Abstract
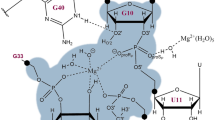
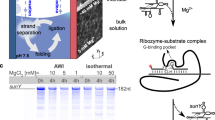
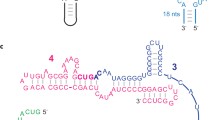
The “RNA world” hypothesis proposes that—early in the evolution of life—RNA molecules played important roles both in information storage and in enzymatic functions. However, this hypothesis seems to be inconsistent with the concept that life may have emerged under hydrothermal conditions since RNA molecules are considered to be labile under such extreme conditions. Presently, the possibility that the last common ancestor of the present organisms was a hyperthermophilic organism which is important to support the hypothesis of the hydrothermal origin of life has been subject of strong discussions. Consequently, it is of importance to study the behavior of RNA molecules under hydrothermal conditions from the viewpoints of stability, catalytic functions, and storage of genetic information of RNA molecules and determination of the upper limit of temperature where life could have emerged. In the present work, self-cleavage of a natural hammerhead ribozyme was examined at temperatures 10–200 °C. Self-cleavage was investigated in the presence of Mg2+, which facilitates and accelerates this reaction. Self-cleavage of the hammerhead ribozyme was clearly observed at temperatures up to 60 °C, but at higher temperatures self-cleavage occurs together with hydrolysis and with increasing temperature hydrolysis becomes dominant. The influence of the amount of Mg2+ on the reaction rate was also investigated. In addition, we discovered that the reaction proceeds in the presence of high concentrations of monovalent cations (Na+ or K+), although very slowly. Furthermore, at high temperatures (above 60 °C), monovalent cations protect the ribozyme against degradation.








Similar content being viewed by others
References
Boussau B, Blanquart S, Necsulea A, Lartillot N, Gouy M (2008) Parallel adaptations to high temperatures in the Archaean eon. Nature 456:942–945
Cech TR (1986) A model for the RNA-catalyzed replication of RNA. Proc Natl Acad Sci USA 83:4360–4363
Chval Z, Chvalova D, Leclerc F (2011) Modeling the RNA 2′OH activation: possible roles of metal ion and nucleobase as catalysts in self-cleaving ribozymes. J Phys Chem B 115:10943–10956
Cochrane JC, Strobel SA (2008) Catalytic strategies of self-cleaving ribozymes. Acc Chem Res 41:1027–1035
Corliss JB, Baross JA, Hoffman SE (1981) An hypothesis concerning the relationship between submarine hot springs and the origin of life on Earth. Oceanol Acta (Suppl) 4:59–69
Curtis EA, Bartel DP (2001) The hammerhead cleavage reaction in monovalent cations. RNA 7:546–552
Daròs JA, Elena SF, Flores R (2006) Viroids: an Ariadne’s thread into the RNA labyrinth. EMBO Rep 7:593–598
Delan-Forino C, Maurel MC, Torchet C (2011) Replication of Avocado Sunblotch Viroid in the yeast Saccharomyces cerevisiae. J Virology 85:3229–3238
Di Giulio M (2000) The late stage of genetic code structuring took place at a high temperature. Gene 261:189–195
Di Giulio M (2005) The ocean abysses witnessedthe origine of the genetic code. Gene 346:7–12
Diener TO (2001) The viroid: biological oddity or evolutionary fossil? Adv Virus Res 57:137–184
Dundas I (1998) Was the environment for primordial life hypersaline? Extremophiles 2:375–377
Feig AL, Ammons GE, Uhlenbeck OC (1998) Cryoenzymology of the hammerhead ribozyme. RNA 4:1251–1258
Forterre P (1996) A hot topic: the origin of hyperthermophiles. Cell 85:789–792
Galtier N, Tourasse N, Gouy M (1999) A nonhyperthermophilic common ancestor to extant life forms. Science 283:220–221
Gilbert W (1986) Origin of life: the RNA world. Nature 319:618–618
Góra-Sochacka A (2004) Viroids: unusual small pathogenic RNAs. Acta Biochim Pol 51:587–607
Järvinen P, Oivane M, Lönnberg H (1991) Interconversion and phosphoester hydrolysis of 2′,5′- and 3′,5′-dinucleotide monophosphate: kinetics and mechanisms. J Org Chem 56:5396–5401
Jonson L, Söll D (1971) Temperature dependence of the aminoacylation of tRNA by Bacillus stearothermophilus. Bioplymers 10:2209–2221
Kaddour H, Vergne J, Hervé G, Maurel M-C (2011) High-pressure analysis of a hammerhead ribozyme from Chrysanthemum chlorotic mottle viroid reveals two different populations of self-cleaving molecule. FEBS J 278:3739–3747
Kawamura K (1999) Monitoring of hydrothermal reactions in 3 ms using fused-silica capillary tubing. Chem Lett 28:125–126
Kawamura K (2000) Monitoring hydrothermal reactions on the millisecond time scale using a micro-tube flow reactor and kinetics of ATP hydrolysis for the RNA world hypothesis. Bull Chem Soc Jpn 73:1805–1811
