Abstract
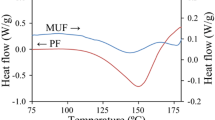
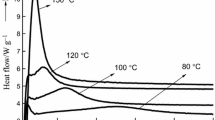
The cure properties of cure-accelerated phenol-urea-formaldehyde (PUF) resins with different catalysts [calcium oxide (CaO), sodium carbonate (Na2CO3), zinc oxide (ZnO), and magnesium oxide (MgO)] were investigated by gelation test and differential scanning calorimetry (DSC) analysis. The results indicated that catalysts such as Na2CO3, ZnO, and MgO were capable of increasing the curing rate and decreasing the curing temperature of PUF resins, however, the CaO inhibited the cure reaction. The formation of methylene bridges was considered to be the main reaction during curing. For the ZnO- and MgO-accelerated PUF resins, the addition reaction of formaldehyde with free phenolic site may act as subsidiary reaction. The activation energies (E a ) of cure-accelerated PUF resins other than CaO-acceleration were much lower than the control resin. The effects of catalysts and hot press temperature on adhesive performances of PUF resins were also discussed by plywood test. The PUF resins with Na2CO3, ZnO, and MgO had higher wet shear strength than the control resin. Hot press temperature had a strong influence on the wet shear strength as well as the catalysts. Among the catalysts, MgO had more significant improving effect on both the curing process and the wet shear strength of PUF resin.
Zusammenfassung
Mittels Geliertest und Differentialrasterkalorimetrie (DSC) wurden die Aushärtungseigenschaften von Phenol-Harnstoff-Formaldehydharzen (PUF), denen verschiedene Katalysatoren (Calciumoxid (CaO), Natriumcarbonat (Na2CO3), Zinkoxid (ZnO) und Magnesiumoxid (MgO)) als Härtungsbeschleuniger zugesetzt wurden, untersucht. Die Ergebnisse zeigten, dass die Katalysatoren Na2CO3, ZnO und MgO in der Lage sind, die Aushärtungsgeschwindigkeit zu erhöhen und die Aushärtungstemperatur von PUF Harzen zu senken, wohingegen CaO die Aushärtungsreaktion hemmte. Die Bildung von Methylenbrücken wurde als Hauptreaktion bei der Aushärtung angesehen. Bei den mit ZnO- und MgO beschleunigten PUF-Harzen kann die Anlagerung von Formaldehyd an die freien phenolischen OH-Gruppen eine Nebenreaktion darstellen. Die Aktivierungsenergien (E a ) von mit Härtungsbeschleunigern versetzen PUF-Harzen waren, außer bei CaO, viel niedriger als die des Kontrollharzes. Der Einfluss der Katalysatoren und der Heißpresstemperatur auf das Klebstoffverhalten von PUF-Harzen wurde auch an Sperrholz geprüft. PUF-Harze mit zugesetztem Na2CO3, ZnO und MgO wiesen eine höhere Nassscherfestigkeit als das Kontrollharz auf. Die Heißpresstemperatur hatte ebenfalls einen starken Einfluss auf die Nassscherfestigkeit. Unter allen Katalysatoren hatte MgO den größten positiven Einfluss auf sowohl das Aushärten als auch die Nassscherfestigkeit des PUF-Harzes.


