Abstract
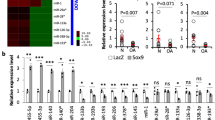
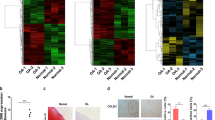
Cartilage degeneration in the course of osteoarthritis (OA) is associated with an alteration in chondrocyte metabolism. In order to identify molecules representing putative key regulators for diagnosis and therapeutic intervention, we analyzed gene expression and microRNA (miR) levels in OA and normal knee cartilage using a customized cartilage cDNA array and quantitative RT-PCR. Among newly identified candidate molecules, H19, IGF2, and ITM2A were significantly elevated in OA compared to normal cartilage. H19 is an imprinted maternally expressed gene influencing IGF2 expression, whose transcript is a long noncoding (lnc) RNA of unknown biological function harboring the miR-675. H19 and IGF2 mRNA levels did not correlate significantly within cartilage samples suggesting that deregulation by imprinting effects are unlikely. A significant correlation was, however, observed for H19, COL2A1, and miR-675 expression levels in OA tissue, and functional regulation of these candidate molecules was assessed under anabolic and catabolic conditions. Culture of chondrocytes under hypoxic signaling showed co-upregulation of H19, COL2A1, and miRNA-675 levels in close correlation. Proinflammatory cytokines IL-1β and TNF-α downregulated COL2A1, H19, and miR-675 significantly without close statistical correlation. In conclusion, this is the first report demonstrating deregulation of an lncRNA and its encoded miR in the context of OA-affected cartilage. Stress-induced regulation of H19 expression by hypoxic signaling and inflammation suggests that lncRNA H19 acts as a metabolic correlate in cartilage and cultured chondrocytes, while the miR-675 may indirectly influence COL2A1 levels. H19 may not only be an attractive marker for cell anabolism but also a potential target to stimulate cartilage recovery.




Similar content being viewed by others
References
Sandell LJ, Aigner T (2001) Articular cartilage and changes in arthritis. An introduction: cell biology of osteoarthritis. Arthritis Res 2:107–113
Goldring MB, Marcu KB (2009) Cartilage homeostasis in health and rheumatic diseases. Arthritis Res Ther 3:224
Boeuf S, Steck E, Pelttari K et al (2008) Subtractive gene expression profiling of articular cartilage and mesenchymal stem cells: serpins as cartilage-relevant differentiation markers. Osteoarthr Cartil 1:48–60
Aigner T, Zien A, Gehrsitz A et al (2001) Anabolic and catabolic gene expression pattern analysis in normal versus osteoarthritic cartilage using complementary DNA-array technology. Arthritis Rheum 12:2777–2789
Brew CJ, Clegg PD, Boot-Handford RP et al (2010) Gene expression in human chondrocytes in late osteoarthritis is changed in both fibrillated and intact cartilage without evidence of generalised chondrocyte hypertrophy. Ann Rheum Dis 1:234–240
Aigner T, Fundel K, Saas J et al (2006) Large-scale gene expression profiling reveals major pathogenetic pathways of cartilage degeneration in osteoarthritis. Arthritis Rheum 11:3533–3544
Tusher VG, Tibshirani R, Chu G (2001) Significance analysis of microarrays applied to the ionizing radiation response. Proc Natl Acad Sci U S A 9:5116–5121
Han J, Yang T, Gao J et al (2010) Specific microRNA expression during chondrogenesis of human mesenchymal stem cells. Int J Mol Med 3:377–384
Fehrenbacher A, Steck E, Roth W et al (2006) Long-term mechanical loading of chondrocyte-chitosan biocomposites in vitro enhanced their proteoglycan and collagen content. Biorheology 6:709–720
Hu SI, Carozza M, Klein M et al (1998) Human HtrA, an evolutionarily conserved serine protease identified as a differentially expressed gene product in osteoarthritic cartilage. J Biol Chem 51:34406–34412
Cai X, Cullen BR (2007) The imprinted H19 noncoding RNA is a primary microRNA precursor. RNA 3:313–316
Dudek KA, Lafont JE, Martinez-Sanchez A et al (2010) Type II collagen expression is regulated by tissue-specific miR-675 in human articular chondrocytes. J Biol Chem 32:24381–24387
Duval E, Leclercq S, Elissalde JM et al (2009) Hypoxia-inducible factor 1alpha inhibits the fibroblast-like markers type I and type III collagen during hypoxia-induced chondrocyte redifferentiation: hypoxia not only induces type II collagen and aggrecan, but it also inhibits type I and type III collagen in the hypoxia-inducible factor 1alpha-dependent redifferentiation of chondrocytes. Arthritis Rheum 10:3038–3048
