Abstract
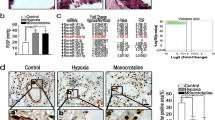
Abnormal pulmonary arterial smooth muscle cells (PASMCs) proliferation is an important pathological process in hypoxic pulmonary arterial hypertension. Mitochondrial dynamics and quality control have a central role in the maintenance of the cell proliferation–apoptosis balance. However, the molecular mechanism is still unknown. We used hypoxic animal models, cell biology, and molecular biology to determine the effect of mitofusin 1 (Mfn1) on hypoxia-mediated PASMCs mitochondrial homeostasis. We found that Mfn1 expression was increased in hypoxia, which was crucial for hypoxia-induced mitochondrial dysfunction and smooth muscle cell proliferation as well as hypoxia-stimulated cell-cycle transition from the G0/G1 phase to S phase. Subsequently, we studied the role of microRNAs in mitochondrial function associated with PASMC proliferation under hypoxic conditions. The promotive effect of Mfn1 on pulmonary vascular remodeling was alleviated in the presence of miR-125a agomir, and miR-125a antagomir mimicked the hypoxic damage effects to mitochondrial homeostasis. Moreover, in vivo and in vitro treatment with miR-125a agomir protected the pulmonary vessels from mitochondrial dysfunction and abnormal remodeling. In the present study, we determined that mitochondrial homeostasis, particularly Mfn1, played an important role in PASMCs proliferation. MiR-125a, an important underlying factor, which inhibited Mfn1 expression and decreased PASMCs disordered growth during hypoxia. These results provide a theoretical basis for the prevention and treatment of pulmonary vascular remodeling.
Key messages
-
Hypoxia leads to upregulation of mitofusin 1 (Mfn1) both in vivo and in vitro.
-
Mfn1 is involved in hypoxia-induced PASMCs proliferation.
-
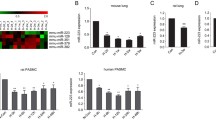
Mfn1-mediated mitochondrial homeostasis is regulated by miR-125a.
-
MiR-125a plays a role in PASMCs oxidative phosphorylation and glycolysis.








Similar content being viewed by others
References
Humbert M, Morrell NW, Archer SL, Stenmark KR, MacLean MR, Lang IM, Christman BW, Weir EK, Eickelberg O, Voelkel NF et al (2004) Cellular and molecular pathobiology of pulmonary arterial hypertension. J Am Coll Cardiol 43(12 Suppl S):13S–24S
Chiong M, Cartes-Saavedra B, Norambuena-Soto I, Mondaca-Ruff D, Morales PE, Garcia-Miguel M, Mellado R (2014) Mitochondrial metabolism and the control of vascular smooth muscle cell proliferation. Front Cell Dev Biol 2:72
Osteryoung KW, Nunnari J (2003) The division of endosymbiotic organelles. Science 302(5651):1698–1704
Moon HE, Paek SH (2015) Mitochondrial dysfunction in Parkinson’s disease. Exp Neurobiol 24(2):103–116
Chen H, McCaffery JM, Chan DC (2007) Mitochondrial fusion protects against neurodegeneration in the cerebellum. Cell 130(3):548–562
Marsboom G, Toth PT, Ryan JJ, Hong Z, Wu X, Fang YH, Thenappan T, Piao L, Zhang HJ, Pogoriler J et al (2013) Dynamin-related protein 1-mediated mitochondrial mitotic fission permits hyperproliferation of vascular smooth muscle cells and offers a novel therapeutic target in pulmonary hypertension. Circ Res 110(11):1484–1497
Chen H, Detmer SA, Ewald AJ, Griffin EE, Fraser SE, Chan DC (2003) Mitofusins Mfn1 and Mfn2 coordinately regulate mitochondrial fusion and are essential for embryonic development. J Cell Biol 160(2):189–200