Kawamura K (2003) Kinetic analysis of cleavage of ribose phosphodiester bond within guanine and cytosine rich oligonucleotides and dinucleotides at 65–200°C and its implications on the chemical evolution of RNA. Bull Chem Soc Jpn 76:153–162
Kawamura K (2004) Behavior of RNA under hydrothermal conditions and the origins of life. Int J Astrobiol 3:301–309
Kawamura K (2005) A new probe for the indirect measurement of the conformation and interaction of biopolymers at extremely high temperatures using a capillary flow hydrothermal reactor system for UV–visible spectrophotometry. Anal Chim Acta 543:236–241
Kawamura K, Maeda J (2007) Kinetic analysis of oligo(C) formation from the 5′-monophosphorimidazolide of cytidine with Pb(II) ion catalyst at 10–75°C. Orig Life Evol Biosph 37:153–165
Kawamura K, Maeda J (2008) Kinetics and activation parameter analysis for the prebiotic oligocytidylate formation on Na+-montmorillonite at 0–100°C. J Phys Chem A 112:8015–8023
Kawamura K, Nagayoshi H (2007) Behavior of DNA under hydrothermal conditions with MgCl2 additive using an in situ UV–visible spectrophotometer. Thermochim Acta 466:63–68
Kawamura K, Umehara M (2001) Kinetic analysis of the temperature dependence of the template-directed formation of oligoguanylate from the 5′-phosphorimidazolide of guanosine on a poly(C) template with Zn2+. Bull Chem Soc Jpn 74:927–935
Kawamura K, Kameyama N, Matumoto O (1999) Kinetics of hydrolysis of ribonucleotide polymers in aqueous solution ate elevated temperatures: implications of chemical evolution of RNA and primitive ribonuclease. Viva Origino 27:107–118
Lazcano A, Miller SL (1996) The origin and early evolution of life: prebiotic chemistry, the pre-RNA world, and time. Cell 85:793–798
Marguet E, Forterre P (1994) DNA stability at temperatures typical for hyperthermophiles. Nucleic Acids Res 22:1681–1686
Miller SL, Lazcano A (1995) The origin of life—did it occur at high temperatures? J Mol Evol 41:689–692
Murray JB, Dunham CM, Scott WG (2002) A pH-dependent conformational change, rather than the chemical step, appears to be rate-limiting in the hammerhead ribozyme cleavage reaction. J Mol Biol 315:121–130
Nisbet EG (1986) Origin of life: RNA and hot-water springs. Nature 322:206–206
Orgel LE (2004) Prebiotic chemistry and the origin of the RNA world. Crit Rev Biochem Mol Biol 39:99–123
Pace NR (1991) Origin of life—facing up to the physical setting. Cell 65:531–533
Peracchi A (1999) Origins of the temperature dependence of hammerhead ribozyme catalysis. Nucl Acids Res 27:2875–2882
Russell MJ, Hall AJ (1997) The emergence of life from iron monosulphide bubbles at a submarine hydrothermal redox and pH front. J Geol Soc 154:377–402
Russell MJ, Hall AJ, Cairns-Smith AG, Braterman PS (1988) Submarine hot springs and the origin of life. Nature 336:117–117
Tehei M, Franzetti B, Maurel MC, Vergne J, Hountondji C, Zaccai G (2002) The search for traces of life: the protective effect of salt on biological macromolecules. Extremophiles 6:427–430
White RH (1984) Hydrolytic stability of biomolecules at high temperatures and its implication for life at 250°C. Nature 310:430–432
Zhuo DM, Usman N, Wincott FE, Matulic-Adamic J, Orita M, Zhang LH, Komiyama M, Kumar PKR, Taira K (1996) Evidence for the rate-limiting departure of the 5′-oygen in nonenzymatic and hammerhead ribozyme-catalyzed reactions. J Am Chem Soc 118:5862–5866
Ztouti M, Kaddour H, Miralles F, Simian C, Vergne J, Hervé G, Maurel M-C (2009) Adenine, a hairpin ribozyme cofactor—high-pressure and competition studies. FEBS J 276:2574–2588
Acknowledgments
Nizar El-Murr was the recipient of the Jasso Scholarship for short-term studies in Japan. This research was supported by 21200004 on Innovative Areas from MEXT in Japan, a JSPS Fellowship for Marie-Christine Maurel research in Japan (S10186), and a 20540476 Grant-in-Aid for Scientific Research from JSPS in Japan. We are grateful for EPOV program of CNRS and CNES grants for supporting this work. We also thank Dr. Anne-Lise Haenni for English improvements.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Sven Thatje
Electronic supplementary materials
Gel electrophoresis data and CD spectral changes are available.
ESM 1
(DOC 346 kb)
Rights and permissions
About this article
Cite this article
El-Murr, N., Maurel, MC., Rihova, M. et al. Behavior of a hammerhead ribozyme in aqueous solution at medium to high temperatures. Naturwissenschaften 99, 731–738 (2012). https://doi.org/10.1007/s00114-012-0954-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00114-012-0954-9