Abstract
Objective
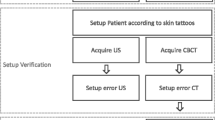
The accuracy of a transperineal three-dimensional ultrasound system (3DUS) was assessed for prostate positioning and compared to fiducial- and bone-based positioning in kV cone beam computed tomography (CBCT) during definitive radiotherapy of prostate cancer.
Methods
Each of the 7 patients had three fiducial markers implanted into the prostate before treatment. Prostate positioning was simultaneously measured by 3DUS and CBCT before each fraction. In total, 177 pairs of 3DUS and CBCT scans were collected. Bone-match and seed-match were performed for each CBCT. Using seed-match as a reference, the accuracy of 3DUS and bone-match was evaluated. Systematic and random errors as well as optimal setup margins were calculated for 3DUS and bone-match.
Results
The discrepancy between 3DUS and seed-match in CBCT (average ± standard deviation) was 0.0 ± 1.7 mm laterally, 0.2 ± 2.0 mm longitudinally, and 0.3 ± 1.7 mm vertically. Using seed-match as a reference, systematic errors for 3DUS were 1.2 mm, 1.1 mm, and 0.9 mm; and random errors were 1.4 mm, 1.8 mm, and 1.6 mm, on lateral, longitudinal, and vertical axes, respectively. By analogy, the difference of bone-match to seed-match was 0.1 ± 1.1 mm laterally, 1.3 ± 3.8 mm longitudinally, and 1.3 ± 4.5 mm vertically. Systematic errors were 0.5 mm, 2.2 mm, and 2.6 mm; and random errors were 1.0 mm, 3.1 mm, and 3.9 mm on lateral, longitudinal, and vertical axes, respectively. The accuracy of 3DUS was significantly higher than that of bone-match on longitudinal and vertical axes, but not on the lateral axis.
Conclusion
Image-guided radiotherapy of prostate cancer based on transperineal 3DUS was feasible, with overall small discrepancy to seed-match in CBCT in this retrospective study. Compared to bone-match, transperineal 3DUS achieved higher accuracy on longitudinal and vertical axes.
Zusammenfassung
Zielsetzung
Bewertung der Genauigkeit eines transperinealen dreidimensionalen Ultraschallsystems (3DUS) für die Prostatapositionierung und Vergleich mit Goldmarker- und knochenbasierter Positionierung in Kilovolt-„Cone-beam“-Computertomographie (CBCT) bei einer definitiven Strahlentherapie des Prostatakarzinoms.
Methoden
Vor Behandlung wurden 7 Patienten je drei Goldmarker in die Prostata implantiert. Die Prostataposition wurde zeitgleich von 3DUS und CBCT vor jeder Fraktion gemessen. Insgesamt wurden 177 Paare von 3DUS- und CBCT-Scans gewonnen. Knochen- und Goldmarker-Koregistrierungen wurden für jede CBCT durchgeführt. Goldmarker-Koregistrierung wurden als Referenz genutzt, um die Genauigkeit der 3DUS und Knochen-Koregistrierung zu bewerten. Systematische und zufällige Fehler sowie optimale Sicherheitsabstände wurden jeweils für 3DUS und Knochen-Koregistrierung berechnet.
Ergebnisse
Die Diskrepanz zwischen 3DUS und Goldmarker-Koregistrierung in CBCT (Mittelwert ± Standardabweichung) betrug lateral 0,0 ± 1,7 mm, longitudinal 0,2 ± 2,0 mm und vertikal 0,3 ± 1,7 mm. Mit Goldmarker-Koregistrierung als Referenz betrugen systematische Fehler für 3DUS 1,2, 1,1 und 0,9 mm, zufällige Fehler 1,4, 1,8 und 1,6 mm jeweils in lateraler, longitudinaler und vertikaler Richtung. In Analogie dazu betrug die Differenz der Knochen-Koregistrierung zur Goldmarker-Koregistrierung 0,1 ± 1,1, 1,3 ± 3,8 und 1,3 ± 4,5 mm in lateraler, longitudinaler und vertikaler Richtung; systematische Fehler waren 0,5, 2,2 und 2,6 mm und zufällige Fehler 1,0, 3,1 und 3,9 mm jeweils in lateraler, longitudinaler und vertikaler Richtung. Die Genauigkeit der 3DUS war signifikant höher als die der Knochen-Koregistrierung in longitudinaler und vertikaler, aber nicht in lateraler Richtung.
Schlussfolgerung
In der retrospektiven Studie war die bildgeführte Strahlentherapie durch den transperinealen 3DUS praktikabel beim Prostatakarzinom mit insgesamt geringer Diskrepanz zu Goldmarker-Koregistrierung in CBCT. Der transperineale 3DUS erzielte eine höhere Genauigkeit in longitudinaler und vertikaler Richtung als die Knochen-Koregistrierung.


