Abstract
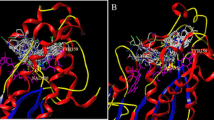
Enoyl-ACP reductase is the key enzyme involved in FAS-II synthesis of mycolic acid in bacterial cell wall and is a promising target for discovering new chemical entity. The designed pharmacophores are the possible better tools to combat mutation in enoyl-ACP enzyme, which leads to a decrease in volume of triclosan binding site. Compound 3a showed H-bonding interactions similar to that of triclosan with enoyl-ACP enzyme and with a better docking score (C score 8.81), while the compound 3f showed additional interaction with MET98.H amino acid residue. The 3D-QSAR computations also support the docking study to develop novel pyrrole-based derivatives.
Graphical abstract
Molecular docking 3D-QSAR studies and synthesis of active analogs of pyrrole carbaldehyde as better receptor fit pharmacophore for enoyl-ACP reductase along with in vitro antitubercular activity.










Similar content being viewed by others
References
Agarwal A, Pearson PP, Taylor EW, Li HB, Dahlgren T, Herslof M, Yang Y, Lambert G, David L (1993) Three-dimensional quantitative structure–activity relationships of 5-HT receptor binding data for tetrahydropyridinylindole derivatives: a comparison of the Hansch and CoMFA methods. J Med Chem 36:4006–4014
Baldock C, Rafferty JB, Sedelnikova SE, Baker PJ, Stuitje AR, Slabas AR, Hawkes TR, Rice DW (1996) A mechanism of drug action revealed by structural studies of enoyl reductase. Science 274:2107–2110
Banerjee A, Dubnau E, Quemard A, Balasubramanian V, Um KS, Wilson T, Collins D, de-Lisle G, Jacobs WR Jr (1994) InhA, a gene encoding a target for isoniazid and ethionamide in Mycobacterium tuberculosis. Science 263:227–230
Biava M, Fioravanti R, Porretta GC, Sleiter G, Ettorre A, Deidda D, Lampis G, Pompei R (1997) New toluidine derivatives with antimycobacterial and antifungal activities. Med Chem Res 7:228–250
Bush BL, Nachbar RB (1993) Sample-distance partial least squares: PLS optimized for many variables, with application to CoMFA. J Comput Aided Mol Des 7:587–619
Chhibber M, Kumar G, Parasuraman P, Ramya TN, Surolia N, Surolia A (2006) Novel diphenyl ethers: design, docking studies, synthesis and inhibition of enoyl-ACP reductase of Plasmodium falciparum and Escherichia coli. Bioorg Med Chem 14:8086–8098
Chin WW (1998) The partial least squares approach to structural equation modeling. In: Marcoulides GA (ed) Modern methods for business research. Lawrence Erlbaum Associates, Mahwah, NJ, pp 295–336
Clark M, Richard D, Cramer RD III, Opdenbosch NV (1989) Validation of the general purpose tripos 5.2 force field. J Comput Chem 10:982–1012
Clark M, Cramer RD III, Jones DM, Patterson DE, Simeroth PE (1990) Comparative molecular field analysis (CoMFA) 2. Toward its use with 3D-structural databases. Tetrahedron Comp Methodol 3:47–59
Clark RD, Strizhev A, Leonard JM, Blake JF, Matthew JB (2002) Consensus scoring for ligand/protein interactions. J Mol Graph Model 20:281–295
Cramer RD III, Bunce JD, Patterson DE, Frank IE (1988) Cross validation, bootstrapping, and partial least squares compared with multiple regression in conventional QSAR studies. Mol Inf 7:18–25
