Abstract
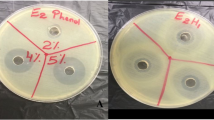
Propolis is a material manufactured by bees and contains beeswax, bee salivary secretions and plant resins. Propolis preparations have been used for millennia by humans in food, cosmetics and medicines due to its antibacterial effects. Within the hive, propolis plays an important role in bees’ health, with much of its bioactivity largely dependent on the plant resins the bees select for its production. Few chemical studies are available on the chemistry of propolis produced by Australian honeybees (Apis mellifera, Apidae). This study aimed to determine the chemical composition as well as in vitro antimicrobial effects of propolis harvested from honeybees in subtropical eastern Australia. Honeybee propolis was produced using plastic frames and multiple beehives in two subtropical sites in eastern Australia. Methanolic extracts of propolis were analysed by liquid chromatography with ultraviolet detection and high-resolution mass spectrometry (ultra-high-pressure liquid chromatography (UHPLC)-UV-high-resolution tandem mass spectrometry (HR-MS/MS)) and by gas chromatography mass spectrometry (GC-MS). The resulting chemical data were dereplicated for compound characterisation. The two crude extracts in abs. ethanol were tested in vitro by the agar diffusion and broth dilution methods, using a phenol standard solution as the positive control and abs. ethanol as the negative control. Chemical constituents were identified to be pentacyclic triterpenoids and C-prenylated flavonoids, including Abyssinoflavanone VII, Propolin C and Nymphaeol C. The two propolis crude extracts showed bactericidal effects at the minimal inhibitory concentrations of 0.37–2.04 mg mL−1 against Staphylococcus aureus ATCC 25923. However, the extracts were inactive against Klebsiella pneumoniae ATCC 13883 and Candida albicans ATCC 10231. The antistaphylococcal potential of propolis was discussed, also in relation to honeybees’ health, as it warrants further investigations on the social and individual immunities of Australian honeybees.


Similar content being viewed by others
References
Abu-Mellal A, Koolaji N et al (2012) Prenylated cinnamate and stilbenes from Kangaroo Island propolis and their antioxidant activity. Phys Chem Chem Phys 77:251–259. doi:10.1016/j.phytochem.2012.01.012
Banskota AH, Tezuka Y et al (2001) Hepatoprotective and anti-Helicobacter pylori activities of constituents from Brazilian propolis. Phytomedicine 8(1):16–23. doi:10.1078/0944-7113-00004
Berenbaum MR, Johnson RM (2015) Xenobiotic detoxification pathways in honey bees. Curr Opin Insect Sci 10:51–58
Chen C-N, Wu C-L et al (2004) Propolin C from propolis induces apoptosis through activating caspases, Bid and cytochrome C release in human melanoma cells. Biochem Pharm 67(1):53–66
Chen YW, Wu SW et al (2008) Characterisation of Taiwanese propolis collected from different locations and seasons. J Sci Food Agric 88(3):412–419
Cui L, Ndinteh DT et al (2007) Isoprenylated Flavonoids from the Stem Bark of Erythrina abyssinica. J Nat Prod 70(6):1039–1042
de Rijke E, Out P et al (2006) Analytical separation and detection methods for flavonoids. J Chromatogr A 1112(1):31–63
Drescher N, Wallace HM et al (2014) Diversity matters: how bees benefit from different resin sources. Oecologia 176(4):943–953
Freitas SF, Shinohara L et al (2006) In vitro effects of propolis on Giardia duodenalis trophozoites. Phytomedicine 13(3):170–175. doi:10.1016/j.phymed.2004.07.008
Ghisalberti E, Jefferies P et al (1978) Constituents of propolis. Cell Mol Life Sci 34(2):157–158
Greco MK, Hoffmann D et al (2010) The alternative Pharaoh approach: stingless bees mummify beetle parasites alive. Naturwissenschaften 97(3):319–323
Inui S, Hosoya T et al (2012a) Solophenols B–D and solomonin: new prenylated polyphenols isolated from propolis collected from the Solomon Islands and their antibacterial activity. J Agric Food Chem 60(47):11765–11770
Inui S, Shimamura Y et al (2012b) A new prenylflavonoid isolated from propolis collected in the Solomon Islands. Biosci Biotechnol Biochem 76(5):1038–1040
Kumazawa S, Hamasaka T et al (2004) Antioxidant activity of propolis of various geographic origins. Food Chem 84(3):329–339
Kumazawa S, Ueda R et al (2007) Antioxidant prenylated flavonoids from propolis collected in Okinawa, Japan. J Agric Food Chem 55(19):7722–7725
Kumazawa S, Nakamura J et al (2008) Plant origin of Okinawan propolis: honeybee behavior observation and phytochemical analysis. Naturwissenschaften 95(8):781–786. doi:10.1007/s00114-008-0383-y
Kumazawa S, Murase M et al (2014) Analysis of antioxidant prenylflavonoids in different parts of Macaranga tanarius, the plant origin of Okinawan propolis. Asian Pac J Trop Med 7(1):16–20