Similar content being viewed by others
References
Arcangeli G, Saracino B, Gomellini S et al (2010) A prospective phase III randomized trial of hypofractionation versus conventional fractionation in patients with high-risk prostate cancer. Int J Radiat Oncol Biol Phys 78:11–18
Aubry JF, Beaulieu L, Girouard LM et al (2004) Measurements of intrafraction motion and interfraction and intrafraction rotation of prostate by three-dimensional analysis of daily portal imaging with radiopaque markers. Int J Radiat Oncol Biol Phys 60:30–39
Ballhausen H, Hieber S, Li M et al (2014) Technical note: millimeter precision in ultrasound based patient positioning: experimental quantification of inherent technical limitations. Med Phys 41:081718
Beltran C, Herman M, Davis B (2008) Planning target margin calculations for prostate radiotherapy based on intrafraction and interfraction motion using four localization methods. Int J Radiat Oncol Biol Phys 70:289–295
Boda-Heggemann J, Kohler FM, Kupper B et al (2008) Accuracy of ultrasound-based (BAT) prostate-repositioning: a three-dimensional on-line fiducial-based assessment with cone-beam computed tomography. Int J Radiat Oncol Biol Phys 70:1247–1255
Cahlon O, Zelefsky MJ, Shippy A et al (2008) Ultra-high dose (86.4 Gy) IMRT for localized prostate cancer: toxicity and biochemical outcomes. Int J Radiat Oncol Biol Phys 71:330–337
Chandra A, Dong L, Huang E et al (2003) Experience of ultrasound-based daily prostate localization. Int J Radiat Oncol Biol Phys 56:436–447
Crehange G, Mirjolet C, Gauthier M et al (2012) Clinical impact of margin reduction on late toxicity and short-term biochemical control for patients treated with daily on-line image guided IMRT for prostate cancer. Radiother Oncol 103:244–246
Crook JM, Raymond Y, Salhani D et al (1995) Prostate motion during standard radiotherapy as assessed by fiducial markers. Radiother Oncol 37:35–42
Cury FL, Shenouda G, Souhami L et al (2006) Ultrasound-based image guided radiotherapy for prostate cancer: comparison of cross-modality and intramodality methods for daily localization during external beam radiotherapy. Int J Radiat Oncol Biol Phys 66:1562–1567
Dearnaley D, Syndikus I, Sumo G et al (2012) Conventional versus hypofractionated high-dose intensity-modulated radiotherapy for prostate cancer: preliminary safety results from the CHHiP randomised controlled trial. Lancet Oncol 13:43–54
Dearnaley DP, Jovic G, Syndikus I et al (2014) Escalated-dose versus control-dose conformal radiotherapy for prostate cancer: long-term results from the MRC RT01 randomised controlled trial. Lancet Oncol 15:464–473
Dobler B, Mai S, Ross C et al (2006) Evaluation of possible prostate displacement induced by pressure applied during transabdominal ultrasound image acquisition. Strahlenther Onkol 182:240–246
Fargier-Voiron M, Presles B, Pommier P et al (2016) Evaluation of a new transperineal ultrasound probe for inter-fraction image-guidance for definitive and post-operative prostate cancer radiotherapy. Phys Med 32:499–505
Fargier-Voiron M, Presles B, Pommier P et al (2014) Impact of probe pressure variability on prostate localization for ultrasound-based image-guided radiotherapy. Radiother Oncol 111:132–137
Foster RD, Solberg TD, Li HS et al (2010) Comparison of transabdominal ultrasound and electromagnetic transponders for prostate localization. J Appl Clin Med Phys 11:2924
Gadia R, Leite ET, Gabrielli FG et al (2013) Outcomes of high-dose intensity-modulated radiotherapy alone with 1 cm planning target volume posterior margin for localized prostate cancer. Radiat Oncol 8:285
Ghilezan MJ, Jaffray DA, Siewerdsen JH et al (2005) Prostate gland motion assessed with cine-magnetic resonance imaging (cine-MRI). Int J Radiat Oncol Biol Phys 62:406–417
Kuban DA, Dong L, Cheung R et al (2005) Ultrasound-based localization. Semin Radiat Oncol 15:180–191
Kupelian P, Willoughby T, Mahadevan A et al (2007) Multi-institutional clinical experience with the Calypso system in localization and continuous, real-time monitoring of the prostate gland during external radiotherapy. Int J Radiat Oncol Biol Phys 67:1088–1098
Kupelian PA, Willoughby TR, Meeks SL et al (2005) Intraprostatic fiducials for localization of the prostate gland: monitoring intermarker distances during radiation therapy to test for marker stability. Int J Radiat Oncol Biol Phys 62:1291–1296
Letourneau D, Martinez AA, Lockman D et al (2005) Assessment of residual error for online cone-beam CT-guided treatment of prostate cancer patients. Int J Radiat Oncol Biol Phys 62:1239–1246
Li M, Ballhausen H, Hegemann NS et al (2015) A comparative assessment of prostate positioning guided by three-dimensional ultrasound and cone beam CT. Radiat Oncol 10:82
McNair HA, Mangar SA, Coffey J et al (2006) A comparison of CT- and ultrasound-based imaging to localize the prostate for external beam radiotherapy. Int J Radiat Oncol Biol Phys 65:678–687