Similar content being viewed by others
References
Christiansen AW, Gollob L (2003) Differential scanning calorimetry of phenol-formaldehyde resols. J Appl Polym Sci 30(6):2279–2289
Effendi A, Gerhauser H, Bridgwater AV (2008) Production of renewable phenolic resins by thermochemical conversion of biomass: a review. Renew Sustain Energy Rev 12(8):2092–2116
Fan DB, Chang JM, Li JZH, Mao A, Zhang LT (2009a) 13C-NMR Study on the Structure of Phenol-Urea-Formaldehyde Resins Prepared by Methylolureas and Phenol. J Appl Polym Sci 112(4):2195–2202
Fan DB, Li JZH, Chang JM, Gou JSH, Jiang JX (2009b) Chemical structure and curing behavior of phenol-urea-formaldehyde cocondensed resins of high urea content. J Adhes Sci Technol 23(13–14):1787–1797
Fan DB, Li JZH, Chang JM (2009c) On the structure and cure acceleration of phenol-urea-formaldehyde resins with different catalysts. Eur Polym J 45(10):2849–2857
Fraser DA, Hall RW, Raum AL (1957) Preparation of ‘high-ortho’ novolak resins I. Metal ion catalysis and orientation effect. J Appl Chem 7(12):676–689
He GB, Riedl B (2003) Phenol-urea-formaldehyde cocondensed resol resins: their synthesis, curing kinetics, and network properties. J Polym Sci, Part B 41(16):1929–1938
He GB, Yan N (2005) Influence of the synthesis conditions on the curing behavior of phenol–urea–formaldehyde resol resins. J Appl Polym Sci 95(6):1368–1375
Kissinger HE (1957) Reaction kinetics in differential thermal analysis. Anal Chem 29:1702–1706
Klasnja B, Kopitovic S (1992) Lignin-phenol-formaldehyde resins as adhesives in the production of plywood. Holz Roh- Werkst 50(7–8):282–285
Park BD, Riedl B, Hsu E, Shields J (1999) Differential scanning calorimetry of phenol–formaldehyde resins cure-accelerated by carbonates. Polymer 40(7):1689–1699
Pizzi A (1979a) Phenolic and tannin-based adhesive resins by reactions of coordinated metal ligands. I. Phenolic chelates. J Appl Polym Sci 24(5):1247–1255
Pizzi A (1979b) Phenolic and tannin-based adhesive resins by reactions of coordinated metal ligands. II. Tannin adhesive preparation, characteristics, and application. J Appl Polym Sci 24(5):1257–1268
Pizzi A (1994a) Advanced wood adhesives technology. Dekker, New York
Pizzi A (1994b) Handbook of adhesive technology. Dekker, New York
Pizzi A, Garcia R, Wang S (1997) On the networking mechanisms of additives-accelerated phenol–formaldehyde polycondensates. J Appl Polym Sci 66(2):255–266
Tomita B, Hse CY (1992) Cocondensation of urea with methylolphenols in acidic conditions. J Polym Sci, Part A 30(8):1615–1624
Tomita B, Hse CY (1993) Synthesis and structural analysis of cocondensed resins from urea and methylolphenols. Mokuzai Gakkaishi 39:1276–1284
Tomita B, Hse CY (1998) Phenol-urea-formaldehyde (PUF) co-condensed wood adhesives. Int J Adhes Adhes 18(2):69–79
Turunen M, Alvila L, Pakkanen TT, Rainio J (2003) Modification of phenol–formaldehyde resol resins by lignin, starch, and urea. J Appl Polym Sci 88(2):582–588
Vázquez G, López-Suevos F, Villar-Garea A, González-Alvarez J, Antorrena G (2004) 13C-NMR analysis of phenol-urea-formaldehyde prepolymers and phenol-urea-formaldehyde-tannin adhesives. J Adhes Sci Technol 18(13):1529–1543
Vázquez G, López-Suevos F, González-Alvarez J, Antorrena G (2005) Curing process of phenol-urea-formaldehyde-tannin, (PUFT) adhesives: kinetic studies by DSC and DMA. J Therm Anal Calorim 82(1):143–149
Zhao C, Pizzi A, Garnier S (1999) Fast advancement and hardening acceleration of low condensation alkaline PF resins by ester and copolymerized urea. J Appl Polym Sci 74(2):359–378
Zhao C, Pizzi A, Kuhn A, Garnier S (2000) Fast advancement and hardening acceleration of low condensation alkaline phenol-formaldehyde resins by esters and copolymerized urea. II. Esters during resin reaction and effect of guanidine salts. J Appl Polym Sci 77(2):249–259
Acknowledgements
The authors are very grateful for financial support from Chinese National Science and Technology planning (Project 2006BAD07A07-10).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fan, DB., Chang, JM., Li, JZ. et al. Cure properties and adhesive performances of cure-accelerated phenol-urea-formaldehyde resins. Eur. J. Wood Prod. 69, 213–220 (2011). https://doi.org/10.1007/s00107-010-0414-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00107-010-0414-1