Abstract
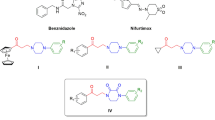
Synthesis and evaluation of the bioactivity of spiroheterocycles (STC) against Trypanosoma cruzi are described. Selectivity indices were improved for two compounds versus the leads 17 and 20, the spiro-thiochromanone (STC) derivatives 17–26, thus increasing the therapeutic interest of our family. As our previous studies conducted on the structure of pharmacophore sites of our compounds made us hypothesize the existence of original sites, STC can be considered as promising tools further anti-trypanosoma studies, as probes for affinity chemotherapy. Compounds 17 and 20 are more potent and more selective than benznidazole and nifurtimox.
Graphical abstract
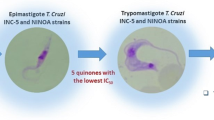
The highest anti-trypanosoma cruzi (TC) activity is obtained for compounds 17 and 20 which exhibited low IC50 values (1.5 and 3.78 μM), up to sixfold lower than clinical drugs, nifurtimox (Nfx) or up to fourfold lower than benznidazole (Bdz).





Similar content being viewed by others
References
Akkurt M, Yildirim SO, Badri R, Kerbal A, Ben Hadda T, Buyukgungor O (2006) 4-p-Anisyl-1,2′-diphenyl-3-p-tolyl-2′,3′-dihydro-spiro[2-pyrazoline-5,3,0-isoquinolin]-4′(1′H)-one. Acta Cryst E62:o5022–o5023
Akkurt M, Yıldırım SÖ, Kerbal A, Bennani B, Daoudi M, Chohan ZH, McKee V, Ben Hadda T (2009) Crystal structure of 3′-(4-methoxyphenyl)-2-phenyl-4′-(4-ethoxyphenyl)-1,2-dihydro-4H,4′H-spiro [isoquinoline-3,5′-isoxazol]-4-one. J Chem Crystallogr. doi:10.1007/s10870-009-9639-4
Al Houari G, Kerbal A, Larbi NB, Bennani B, Ben Hadda T, Stoeckli-Evans H (2008a) 4′-Methyl-3-(4-nitrophenyl)-4-phenyl-4,5,1′,2′,3′,4′-hexahydrospiro[isoxazole-5,2′-naphthalen]-1′-one. Acta Cryst E64:o509–o510
Al Houari G, Kerbal A, Bennani B, Baba MF, Daoudi M, Ben Hadda T (2008b) Drug design of new antitubercular agents: 1,3-dipolar cycloaddition reaction of arylnitriloxides and 3-para-methoxy-benzylidene-isochroman-4-ones. Arkivoc xii:42–50
Anaflous A, Benchat N, Mimouni M, Abouricha S, Ben Hadda T, El-Bali B, Hakkou A, Hacht B (2004) Armed imidazo [1,2-a] pyrimidines (pyridines):evaluation of antibacterial activity. Lett Drug Des Discov 1:224–229
Badri R, Kerbal A, Najib B, El-Bali B, Escudie J, Ranaivonjatovo H, Bolte M (1999) 3′-(p-Nitrophenyl)-2-phenyl-4′-(p-tolyl)spiro[isoquinoline-3(2H),5′(4′H)-isoxazolin]-4(1H)-one. Acta Cryst C55. IUC9900165
Ben Hadda T, Benchat N, El-Bali B, Abouricha S, Moueqqit M, Mimouni M. (2003) Impact of dimroth rearrangement on anti-tuberculosis activity of 3-armed-imidazo[1,2-a]pyrimidines IMP (-pyridines) IP. Med Pharm Chem 1–18 online. Paper N°: 0301001
Ben Hadda T, Badri R, Kerbal A, Filali Baba B, Akkurt M, Demailly G, Benazza M (2007) Synthesis and antitubercular activity of spiroheterocycles: 2,2′,4′,5′-tetra-substituted-1,2,2′,4′-tetrahydro-4H-spiro[isoquinoline-3,3′-pyrazol]-4-ones. Arkivoc xiv:276–288
Ben Hadda T, Badri R, Kerbal A, Bennani B, Al Houari G, Filali Baba B, Akkurt M, Demailly G, Benazza M (2008) Looking for a new antitubercular pharmacophore site: synthesis and bioactivity of spiroheterocycles 2,3′,4′-tri-subsituted-1,2-dihydro-4H,4′H-spiro [isoquinoline-3,5′-isoxazol]-4-ones. Arkivoc ii:1–13