Cramer RD III, Patterson DE, Bunce JD (1989) Recent advances in comparative molecular field analysis (CoMFA). Prog Clin Biol Res 291:161–165
Deidda D, Lampis G, Fioravanti R, Biava M, Porretta GC, Zanetti S, Pompei R (1998) Bactericidal activities of the pyrrole derivative BM212 against multidrug-resistant and intramacrophagic Mycobacterium tuberculosis strains. Antimicrob Agents Chemother 42:3035–3037
Eldridge MD, Murray CW, Auton TR, Paolini GV, Mee RP (1997) Empirical scoring functions: I. The development of a fast empirical scoring function to estimate the binding affinity of ligands in receptor complexes. J Comput Aided Mol Des 11:425–445
Espinal MA, Laszlo A, Simonsen L, Boulahbal F, Kim SJ, Reniero A, Hoffner S, Rieder HL, Binkin N, Dye C, Williams R, Raviglione MC (2001) Global trends in resistance to antituberculosis drugs. N Engl J Med 344:1294–1303
Faustini A, Hall AJ, Perucci CA (2006) Risk factors for multidrug resistant tuberculosis in Europe: a systematic review. Thorax 61:158–163
Fioravanti R, Biava M, Porretta GC, Artico M, Lampis G, Deidda D, Pompei R (1997) N-substituted 1-aryl-2(1H-imidazol-1-yl)1-ethanamines with broad spectrum in vitro antimycobacterial and antifungal activities. Med Chem Res 7:87–97
Fischer F, Matthisson M, Herrling P (2004) List of drugs in development for neurodegenerative diseases. Neurodegener Dis 1:50–70
Franzblau SG, Witzig RS, McLaughlin JC, Torres P, Madico G, Hernandez A, Degnan MT, Cook MB, Quenzer VK, Ferguson RM, Gilman RH (1998) Rapid, low-technology MIC determination with clinical Mycobacterium tuberculosis isolates by using the Microplate Alamar Blue Assay. J Clin Microbiol 3:362–366
Heath RJ, Rock CO (1995) Enoyl-acyl carrier protein reductase (FabI) plays a determinant role in completing cycles of fatty acid elongation in Escherichia coli. J Biol Chem 270:26538–26542
Heath RJ, Li J, Roland GE, Rock CO (2000) Inhibition of the Staphylococcus aureus NADPH-dependent enoyl-acyl carrier protein reductase by triclosan and hexachlorophene. J Biol Chem 275:4654–4659
Johnsson K, Schultz PG (1994) Mechanistic studies of the oxidation of isoniazid by the catalase peroxidase from Mycobacterium tuberculosis. J Am Chem Soc 116:7425–7426
Johnsson K, King DS, Schultz PG (1995) Studies on the mechanism of action of isoniazid and ethionamide in the chemotherapy of tuberculosis. J Am Chem Soc 117:5009–5010
Jones G, Willett P, Glen RC, Leach AR, Taylor R (1997) Development and validation of a genetic algorithm for flexible docking. J Mol Biol 267:727–748
Joshi SD, Vagdevi HM, Vaidya VP, Gadaginamath GS (2008) Synthesis of new 4-pyrrol-1-yl benzoic acid hydrazide analogs and some derived oxadiazole, triazole and pyrrole ring systems, a novel class of potential antibacterial and antitubercular agents. Eur J Med Chem 43:1989–1996
Joshi SD, More UA, Kulkarni VH (2013a) Synthesis, antimicrobial and cytotoxic activity of new heterocyclic hybrids based on 2,5-dimethylpyrrole and pyrrole scaffolds. Indian J Pharm Sci 75:310–323
Joshi SD, More UA, Pansuriya K, Aminabhavi TM, Gadad AK (2013b) Synthesis and molecular modeling studies of novel pyrrole analogs as antimycobacterial agents. J Saudi Chem Soc. doi:10.1016/j.jscs.2013.09.002