Pyakurel A, Savoia C, Hess D, Scorrano L (2015) Extracellular regulated kinase phosphorylates mitofusin 1 to control mitochondrial morphology and apoptosis. Mol Cell 58(2):244–254
Park KS, Wiederkehr A, Wollheim CB (2012) Defective mitochondrial function and motility due to mitofusin 1 overexpression in insulin secreting cells. Korean J Physiol Pharmacol 16(1):71–77
Picao-Osorio J, Johnston J, Landgraf M, Berni J, Alonso CR (2015) MicroRNA-encoded behavior in Drosophila. Science. doi:10.1126/science.aad0217
Caruso P, Dempsie Y, Stevens HC, McDonald RA, Long L, Lu R, White K, Mair KM, McClure JD, Southwood M et al (2012) A role for miR-145 in pulmonary arterial hypertension: evidence from mouse models and patient samples. Circ Res 111(3):290–300
Ma C, Li Y, Ma J, Liu Y, Li Q, Niu S, Shen Z, Zhang L, Pan Z, Zhu D (2011) Key role of 15-lipoxygenase/15-hydroxyeicosatetraenoic acid in pulmonary vascular remodeling and vascular angiogenesis associated with hypoxic pulmonary hypertension. Hypertension 58(4):679–688
Furukawa R, Yamada Y, Takenaga M, Igarashi R, Harashima H (2010) Octaarginine-modified liposomes enhance the anti-oxidant effect of lecithinized superoxide dismutase by increasing its cellular uptake. Biochem Biophys Res Commun 404(3):796–801
Mack SC, Agnihotri S, Bertrand KC, Wang X, Shih DJ, Witt H, Hill N, Zayne K, Barszczyk M, Ramaswamy V et al (2015) Spinal myxopapillary ependymomas demonstrate a Warburg phenotype. Clin Cancer Res 21(16):3750–3758
Zhao L, Oliver E, Maratou K, Atanur SS, Dubois OD, Cotroneo E, Chen CN, Wang L, Arce C, Chabosseau PL et al (2015) The zinc transporter ZIP12 regulates the pulmonary vascular response to chronic hypoxia. Nature 524(7565):356–360
Li X, Zhang X, Leathers R, Makino A, Huang C, Parsa P, Macias J, Yuan JX, Jamieson SW, Thistlethwaite PA (2009) Notch3 signaling promotes the development of pulmonary arterial hypertension. Nat Med 15(11):1289–1297
Romaine SP, Tomaszewski M, Condorelli G, Samani NJ (2013) MicroRNAs in cardiovascular disease: an introduction for clinicians. Heart 101(12):921–928
Yin Y, Wu X, Yang Z, Zhao J, Wang X, Zhang Q, Yuan M, Xie L, Liu H, He Q (2013) The potential efficacy of R8-modified paclitaxel-loaded liposomes on pulmonary arterial hypertension. Pharm Res 30(8):2050–2062
Biswas S, Deshpande PP, Perche F, Dodwadkar NS, Sane SD, Torchilin VP (2013) Octa-arginine-modified pegylated liposomal doxorubicin: an effective treatment strategy for non-small cell lung cancer. Cancer Lett 335(1):191–200
Fike CD, Dikalova A, Kaplowitz MR, Cunningham G, Summar M, Aschner JL (2015) Rescue treatment with L-citrulline inhibits hypoxia-induced pulmonary hypertension in newborn pigs. Am J Respir Cell Mol Biol 53(2):255–264
Arnold B, Cassady SJ, VanLaar VS, Berman SB (2010) Integrating multiple aspects of mitochondrial dynamics in neurons: age-related differences and dynamic changes in a chronic rotenone model. Neurobiol Dis 41(1):189–200
Lugus JJ, Ngoh GA, Bachschmid MM, Walsh K (2011) Mitofusins are required for angiogenic function and modulate different signaling pathways in cultured endothelial cells. J Mol Cell Cardiol 51(6):885–893
Park YY, Nguyen OT, Kang H, Cho H (2014) MARCH5-mediated quality control on acetylated Mfn1 facilitates mitochondrial homeostasis and cell survival. Cell Death Dis 5:e1172