Peeters ST, Heemsbergen WD, Koper PC et al (2006) Dose-response in radiotherapy for localized prostate cancer: results of the Dutch multicenter randomized phase III trial comparing 68 Gy of radiotherapy with 78 Gy. J Clin Oncol 24:1990–1996
Peng LC, Yang CC, Sim S et al (2007) Dose comparison of megavoltage cone-beam and orthogonal-pair portal images. J Appl Clin Med Phys 8:10–20
Perks JR, Lehmann J, Chen AM et al (2008) Comparison of peripheral dose from image-guided radiation therapy (IGRT) using kV cone beam CT to intensity-modulated radiation therapy (IMRT). Radiother Oncol 89:304–310
Pollack A, Zagars GK, Starkschall G et al (2002) Prostate cancer radiation dose response: results of the M. D. Anderson phase III randomized trial. Int J Radiat Oncol Biol Phys 53:1097–1105
Robinson D, Liu D, Steciw S et al (2012) An evaluation of the Clarity 3D ultrasound system for prostate localization. J Appl Clin Med Phys 13:3753
Serago CF, Buskirk SJ, Igel TC et al (2006) Comparison of daily megavoltage electronic portal imaging or kilovoltage imaging with marker seeds to ultrasound imaging or skin marks for prostate localization and treatment positioning in patients with prostate cancer. Int J Radiat Oncol Biol Phys 65:1585–1592
Shinohara K, Roach M 3rd (2008) Technique for implantation of fiducial markers in the prostate. Urology 71:196–200
Skarsgard D, Cadman P, El-Gayed A et al (2010) Planning target volume margins for prostate radiotherapy using daily electronic portal imaging and implanted fiducial markers. Radiat Oncol 5:52
Van Der Heide UA, Kotte AN, Dehnad H et al (2007) Analysis of fiducial marker-based position verification in the external beam radiotherapy of patients with prostate cancer. Radiother Oncol 82:38–45
Van Der Meer S, Bloemen-Van Gurp E, Hermans J et al (2013) Critical assessment of intramodality 3D ultrasound imaging for prostate IGRT compared to fiducial markers. Med Phys 40:071707
Van Herk M, Remeijer P, Rasch C et al (2000) The probability of correct target dosage: dose-population histograms for deriving treatment margins in radiotherapy. Int J Radiat Oncol Biol Phys 47:1121–1135
Wertz H, Lohr F, Dobler B et al (2007) Dosimetric consequences of a translational isocenter correction based on image guidance for intensity modulated radiotherapy (IMRT) of the prostate. Phys Med Biol 52:5655–5665
Wu Q, Ivaldi G, Liang J et al (2006) Geometric and dosimetric evaluations of an online image-guidance strategy for 3D-CRT of prostate cancer. Int J Radiat Oncol Biol Phys 64:1596–1609
Zelefsky MJ, Levin EJ, Hunt M et al (2008) Incidence of late rectal and urinary toxicities after three-dimensional conformal radiotherapy and intensity-modulated radiotherapy for localized prostate cancer. Int J Radiat Oncol Biol Phys 70:1124–1129
Acknowledgements
We thank Patrick Dominik Thum, Andrea Beisel, Anne Kolberg, Gabriela Danilkiewicz, and Anja Weber for their excellent work as radiation therapists.
Funding
Elekta Germany supports research at the university hospital of the Ludwig Maximilian University. Funding for research with the Clarity® system has been received from Elekta. Elekta had no influence on study design; nor on data collection, analysis, or interpretation; nor on the writing of the manuscript; nor on the decision to submit the manuscript for publication.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
H. Ballhausen, N.-S. Hegemann, M. Reiner, S. Tritschler, C. Gratzke, F. Manapov, S. Corradini, and U. Ganswindt declare that they have no competing interests. Elekta supported various congress presentations by C. Belka and M. Li.
Ethical standards
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Additional information
Authors’ contributions
M. Li conceived the study, performed measurements, and drafted the manuscript. H. Ballhausen performed the statistical work and contributed to writing the manuscript. N.-S. Hegemann organized fiducial implantations and provided helpful suggestions. F. Manapov performed measurements and provided helpful suggestions. M. Reiner, U. Ganswindt, S. Corradini, and C. Belka helped with study design and provided useful suggestions. S. Tritschler and C. Gratzke implanted fiducial markers in prostates and provided helpful advices. All authors read and approved the final manuscript.
Rights and permissions
About this article
Cite this article
Li, M., Ballhausen, H., Hegemann, NS. et al. Comparison of prostate positioning guided by three-dimensional transperineal ultrasound and cone beam CT. Strahlenther Onkol 193, 221–228 (2017). https://doi.org/10.1007/s00066-016-1084-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00066-016-1084-7
Keywords
- Prostate cancer
- Radiotherapy, image-guided
- Radiotherapy, intensity-modulated
- Organs at risk
- Radiation oncologists