Joshi SD, More UA, Aminabhavi TM, Badiger AM (2014a) Two- and three-dimensional QSAR studies on a set of antimycobacterial pyrroles: CoMFA, Topomer CoMFA, and HQSAR. Med Chem Res 23:107–126
Joshi SD, More UA, Dixit SR, Korat HH, Aminabhavi TM, Badiger AM (2014b) Synthesis, characterization, biological activity, and 3D-QSAR studies on some novel class of pyrrole derivatives as antitubercular agents. Med Chem Res 23:1123–1147
Kitagawa H, Kumura K, Takahata S, Iida M, Atsumi K (2007) 4-Pyridone derivatives as new inhibitors of bacterial enoyl-ACP reductase FabI. Bioorg Med Chem 15:1106–1116
Kuntz ID, Blaney JM, Oatley SJ, Langridge R, Ferrin TE (1982) A geometric approach to macromolecule–ligand interactions. J Mol Biol 161:269–288
La Rosa V, Poce G, Canseco JO, Buroni S, Pasca MR, Biava M, Raju RM, Porretta GC, Alfonso S, Battilocchio C, Javid B, Sorrentino F, Loerger TR, Sacchettinic JC, Manetti F, Botta M, Logu AD, Rubin EJ, Rossi ED (2012) MmpL3 is the cellular target of the antitubercular pyrrole derivative BM212. Antimicrob Agents Chemother 56:324–331
Levy CW, Roujeinikova A, Sedelnikova S, Baker PJ, Stuitje AR, Slabas AR, Rice DW, Rafferty JB (1999) Molecular basis of triclosan activity. Nature 398:383–384
Lourenco MCS, de Souza MVN, Pinheiro AC, Ferreira ML, Goncalves RSB, Nogueira TCM, Peralta MA (2007) Evaluation of anti-tubercular activity of nicotinic and isoniazid analogues. ARKIVOC 15:181–191
Lu H, Tonge PJ (2008) Inhibitors of FabI, an enzyme drug target in the bacterial fatty acid biosynthesis pathway. Acc Chem Res 41:11–20
Luckner SR, Liu N, Ende CW, Tonge PJ, Kisker C (2010) A slow, tight binding inhibitor of InhA, the enoyl-acyl carrier protein reductase from Mycobacterium tuberculosis. J Biol Chem 285:14330–14337
Mallegol T, Gmouh S, Meziane MAA, Desce MB, Mongin O (2005) Practical and efficient synthesis of tris(4-formylphenyl)amine, a key building block in materials chemistry. Synthesis 11:1771–1774
McMurry LM, Oethinger M, Levy SB (1998) Triclosan targets lipid synthesis. Nature 394:531–532
More UA, Joshi SD, Aminabhavi TM, Gadad AK, Mallikarjuna NN, Kulkarni VH (2014) Design, synthesis, molecular docking and 3D-QSAR studies of potent inhibitors of enoyl-acyl carrier protein reductase as potential antimycobacterial agents. Eur J Med Chem 71:199–218
Muegge I, Martin YC (1999) A general and fast scoring function for protein–ligand interactions: a simplified potential approach. J Med Chem 42:791–804
Park HS, Yoon YM, Jung SJ, Kim CM, Kim JM, Kwak JH (2007) Antistaphylococcal activities of CG400549, a new bacterial enoyl-acyl carrier protein reductase (FabI) inhibitor. J Antimicrob Chemother 60:568–574
Quemard A, Lacave C, Laneelle G (1991) Isoniazid inhibition of mycolic acid synthesis by cell extracts of sensitive and resistant strains of Mycobacterium aurum. Antimicrob Agents Chemother 35:1035–1039
Quemard A, Sacchettini JC, Dessen A, Vilcheze C, Bittma R, Jacobs WR Jr, Blanchard JS (1995) Enzymatic characterization of the target for isoniazid in Mycobacterium tuberculosis. Biochemistry 34:8235–8241
Raviglione M, Marais B, Floyd K, Lönnroth K, Getahun H, Migliori GB, Harries AD, Nunn P, Lienhardt C, Graham S, Chakaya J, Weyer K, Cole S, Kaufmann SHE, Zumla A (2012) Scaling up interventions to achieve global tuberculosis control: progress and new developments. Lancet 379:1902–1913