Bennani B, Filalibaba B, El-Fazazi A, Houari GA, Bitit N, Kerbal A, El-Bali B, Bolte M (2002) 2′,3′-Diphenyl-4′-p-tolylspiro[isothiachroman-3,5′-isoxazolidin]-4(2H)-one. Acta Cryst E58:o312–o313
Bennani B, Jalbout AF, Baba BF, Larbi NB, Boukir A, Kerbal A, Mimouni M, Ben Hadda T, Trzaskowski B (2007a) Synthesis of new spiro-[isothiochromene-3,5′-isoxazolidin]-4(1H)-ones. Heterocycl Chem 44(3):711–716
Bennani B, Kerbal A, Daoudi M, Filali Baba B, Al Houari G, Jalbout AF, Mimouni M, Benazza M, Demailly G, Akkurt M, Yýldýrým SÖ, Ben Hadda T (2007b) Combined drug design of potential Mycobacterium tuberculosis and HIV-1 inhibitors: 3′,4′-di-substituted-4′H-spiro[isothiochromene-3,5′-isoxazol]-4(1H)-one. Arkivoc xvi:19–40
Castro JA, Diaz de Toranzo EG (1988) Toxic effects of nifurtimox and benznidazole, two drugs used against American trypanosomiasis (Chagas’ disease). Biomed Environ Sci 1(1):19–33
Changtam C, de Koning HP, Ibrahim H, Sajid MS, Gould MK, Suksamrarn A (2010) Curcuminoid analogs with potent activity against Trypanosoma and Leishmania species. Eur J Med Chem 45:941–956
Chohan ZH, Sumrra SH, Youssoufi MH, Ben Hadda T (2010a) Metal based biologically active compounds: design, synthesis and antibacterial/antifungal/cytotoxic properties of triazole derived Schiff bases and their oxovanadium(IV) complexes. Eur J Med Chem 45:2739–2747
Chohan ZH, Youssoufi MH, Jarrahpour A, Ben Hadda T (2010b) Identification of antibacterial and antifungal pharmacophore sites for potent bacteria and fungi inhibition: indolenyl sulfonamide derivatives. Eur J Med Chem 45:1189–1199
Chohan ZH, Sumrra SH, Youssoufi MH, Ben Hadda T (2010c) Design and synthesis of triazole schiff bases and their oxovanadium(IV) complexes as antimicrobial agents. J Coord Chem 63(22):3981–3998
Chohan ZH, Shad HA, Toupet L, Ben Hadda T, Akkurt M (2010d) Structure of a new bioactive agent containing combined antibacterial and antifungal pharmacophore sites: 4-{[(E)-(5-bromo-2-hydroxyphenyl)methylidene]amino}-N-(5-methyl-1,2-oxazol-3-yl)benzene-sulfonamide. J Chem Crystallogr. doi:10.1007/s10870-010-9856-x
Diaz EG, Montalto DMM, Castro JA (2004) Reactions of nifurtimox with critical sulfhydryl-containing biomolecules: their potential toxicological relevance. J Appl Toxicol 24(3):189–195
Ertl P, Rohde B, Selzer P (2000) Fast calculation of molecular polar surface area as a sum of fragment-based contributions and its application to the prediction of drug transport properties. J Med Chem 43(20):3714–3717
Jarrahpour A, Motamedifar M, Zareil M, Youssoufi MH, Mimouni M, Chohan ZH, Ben Hadda T (2010) Petra, osiris and molinspiration together as a guide in drug design: predictions and correlation structure/antibacterial activity relationships of new N-sulfonyl monocyclic β-lactams (part II). Phosphorus, Sulfur, Silicon Relat Elem 185:491–497
Jawarkar RD, Mahajan DT, Masand VH, Ben Hadda T, Kurhade GH (2010a) COMFA analysis and toxicity risk assessment of coumarin analogues as Mao-A inhibitors: attempting better insight in drug design. Der Pharm Lett 2(6):350–357
Jawarkar RD, Masand VH, Patil KN, Mahajan DT, Youssoufi MH, Ben Hadda T, Kumbhare SL (2010b) 3D-QSAR study on coumarin analogues as potent inhibitors of MAO-B using a CoMFA approach. Der Pharm Chem 2(6):302–310
Moll C, Peris C, Moreno A, Muñoz J, Guañabens N (2008) Severe invalidating pain syndrome associated with benznidazole therapy for Chagas’ disease. Clin Rheumatol 27(2):269–270