Chen H, Vermulst M, Wang YE, Chomyn A, Prolla TA, McCaffery JM, Chan DC (2010) Mitochondrial fusion is required for mtDNA stability in skeletal muscle and tolerance of mtDNA mutations. Cell 141(2):280–289
Kim KY, Perkins GA, Shim MS, Bushong E, Alcasid N, Ju S, Ellisman MH, Weinreb RN, Ju WK (2015) DRP1 inhibition rescues retinal ganglion cells and their axons by preserving mitochondrial integrity in a mouse model of glaucoma. Cell Death Dis 6:e1839
Bui HT, Shaw JM (2013) Dynamin assembly strategies and adaptor proteins in mitochondrial fission. Curr Biol 23(19):R891–R899
Zhang D, Ma C, Li S, Ran Y, Chen J, Lu P, Shi S, Zhu D (2012) Effect of mitofusin 2 on smooth muscle cells proliferation in hypoxic pulmonary hypertension. Microvasc Res 84(3):286–296
Ishihara N, Eura Y, Mihara K (2004) Mitofusin 1 and 2 play distinct roles in mitochondrial fusion reactions via GTPase activity. J Cell Sci 117(Pt 26):6535–6546
Song Z, Ghochani M, McCaffery JM, Frey TG, Chan DC (2009) Mitofusins and OPA1 mediate sequential steps in mitochondrial membrane fusion. Mol Biol Cell 20(15):3525–3532
Lin CJ, Lee CC, Shih YL, Lin CH, Wang SH, Chen TH, Shih CM (2012) Inhibition of mitochondria- and endoplasmic reticulum stress-mediated autophagy augments temozolomide-induced apoptosis in glioma cells. PLoS One 7(6):e38706
Ryan JJ, Marsboom G, Fang YH, Toth PT, Morrow E, Luo N, Piao L, Hong Z, Ericson K, Zhang HJ et al (2013) PGC1alpha-mediated mitofusin-2 deficiency in female rats and humans with pulmonary arterial hypertension. Am J Respir Crit Care Med 187(8):865–878
Fang X, Chen X, Zhong G, Chen Q, Hu C (2016) Mitofusin 2 downregulation triggers pulmonary artery smooth muscle cell proliferation and apoptosis imbalance in rats with hypoxic pulmonary hypertension via the PI3K/Akt and mitochondrial apoptosis pathways. J Cardiovasc Pharmacol 67(2):164–174
Lou Y, Li R, Liu J, Zhang Y, Zhang X, Jin B, Liu Y, Wang Z, Zhong H, Wen S et al (2015) Mitofusin-2 over-expresses and leads to dysregulation of cell cycle and cell invasion in lung adenocarcinoma. Med Oncol 32(4):132
Ding Y, Gao H, Zhao L, Wang X, Zheng M (2015) Mitofusin 2-deficiency suppresses cell proliferation through disturbance of autophagy. PLoS One 10(3):e0121328
Estela A, Pla-Martín D, Sánchez-Piris M, Sesaki H, Palau F (2011) Charcot-Marie-Tooth-related gene GDAP1 complements cell cycle delay at G2/M phase in Saccharomyces cerevisiae fis1 gene-defective cells. J Biol Chem 286(42):36777–36786
Losón OC, Song Z, Chen H, Chan DC (2013) Fis1, Mff, MiD49, and MiD51 mediate Drp1 recruitment in mitochondrial fission. Mol Biol Cell 24(5):659–667
Renault TT, Floros KV, Elkholi R, Corrigan KA, Kushnareva Y, Wieder SY, Lindtner C, Serasinghe MN, Asciolla JJ, Buettner C et al (2015) Mitochondrial shape governs BAX-induced membrane permeabilization and apoptosis. Mol Cell 57(1):69–82
Schrepfer E, Scorrano L (2016) Mitofusins, from mitochondria to metabolism. Mol Cell 61(5):683–694
Bach D, Pich S, Soriano FX, Vega N, Baumgartner B, Oriola J, Daugaard JR, Lloberas J, Camps M, Zierath JR et al (2003) Mitofusin-2 determines mitochondrial network architecture and mitochondrial metabolism. J Biol Chem 278(19):17190–17197