Seefeld MA, Miller WH, Kenneth AN, Burgess WJ, DeWolf WE Jr, Elkins PA, Head MS, Jakas DR, Janson CA, Keller PM, Manley PJ, Moore TD, Payne DJ, Pearson S, Polizzi BJ, Qiu X, Rittenhouse SF, Uzinskas IN, Wallis NG, Huffman WF (2003) Indole naphthyridinones as inhibitors of bacterial enoyl-ACP reductases FabI and FabK. J Med Chem 46:1627–1635
Sivaraman S, Sullivan TJ, Johnson F, Novichenok P, Cui G, Simmerling C, Tonge PJ (2004) Inhibition of the bacterial enoyl reductase FabI by triclosan: a structure–reactivity analysis of FabI inhibition by triclosan analogues. J Med Chem 47:509–518
Sullivan TJ, Truglio JJ, Boyne ME, Novichenok P, Zhang X, Stratton CF, Li HJ, Kaur T, Amin A, Johnson F, Slayden RA, Kisker C, Tonge PJ (2006) High affinity InhA inhibitors with activity against drug-resistant strains of Mycobacterium tuberculosis. ACS Chem Biol 1:43–53
Thomas G (2007) Structure–activity and quantitative structure relationships. medicinal chemistry an introduction, 2nd edn. Wiley, New York, pp 75–110
Tipparaju SK, Mulhearn DC, Klein GM, Chen Y, Tapadar S, Bishop MH, Yang S, Chen J, Ghassemi M, Santarsiero BD, Cook JL, Johlfs M, Mesecar AD, Johnson ME, Kozikowski AP (2008) Design and synthesis of aryl ether inhibitors of the Bacillus anthracis enoyl-ACP reductase. Chem Med Chem 3:1250–1268
Tripos International (2012) Sybyl-X 2.1.1, Tripos International, St. Louis, MO, USA
Varela C, Rittmann D, Singh A, Krumbach K, Bhatt K, Eggeling L, Besra GS, Bhatt A (2012) MmpL genes are associated with mycolic acid metabolism in mycobacteria and Corynebacteria. Chem Biol 19:498–506
Vilsmeier A, Haack A (1927) Uber die einwirkung von halogenphosphor auf alkyl-formanilide. eine neue methode zur darstellung sekundarer und tertiarer p-alkylamino-benzaldehyde. Eur J Inorg Chem 60:119–122
WHO (2012) Global tuberculosis report 2012. World Health Organization, Geneva, Switzerland
World Health Organization (WHO) (2010) Multidrug and extensively drug-resistant TB (M/XDR-TB). (2010) Global report on surveillance and response. WHO/HTM/TB/2010.3. Geneva, Switzerland. http://whqlibdoc.who.int/publications/2010/9789241599191_eng.pdf
Zhang Y, Heym B, Allen B, Young D, Cole S (1992) The catalase–peroxidase gene and isoniazid resistance of Mycobacterium tuberculosis. Nature 358:591–593
Acknowledgments
Authors immensely thank research support from the Board of Research in Nuclear Sciences (BRNS), Bhabha Atomic Research Centre (BARC), Mumbai (File No. 2013/37B/17/BRNS/0417 dated-14/05/2013). We also thank Dr. V. H. Kulkarni, Principal and Mr. H. V. Dambal, President, S.E.T’s College of Pharmacy, Dharwad, India for providing the facilities. The authors are grateful to Mr. Ravindra N. Nadagir and Mr. Shrikant A. Tiwari for their technical assistance.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Joshi, S.D., Kumar, D., More, U.A. et al. Design and development of pyrrole carbaldehyde: an effective pharmacophore for enoyl-ACP reductase. Med Chem Res 25, 672–689 (2016). https://doi.org/10.1007/s00044-016-1517-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-016-1517-y