Abstract
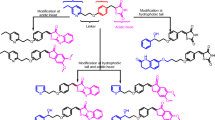
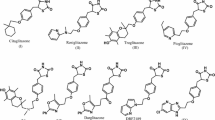
A novel hybrid class of telmisartan–rosiglitazone molecules was synthesized in an attempt to discover a dual peroxisome proliferator-activated receptor gamma (PPARγ) agonist/angiotensin II antagonist for treatment for metabolic syndrome. Almost all the synthesized molecules showed moderate PPARγ activity. However, none of the hybrid analogs showed binding affinity toward the AT1 receptor.













Similar content being viewed by others
References
Balmforth AJ, Lee AJ, Warburton P, Donnelly D, Ball SG (1997) The conformational change responsible for AT1 receptor activation is dependent upon two Juxtaposed asparagine residues on transmembrane helixes III and VII. J Biol Chem 272:4245–4251. doi:10.1074/jbc.272.7.4245
Benson SC, Pershadsingh HA, Ho CI, Chittiboyina A, Desai P, Pravenec M, Qi N, Wang J, Avery MA, Kurtz TW (2004) Identification of telmisartan as a unique angiotensin II receptor antagonist with selective PPARgamma-modulating activity. Hypertension 43:993–1002. doi:10.1161/01.HYP.0000123072.34629.57
Grundy SM, Brewer HB Jr, Cleeman JI, Smith SC Jr, Lenfant C (2004) Definition of metabolic syndrome: report of the National Heart, Lung, and Blood Institute/American Heart Association conference on scientific issues related to definition. Circulation 109:433–438. doi:10.1161/01.CIR.0000111245.75752.C6
Harrold M (2002) Foye’s principles of medicinal chemistry, 5th edn. Lippincott Williams & Wilkins, Philadelphia, pp 533–561
Hoe K-L, Saavedra JM (2002) Site-directed mutagenesis of the gerbil and human angiotensin II AT1 receptors identifies amino acid residues attributable to the binding affinity for the nonpeptidic antagonist losartan. Mol Pharmacol 61:1404–1415. doi:10.1124/mol.61.6.1404
Messerli FH (2002) Vasodilatory edema: a common side effect of antihypertensive therapy. Curr Cardiol Rep 4:479–482. doi:10.1007/s11886-002-0110-9
National Institutes of Health, Treatment of Insulin Resistance in Hypertensive, Obese Adolescents. http://clinicaltrials.gov/show/NCT00185705. Accessed 6 January, 2009
Ohnota M, Orita K, Aizawa Y, Yoshida N, Sakamaki T (2001) Process for the preparation of thiazolidinedione derivatives. WO 2001096321
Okubo T, Tsuchiko F, Wakasawa T, Nambara T (1988) Preparation and antigenic properties of 2-hydroxyestrone-[C-15]-bovine serum albumin conjugate. Chem Pharm Bull (Tokyo) 36:3519–35140
Patny A, Desai PV, Avery MA (2006) Ligand-supported homology modeling of the human angiotensin II type 1 (AT1) receptor: insights into the molecular determinants of telmisartan binding Proteins. Struct Funct Bioinf 65:824–842. doi:10.1002/prot.21196
Pershadsingh HA (2006) Treating the metabolic syndrome using angiotensin receptor antagonists that selectively modulate peroxisome proliferator-activated receptor-gamma. Int J Biochem Cell Biol 38:766–781. doi:10.1016/j.biocel.2005.08.006
Ries UJ, Mihm G, Narr B, Hasselbach KM, Wittneben H, Entzeroth M, van Meel JC, Wienen W, Hauel NH (1993) 6-Substituted benzimidazoles as new nonpeptide angiotensin II receptor antagonists: synthesis, biological activity, and structure–activity relationships. J Med Chem 36:4040–4051. doi:10.1021/jm00077a007
Rouhi AM (2004) Metabolic syndrome. Chem Eng News 82:83–99
Schupp M, Janke J, Clasen R, Unger T, Kintscher U (2004) Angiotensin type 1 receptor blockers induce peroxisome proliferator-activated receptor-gamma activity. Circulation 109:2054–2057. doi:10.1161/01.CIR.0000127955.36250.65
Speth RC (2003) Sarcosine1, glycine8 angiotensin II is an AT1 angiotensin II receptor subtype selective antagonist. Regul Pept 115:203–209. doi:10.1016/S0167-0115(03)00172-1
Speth RC, Harding JW (2001) Radiolabeling of angiotensin peptides. Methods Mol Med 51:275–295
Stouten PFW, Frömmel C, Nakamura H, Sander C (1993) An effective solvation term based on atomic occupancies for use in protein simulations. Mol Simul 10:97–120. doi:10.1080/08927029308022161
Vedejs E, Stults JS (1988) Synthesis of azocine derivatives from thio aldehyde Diels-Alder adducts. J Org Chem 53:2226–2232. doi:10.1021/jo00245a019
Acknowledgements
This investigation was conducted in a facility constructed with support from research facilities improvement program grant number C06 Rr-14503-01 from the National Center for Research Resources, National Institutes of Health. NIH grant 2R42AR44767-02A2 for Bethesda Pharmaceuticals is also acknowledged for partial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chittiboyina, A.G., Mizuno, C.S., Desai, P.V. et al. Design, synthesis, and docking studies of novel telmisartan–glitazone hybrid analogs for the treatment of metabolic syndrome. Med Chem Res 18, 589–610 (2009). https://doi.org/10.1007/s00044-008-9152-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-008-9152-x