Marquez Hernandez I, Cuesta-Rubio O et al (2010) Studies on the constituents of yellow Cuban propolis: GC-MS determination of triterpenoids and flavonoids. J Agric Food Chem 58(8):4725–4730
Massaro FC, Brooks PR et al (2011) Cerumen of Australian stingless bees (Tetragonula carbonaria): gas chromatography–mass spectrometry fingerprints and potential anti-inflammatory properties. Naturwissenschaften 98(4):329–337
Massaro C, Katouli M et al (2014a) Anti-staphylococcal activity of C-methyl flavanones from propolis of Australian stingless bees (Tetragonula carbonaria) and fruit resins of Corymbia torelliana (Myrtaceae). Fitoterapia 95:247–257
Massaro CF, Shelley D et al (2014b) In vitro antibacterial phenolic extracts from “sugarbag” pot-honeys of Australian stingless bees (Tetragonula carbonaria). J Agric Food Chem 62(50):12209–12217
Massaro CF, Smyth TJ et al (2015) Phloroglucinols from anti‐microbial deposit‐resins of Australian stingless bees (Tetragonula carbonaria). Phytother Res 29(1):48–58
Mathe C, Culioli G et al (2004) Characterization of archaeological frankincense by gas chromatography–mass spectrometry. J Chromatogr A 1023(2):277–285
Melliou E, Stratis E et al (2007) Volatile constituents of propolis from various regions of Greece—antimicrobial activity. Food Chem 103(2):375–380
Miorin P, Levy Junior N et al (2003) Antibacterial activity of honey and propolis from Apis mellifera and Tetragonisca angustula against Staphylococcus aureus. J Appl Micro 95(5):913–920
Mutai C, Abatis D et al (2004) Cytotoxic lupane-type triterpenoids from Acacia mellifera. Phys Chem Chem Phys 65(8):1159–1164
Ponte F, Silva A et al (2008) Bee-honey, propolis and Eucalyptus globulus extract: preclinical toxicity study in rodents. Pharmacogn Mag 4(16):278
Popova M, Silici S et al (2005) Antibacterial activity of Turkish propolis and its qualitative and quantitative chemical composition. Phytomedicine 12(3):221–228. doi:10.1016/j.phymed.2003.09.007
Popova M, Dimitrova R et al (2013) Omani propolis: chemical profiling, antibacterial activity and new propolis plant sources. Chem Centr J 7:158–165
Popova M, Reyes M, et al. (2014). Propolis chemical composition and honeybee resistance against Varroa destructor. Nat Prod Res(ahead-of-print): 1–7
Raghukumar R, Vali L et al (2010) Antimethicillin‐resistant Staphylococcus aureus (MRSA) activity of ‘pacific propolis’ and isolated prenylflavanones. Phytother Res 24(8):1181–1187
Righi AA, Alves TR et al (2011) Brazilian red propolis: unreported substances, antioxidant and antimicrobial activities. J Sci Food Agric 91(13):2363–2370
Simone-Finstrom M, Spivak M (2010) Propolis and bee health: the natural history and significance of resin use by honey bees. Apidologie 41(3):295–311
Simone-Finstrom MD, Spivak M (2012) Increased resin collection after parasite challenge: a case of self-medication in honey bees. Plos One 7(3):e34601
Tran VH, Duke RK et al (2012) Propolis with high flavonoid content collected by honey bees from Acacia paradoxa. Phys Chem Chem Phys 81:126–132
Trusheva B, Popova M et al (2011) Indonesian propolis: chemical composition, biological activity and botanical origin. Nat Prod Rep 25(6):606–613
Zhang T, Omar R et al (2014) Chromatographic analysis with different detectors in the chemical characterisation and dereplication of African propolis. Talanta 120:181–190
Acknowledgments
The authors are thankful to Terry Braggins at Analytica Laboratories Ltd, NZ, for HR-MS analyses, to the Craig’s members of ‘Valley Bees’, Gympie, for beekeeping help and to Michael Howes at ‘Tyagarah Apiaries’ and Dieter Horstmann at ‘Eagle Farm’ Tyagarah, NSW, Australia, for supplying honeybee propolis from their beehives.
Funding
This study and CFM were financed through the University of the Sunshine Coast studentships.
Authors’ contributions
Conceived and designed the experiments: CFM. Performed the experiments: CFM, JS and DP. Analysed the data: CFM. Contributed reagents/materials: PB. Wrote the manuscript: CFM, JS and PB.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that the experiments described in this article comply with the current laws for the conduct of scientific research in Australia.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by: Sven Thatje
Electronic supplementary material
The online version of this article contains supplementary materials (UV spectra and diagnostic fragments for compound characterisation), which is available to authorised users.
ESM 1
(DOC 3966 kb)
Rights and permissions
About this article
Cite this article
Massaro, C.F., Simpson, J.B., Powell, D. et al. Chemical composition and antimicrobial activity of honeybee (Apis mellifera ligustica) propolis from subtropical eastern Australia. Sci Nat 102, 68 (2015). https://doi.org/10.1007/s00114-015-1318-z
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00114-015-1318-z