Weinreb O, Amit T, Mandel S et al (2010) Neuroprotective multifunctional iron chelators: from redox-sensitive process to novel therapeutic opportunities. Antioxid Redox Signal 6:919–949
Aigner T, Gebhard PM, Schmid E et al (2003) SOX9 expression does not correlate with type II collagen expression in adult articular chondrocytes. Matrix Biol 4:363–372
Fan Z, Bau B, Yang H et al (2005) Freshly isolated osteoarthritic chondrocytes are catabolically more active than normal chondrocytes, but less responsive to catabolic stimulation with interleukin-1beta. Arthritis Rheum 1:136–143
Cui H (2007) Loss of imprinting of IGF2 as an epigenetic marker for the risk of human cancer. Dis Markers 1–2:105–112
Fu VX, Dobosy JR, Desotelle JA et al (2008) Aging and cancer-related loss of insulin-like growth factor 2 imprinting in the mouse and human prostate. Cancer Res 16:6797–6802
Lipovich L, Johnson R, Lin CY (2010) MacroRNA underdogs in a microRNA world: evolutionary, regulatory, and biomedical significance of mammalian long non-protein-coding RNA. Biochim Biophys Acta 9:597–615
Goshen R, Rachmilewitz J, Schneider T et al (1993) The expression of the H-19 and IGF-2 genes during human embryogenesis and placental development. Mol Reprod Dev 4:374–379
Tanos V, Prus D, Ayesh S et al (1999) Expression of the imprinted H19 oncofetal RNA in epithelial ovarian cancer. Eur J Obstet Gynecol Reprod Biol 1:7–11
Leibovitch MP, Nguyen VC, Gross MS et al (1991) The human ASM (adult skeletal muscle) gene: expression and chromosomal assignment to 11p15. Biochem Biophys Res Commun 3:1241–1250
Matouk IJ, Degroot N, Mezan S et al (2007) The H19 non-coding RNA is essential for human tumor growth. PLoS One 9:e845
Hao Y, Crenshaw T, Moulton T et al (1993) Tumour-suppressor activity of H19 RNA. Nature 6448:764–767
Owen RD, Hosoi J, Montgomery JC et al (1993) Coordinate regulation of collagen II(alpha 1) and H19 expression in immortalized hamster cells. Cell Growth Differ 12:1013–1021
Ripoche MA, Kress C, Poirier F et al (1997) Deletion of the H19 transcription unit reveals the existence of a putative imprinting control element. Genes Dev 12:1596–1604
Smits G, Mungall AJ, Griffiths-Jones S et al (2008) Conservation of the H19 noncoding RNA and H19-IGF2 imprinting mechanism in therians. Nat Genet 8:971–976
Gordeladze JO, Djouad F, Brondello JM et al (2009) Concerted stimuli regulating osteo-chondral differentiation from stem cells: phenotype acquisition regulated by microRNAs. Acta Pharmacol Sin 10:1369–1384
Sheedy FJ, O'Neill LA (2008) Adding fuel to fire: microRNAs as a new class of mediators of inflammation. Ann Rheum Dis 3:iii50–iii55
Kulshreshtha R, Davuluri RV, Calin GA et al (2008) A microRNA component of the hypoxic response. Cell Death Differ 4:667–671
Carthew RW (2006) Gene regulation by microRNAs. Curr Opin Genet Dev 2:203–208
Jones SW, Watkins G, Le Good N et al (2009) The identification of differentially expressed microRNA in osteoarthritic tissue that modulate the production of TNF-alpha and MMP13. Osteoarthr Cartil 4:464–472
Yamasaki K, Nakasa T, Miyaki S et al (2009) Expression of microRNA-146a in osteoarthritis cartilage. Arthritis Rheum 4:1035–1041
Akhtar N, Rasheed Z, Ramamurthy S et al (2010) MicroRNA-27b regulates the expression of matrix metalloproteinase 13 in human osteoarthritis chondrocytes. Arthritis Rheum 5:1361–1371
Miyaki S, Nakasa T, Otsuki S et al (2009) MicroRNA-140 is expressed in differentiated human articular chondrocytes and modulates interleukin-1 responses. Arthritis Rheum 9:2723–2730
Scholer N, Langer C, Dohner H et al (2010) Serum microRNAs as a novel class of biomarkers: a comprehensive review of the literature. Exp Hematol 12:1126–1130
Acknowledgments
The authors thank Michaela Burkhardt for excellent technical assistance and Simone Gantz for professional statistical support. This work was supported by the German Research Foundation (DFG grant Ri707/7-1), German Ministry of Education and Research (BMBF grant 0315579), and the Orthopaedic University Hospital Heidelberg.
Conflict of interest
Authors disclose any commercial or other potential conflict of interest regarded to this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Steck, E., Boeuf, S., Gabler, J. et al. Regulation of H19 and its encoded microRNA-675 in osteoarthritis and under anabolic and catabolic in vitro conditions. J Mol Med 90, 1185–1195 (2012). https://doi.org/10.1007/s00109-012-0895-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-012-0895-y