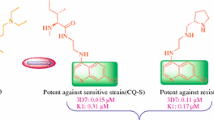
Abstract
Synthesis and evaluation of the antimalarial activity of new pyridine quinoline hybrid molecules against a chloroquine-susceptible strain of Plasmodium falciparum and as inhibitors of β-hematin formation are described. Two novel pyridine quinoline hybrid molecules and one bisquinoline molecule were synthesized and purified by column chromatography and evaluated for antimalarial and haem polymerization inhibition (HPIA) activities. The molecules were found to be very good haem polymerization inhibitors but showed poor antimalarial activity. This was attributed to their low vacuole accumulation ratio (VAR) in comparison to chloroquine. These molecules can be used as templates for designing new antimalarials targeting haem detoxification pathway of malaria parasite.
Graphical Abstract
Synthesis and antimalarial evaluation of novel pyridine quinoline hybrids
B.N Acharya, D. Thavaselvam# and M.P Kaushik*
Discovery Centre, Process Technology Development Division
Defence R & D Establishment, Jhansi Road, Gwalior-474002 (MP) INDIA
#Division of Microbiology, Defence Research and Development Establishment,Gwalior-474002, India

Synthesis and evaluation of antimalarial activity of new pyridine–quinoline hybrid molecules against a chloroquine-susceptible strain of Plasmodium falciparum and as inhibitors of β-hematin formation are described.



Similar content being viewed by others
References
Basilico N, Pagini E, Monti D, Olliaro P, Taramelli D (1998) A microtitre-based method for measuring the haem polymerization inhibitory activity (HPIA) of antimalarial drugs. J Antimicrob Chemother 42:55–60
Bray PG, Hawley SR, Mungthin M, Ward SA (1996) Physicochemical properties correlated with drug resistance and the reversal of drug resistance in Plasmodium falciparum. Mol Pharmacol 50:1559–1566
Dorn A, Stoffel R, Matile H, Bubendorf A, Ridley R (1995) Malarial haemozoin/β-haematin supports haem polymerization in the absence of protein. Nature 374:269–271
Dorn A, Vippagunta SR, Matile H, Jacquet C, Vennerstrom JL, Ridley RG (1998) An assessment of drug-haematin binding as a mechanism for inhibition of haematin polymerisation by quinoline antimalarials. Biochem Pharmacol 55:727–736
Egan TJ, Marques HM (1999) The role of haem in the activity of chloroquine and related antimalarial drugs. Coord Chem Rev 190–192:493–517
Egan TJ, Mavuso WW, Ross DC, Marques HM (1997) Thermodynamic factors controlling the interaction of quinoline antimalarial drugs with ferriprotoporphyrin IX. J Inorg Biochem 68:137–145
Fidock DA, Nomura T, Talley AK, Cooper RA, Dzekunov SM, Ferdig MT, Ursos LMB, Sidhu ABS, Naude KW, Deitsch KW, Su X, Wootton JC, Roepe PD, Wellems TE (2000) Mutations in the P. falciparum digestive food vacuole transmembrane protein PfCRT and evidence for their role in chloroquine resistance. Mol Cell 6:861–871
Goldberg DE, Slater AFG (1992) The pathway of hemoglobin degradation in malaria parasites. Parasitol Today 8:280–283
Hawley SR, Bray PG, Mungthin M, Atkinson J.D, O’Neill PM, Ward SA (1998) Relationship between antimalarial drug activity, accumulation, and inhibition of heme polymerization in Plasmodium falciparum in vitro. Antimicrob Agent Chemother 42:682–686
Hawley S, Bray PC, O’neill PM, Park BK, Ward SA (1996) The role of drug accumulation in 4-aminoquinoline antimalarial potency: the role of structural substitution and physicochemical properties. Biochem Pharmacol 52:723–733
Holland JH (1975) Adaption in natural and artificial system. Ann Arbor, University of Michigan Press
Pagola S, Stephens PW, Bohle DS, Kosar AD, Madsen SK (2000) The structure of malaria pigment β-haematin. Nature 404:307–310
Portela C, Afonso CMM, Pinto MM, Ramos MJ (2003) Receptor–drug association studies in the inhibition of the hematin aggregation process of malaria. FEBS Lett 27435:217–222
Sherman IW (1977) Amino acid metabolism and protein synthesis in malarial parasites. WHO Bull 55:265–276
Solmonov I, Osipova I, Feldman Y, Baehtz C, Kjaer K, Robinson IK, Webster GT, McNaughton D, Wood BR, Weissbuch I, Leiserowitz (2007) Crystal nucleation, growth and morphology of the synthetic malaria pigment β-hematin and the effect thereon by quinoline additives: the malaria pigment as a target of various antimalarial drugs. J Am Chem Soc 129:2615–2627
Trager W, Jensen JB (1976) Human malaria parasite in continuous culture. Science 193:673–675
Acknowledgements
We thank Dr. R. Vijayaraghavan, Director DRDE, Gwalior for his keen interest and encouragement in the present work. We especially thank Mr. Avik Mazumdar and Mr. Basant Lal for NMR analysis and Mr. Asish K. Srivastav for FTIR and ESI-MS analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Narayan Acharya, B., Thavaselvam, D. & Parshad Kaushik, M. Synthesis and antimalarial evaluation of novel pyridine quinoline hybrids. Med Chem Res 17, 487–494 (2008). https://doi.org/10.1007/s00044-008-9092-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-008-9092-5