Orhan İ, Şenol FS, Khan MTH, Ben Hadda T, Bennani B, Kerbal A, Şener B (2009) Acetylcholinesterase inhibitory potentials of several synthetic spiro-heterocycles. International symposium on pharmaceutical sciences, Ankara, 23–26 June
Parvez A, Jyotsna M, Sheikh J, Tiwari V, Dongre R, Ben Hadda T (2010) Predictions and correlations of structure activity relationship of some aminoantipyrine derivatives on the basis of theoretical and experimental ground. Med Chem Res. doi:10.1007/s00044-010-9505-0
Parvez A, Meshram J, Tiwari V, Sheikh J, Dongre R, Shanti M, Khandarkar K, Ahemad M, Ben Hadda T (2010b) Greener synthetic and nanotechnological bridge: theoretical and practical biological approach toward the bactericidal dual action mechanism of azomethine–nanosilver hybrid. Int J Green Nanotech: Phys Chem 2(2):53–61
Parvez A, Meshram J, Youssoufi MH, Ben Hadda T (2010c) Theoretical calculations and experimental verification of the antibacterial potential of some monocyclic beta-lactames containing two synergetic buried antibacterial pharmacophore sites. Phosphorus, Sulfur Silicon Relat Elem 7:1500–1510
Parvez A, Meshram J, Tiwari V, Sheikh J, Dongre R, Youssoufi MH, Ben Hadda T (2010d) Pharmacophores modeling in terms of prediction of theoretical physicochemical properties and verification by experimental correlations of novel coumarin derivatives produced via Betti’s protocol. Eur J Med Chem 45(9):4370–4378
Pinazo M-J, Muñoz J, Posada E, López-Chejade P, Gállego M, Ayala E, Del Cacho E, Soy D, Gascon J (2010) Tolerance of benznidazole in treatment of Chagas’ disease in adults. Antimicrob Agents Chemother 54(11):4896–4899
Prioli RP, Ordovas JM, Rosenberg I, Schaefer EJ, Pereira ME (1987) Similarity of cruzin, an inhibitor of Trypanosoma cruzi neuraminidase, to high-density lipoprotein. Science 238(4832):1417–1419
Rauf A, Liaqat S, Qureshi AM, Yaqub M, Rehman AU, Hassan MU, Chohan ZH, Nasim Faiz UH, Ben Hadda T (2010) Synthesis, characterization, and urease inhibition of 5-substituted-8-methyl-2H-pyrido[1,2-a]pyrimidine-2,4(3H)-diones. Med Chem Res. doi:10.1007/s00044-010-9491-2
Sheikh J, Parvez A, Ingle V, Juneja H, Dongre R, Chohan ZH, Youssoufi MH, Ben Hadda T (2011) Synthesis, biopharmaceutical characterization, antimicrobial and antioxidant activities of 1-(4′-O-β-D-glucopyranosyloxy-2′-hydroxyphenyl)-3-aryl-propane-1,3-diones. Eur J Med Chem 46:1390–1399
Viotti R, Vigliano C, Lococo B, Alvarez MG, Petti M, Bertocchi G, Armenti A (2009) Tolerance of benznidazole in treatment of Chagas’ disease in adults. Exp Rev Anti Infect Ther 7(2):157–163
Acknowledgments
We are grateful to staff of Federal University of Pernambuco, Recife-Brazil for the help in biological assay. Prof. T. Ben Hadda would like to thank the ACTELION; the Biopharmaceutical Company of Swiss, for the online molecular properties calculations.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Ben Hadda, T., Kerbal, A., Bennani, B. et al. Molecular drug design, synthesis and pharmacophore site identification of spiroheterocyclic compounds: Trypanosoma crusi inhibiting studies. Med Chem Res 22, 57–69 (2013). https://doi.org/10.1007/s00044-012-0010-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-012-0010-5