Abstract
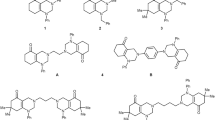
An efficient and direct protocol for the preparation of 1-amidoalkyl-2-naphthols 1a–f employing a multi-component, one-pot condensation reaction of 2-naphthol, heterocyclic aldehydes, and amides in the presence of anhydrous zinc chloride under solvent-free conditions is described. The thermal solvent-free offer advantages such as shorter reaction times, simple work-up and excellent yield. Ring closure of 1a–f gave the pyrazolyl- and indolyl oxazine derivatives 2a–f. On the other hand, the reaction of 2-naphthol, aldehydes, and ammonia solution gave the dipyrazolyl- and di-indolyl oxazine derivatives 3a,b. Some of the newly synthesized compounds showed promising antibacterial and anti-H5N1 activities.




Similar content being viewed by others
References
Betti M (1942) Org Synth 1:381–383
Das B, Laxminarayana K, Ravikanth B, Rao R (2007) Iodine catalyzed preparation of amidoalkyl naphthols in solution and under solvent-free conditions. J Mol Catal A Chem 261:180–183
Hajipour AR, Ghayeb Y, Sheikhan N, Ruoho AE (2009) Brønsted acidic ionic liquid as an efficient and reusable catalyst for one-pot synthesis of 1-amidoalkyl 2-naphthols under solvent-free conditions. Tetrahedron Lett 50:5649–5651
Hashem AI, Youssef ASA, Kandeel KA, Abou-Elmagd WSI (2007) Conversion of some 2(3H)-furanones bearing a pyrazolayl group into other heterocyclic system with study of their antiviral activity. Eur J Med Chem 42:934–939
Kantevari S, Vuppalapati SVN, Nagarapu L (2007) Montmorillonite K10 catalyzed efficient synthesis of amidoalkyl naphthols under solvent free conditions. Catal Commun 8:1857–1862
Kumar A, Rao MS, Ahmad I, Khungar B (2009) A simple and facile synthesis of amidoalkyl naphthols catalyzed by Yb(OTf)3 in ionic liquids. Can J Chem 87:714–719
Lei M, Ma L, Hu LH (2009) Thiamine hydrochloride as a efficient catalyst for the synthesis of amidoalkyl naphthols. Tetrahedron Lett 50:6393–6397
Mahdavinia GH, Bigdeli MA (2009) Wet cyanuric chloride promoted efficient synthesis of amidoalkyl naphthols under solvent-free conditions. Chin Chem Lett 20:383–386
Mahdavinia GH, Bigdeli MA, Heravi MM (2008) Silica supported perchloric acid (HClO4–SiO2): a mild, reusable and highly efficient heterogeneous catalyst for the synthesis of amidoalkyl naphthols. Chin Chem Lett 19:1171–1174
Mossman T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65:55–65
Nagarapu L, Baseeruddin M, Apuri S, Kantevari S (2007) Potassium dodecatungstocobaltate trihydrate (K5CoW12O40·3H2O): a mild and efficient reusable catalyst for the synthesis of amidoalkyl naphthols in solution and under solvent-free conditions. Catal Commun 8:1729–1734
Nagawade RR, Shinde DB (2007) Synthesis of amidoalkyl naphthols by an iodine-catalyzed multicomponent reaction of β-naphthol. Mendeleev Commun 17:299–300
Nandi GC, Samai S, Kumar R, Singh MS (2009) Atom-efficient and environment-friendly multicomponent synthesis of amidoalkyl naphthols catalyzed by P2O5. Tetrahedron Lett 50:7220–7222
Patil SB, Singh PR, Surpur MP, Samant SD (2007) Ultrasound-promoted synthesis of 1-amidoalkyl-2-naphthols via a three-component condensation of 2-naphthol, ureas/amides, and aldehydes, catalyzed by sulfamic acid under ambient conditions. Ultrason Sonochem 14:515–518
Pauwels R, Balzarini J, Baba M, Snoeck R, Schols D, Herdewijn P, Desmyter J, De Clercq E (1988) Rapid and automated tetrazolium-based colorimetric assay for the detection of anti-HIV compounds. J Virol Methods 20(4):309–321
Selvam NP, Perumal PT (2006) A new synthesis of acetamido phenols promoted by Ce(SO4)2. Tetrahedron Lett 47:7481–7483
Shaterian HR, Yarahmadi H (2008) A modified reaction for the preparation of amidoalkyl naphthols. Tetrahedron Lett 49:1297–1300
Shaterian HR, Yarahmadi H, Ghashang M (2008) An efficient, simple and expedition synthesis of 1-amidoalkyl-2-naphthols as ‘drug like’ molecules for biological screening. Bioorg Med Chem Lett 18:788–792
Shen AY, Tsai CT, Chen CL (1999) Synthesis and cardiovascular evaluation of N-substituted aminonaphthols. Eur J Med Chem 34:877–882
Soundararajan V, Tharakaraman K, Raman R, Raguram S, Sasisekharan V, Sasisekharan R (2009) Extrapolating from sequence—the 2009 H1N1 ‘swine’ influenza virus. Nat Biotechnol 27(6):510–513
Srihari G, Nagaraju M, Murthy MM (2007) Solvent-free one-pot synthesis of amidoalkyl naphthols catalyzed by silica sulfuric acid. Helv Chim Acta 90:1497–1504
Su WK, Tang WY, Li JJ (2008) Strontium(II) triflate catalysed condensation of β-naphthol, aldehyde and urea or amides: a facile synthesis of amidoalkyl naphthols. Chem Res 3:123–128
Szatmäri I, Fülöp F (2004) Syntheses and transformations of 1-(α-aminobenzyl)-2-naphthol derivatives. Curr Org Synth 1:155–165
Turgut Z, Pelit E, Köycü A (2007) Synthesis of new 1,3-disubstituted-2,3-dihydro-1H-naphth[1,2-e][1,3]oxazines. Molecules 12:345
Xu Y, Guo Q (2004) Synthesis of heterocyclic compounds under microwave irradiation. Heterocycles 63:903
Acknowledgments
The authors are thankful to Dr. Alaa R.I. Morsy, Central Laboratory for Evaluation of Veterinary Biologics, Agricultural Research Center, Abassia, Cairo, Egypt and Dr. Madeha O.I. Ghobashy, Microbiology Department, Faculty of Science, Ain Shams University for biological investigations.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abou-Elmagd, W.S.I., Hashem, A.I. Synthesis of 1-amidoalkyl-2-naphthols and oxazine derivatives with study of their antibacterial and antiviral activities. Med Chem Res 22, 2005–2013 (2013). https://doi.org/10.1007/s00044-012-0205-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-012-0205-9