Delettre C, Griffoin JM, Kaplan J, Dollfus H, Lorenz B, Faivre L, Lenaers G, Belenguer P, Hamel CP (2001) Mutation spectrum and splicing variants in the OPA1 gene. Hum Genet 109(6):584–591
Lee S, Jeong SY, Lim WC, Kim S, Park YY, Sun X, Youle RJ, Cho H (2007) Mitochondrial fission and fusion mediators, hFis1 and OPA1. Modulate Cellular Senescence 282(31):22977–22983
Ambros V (2004) The functions of animal microRNAs. Nature 431(7006):350–355
McDonald RA, Hata A, MacLean MR, Morrell NW, Baker AH (2012) MicroRNA and vascular remodelling in acute vascular injury and pulmonary vascular remodelling. Cardiovasc Res 93(4):594–604
Nishida N, Mimori K, Fabbri M, Yokobori T, Sudo T, Tanaka F, Shibata K, Ishii H, Doki Y, Mori M (2011) MicroRNA-125a-5p is an independent prognostic factor in gastric cancer and inhibits the proliferation of human gastric cancer cells in combination with trastuzumab. Clin Cancer Res 17(9):2725–2733
Jiang L, Huang Q, Zhang S, Zhang Q, Chang J, Qiu X, Wang E (2010) Hsa-miR-125a-3p and hsa-miR-125a-5p are downregulated in non-small cell lung cancer and have inverse effects on invasion and migration of lung cancer cells. BMC Cancer 10:318
Huber LC, Ulrich S, Leuenberger C, Gassmann M, Vogel J, von Blotzheim LG, Speich R, Kohler M, Brock M (2015) Featured article: microRNA-125a in pulmonary hypertension: regulator of a proliferative phenotype of endothelial cells. Exp Biol Med (Maywood) 240(12):1580–1589
Green DE, Murphy TC, Kang BY, Kleinhenz JM, Szyndralewiez C, Page P, Sutliff RL, Hart CM (2011) The Nox4 inhibitor GKT137831 attenuates hypoxia-induced pulmonary vascular cell proliferation. Am J Respir Cell Mol Biol 47(5):718–726
Crossno JT, Garat CV, Reusch JE, Morris KG, Dempsey EC, McMurtry IF, Stenmark KR, Klemm DJ (2007) Rosiglitazone attenuates hypoxia-induced pulmonary arterial remodeling. Am J Physiol Lung Cell Mol Physiol 292(4):L885–L897
Bauer PM, Bauer EM, Rogers NM, Yao M, Feijoo-Cuaresma M, Pilewski JM, Champion HC, Zuckerbraun BS, Calzada MJ, Isenberg JS (2012) Activated CD47 promotes pulmonary arterial hypertension through targeting caveolin-1. Cardiovasc Res 93(4):682–693
Acknowledgements
This work was supported by the National Natural Science Foundation of China [31471095, 81270113, and 81400353]; the Key Research Plan of National Natural Science Foundation of China [91339107]; the Natural Science Foundation of Heilongjiang Province [QC2014C096]; the Wu Liande Youth Science Foundation [WLD-QN1410]; the Postdoctoral Foundation of Heilongjiang Province [LBH-Z14133]; and the Postdoctoral Foundation of China [2015m571438].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All animal care and experimental procedures were performed in accordance with relevant guidelines and regulations and approved by the Institutional Animal Care and Use Committee (IACUC) of Harbin Medical University.
Conflict of interests
The authors declare that they have no conflict of interests.
Electronic supplementary material
ESM 1
(PDF 418 kb)
Rights and permissions
About this article
Cite this article
Ma, C., Zhang, C., Ma, M. et al. MiR-125a regulates mitochondrial homeostasis through targeting mitofusin 1 to control hypoxic pulmonary vascular remodeling. J Mol Med 95, 977–993 (2017). https://doi.org/10.1007/s00109-017-1541-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-017-1541-5