Abstract
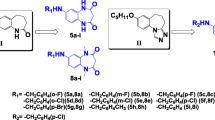
A series of 3-substituted-4-(4-hexyloxyphenyl)-4H-1,2,4-triazole derivatives (3a-s) were synthesized as open-chain analogues of 7-hexyloxyl-4,5-dihydro-[1,2,4]triazolo[4,3-a]quinolines (1c) using 4-hexyloxyaniline, acyl hydrazines, and dimethoxy-N,N-dimethylmethanamine as the starting material. Their anticonvulsant activities were evaluated by the maximal electroshock test (MES test) and their neurotoxicity was evaluated by the rotarod neurotoxicity test (Tox). MES test showed that all open-chain compounds exhibited strong anticonvulsant activity and lower neurotoxicity, and that some possessed obviously stronger activity than compound 1c. Compound 3d, 3-propyl-4-(4-hexyloxyphenyl)-4H-1,2,4-triazole was found to be the most potent with an ED50 value of 5.7 mg/kg and protective index (PI = TD50/ED50) value of 11.5, which was much greater than that of the prototype drug phenytoin (PI = 6.9).

Similar content being viewed by others
References
Al-Soud YA, Al-Masoudi NA, Ferwanah A (2003) Synthesis and properties of new substituted 1,2,4-triazoles: potential antitumor agents. Bioorg Med Chem 11(8):1701–1708
Chen J, Sun XY, Chai KY, Lee JS, Song MS, Quan ZS (2007) Synthesis and anticonvulsant evaluation of 4-(4-alkoxylphenyl)-3-ethyl-4H-1,2,4-triazoles as open-chain analogues of 7-alkoxyl-4,5-dihydro[1,2,4] triazolo[4,3-a]quinolines. Bioorg Med Chem 15:6775–6781
Cui LJ, Xie ZF, Piao HR, Li G, Chai KY, Quan ZS (2005) Synthesis and anticonvulsant activity of 1-substituted-7-benzyloxy-4,5-dihydro-[1,2,4]triazolo[4,3-a]quinoline. Biol Pharm Bull 28:1216–1220
Krall RJ, Penry JK, White BG, Kupferberg HJ, Swinyard EA (1978) Antiepileptic drug development: II. Anticonvulsant drug screening. Epilepsia 19:409–428
Lin Z, Kadaba PK (1997) Molecular targets for the rational design of antiepileptic drugs and related neuroprotective agents. Med Res Rev 17:537–572
Loscher W (1998) New visions in the pharmacology of anticonvulsion. Eur J Pharmacol 342:1–13
Perucca E (1996) The new generation of antiepileptic drugs: advantages and disadvantages. Br J Clin Pharmacol 42:531–543
Porter RJ, Cereghino JJ, Gladding GD, Hessie BJ, Kupferberg HJ, Scoville B (1984) Antiepileptic Drug Development Program. Clev Clin Q 51:293–305
Quan ZS, Wang JM, Rho JR, Kwak KC, Kang HC, Jun CS, Chai KY (2005) Synthesis and anticonvulsant activity of 7-alkoxyl-3.4-dihydro-qunoline-2-ones. Bull Kor Chem Soc 26:1757
Stocks MJ, Cheshire DR, Reynolds R (2004) Efficient and regiospecific one-pot synthesis of substituted 1,2,4-triazoles. Org Lett 6:2969–2971
Xie ZF, Chai KY, Piao HR, Kwak KC, Quan ZS (2005) Synthesis and anticonvulsant activity of 7-alkoxyl-4,5-dihydro-[1,2,4]triazolo[4,3-a]quinolines. Bioorg Med Chem Lett 15:4803–4805
Acknowledgments
This work was supported by the National Natural Science Foundation of China (no. 30460151) and Important Item Foundation of Ministry of Education P.R. China (no. 20070422029).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cui, XS., Chen, J., Chai, KY. et al. Synthesis and anticonvulsant evaluation of 3-substituted-4-(4-hexyloxyphenyl)-4H-1,2,4-triazoles. Med Chem Res 18, 49–58 (2009). https://doi.org/10.1007/s00044-008-9106-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-008-9106-3