Abstract
Background:
The bone substitute NanoBone® consists of nanocrystalline hydroxyapatite embedded in a highly porous matrix of silica gel. It promotes the healing of bone defects and is degraded by osteoclasts during bone remodeling. The present study investigates the interactions of NanoBone® with bone tissue.
Methods:
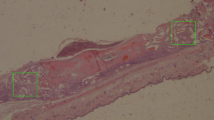
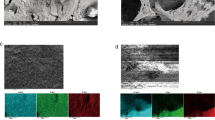
Granules of NanoBone® were implanted in defects of critical size in the mandible of minipigs. Samples were taken after 5 and 10 weeks and demineralized. The composition of the implanted granules was analyzed by means of transmission and scanning electron microscopy and EDX. Enzymeand immunohistochemistry was used to investigate organic components of NanoBone® granules that arised after implantation in the host.
Results:
EDX demonstrated that 5 weeks after implantation the silica gel was degraded and replaced by an organic matrix. Ultrastructurally, the matrix appeared amorphous with only single collagen fibrillae.
PAS-staining indicated the presence of carbohydrates. Immunohistochemically, the bone proteins osteopontin, osteocalcin and BMP-2 were found as constituents of the new matrix. Alkalic phosphatase activity was located in osteoblasts and newly formed bone on NanoBone® and focally in particles. Osteoclasts with ruffled borders, sealing zones, and acid phosphatase activity were situated in resorption lacunae at granule surfaces not covered by new bone.
Conclusions:
In vivo, the silica gel of NanoBone® is replaced by bone matrix glycoproteins with known functions in attraction, adhesion, and differentation of bone cells as osteoblasts and osteoclasts. We assume that the deposition of these molecules supports the early phase of NanoBone® degradation by osteoclasts and promotes the production of new bone tissue.
Similar content being viewed by others
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gerber, T., Holzhüter, G., Götz, W. et al. Nanostructuring of Biomaterials—A Pathway to Bone Grafting Substitute. Eur J Trauma 32, 132–140 (2006). https://doi.org/10.1007/s00068-006-6046-9
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s00068-006-6046-9