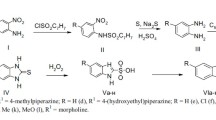
Geometric cis, trans isomers, derivatives of 2-substituted-1,3-dioxanes were designed and studied as antimuscarinic agents. The synthesized compounds were evaluated as perchlorides and methiodides by functional tests with rabbit vas deferens (putative M1), guinea-pig heart (M2) and guinea-pig ileum (M3). The effect of the replacement of a trimethylammonium group with a dimethysulfonium in the two rings was also evaluated. Pharmacological results indicate that the 1,3-dioxane nucleus shows the highest stereoselective values on the studied receptors.
Similar content being viewed by others
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Marucci, G., Piero, A., Brasili, L. et al. Synthesis and Antimuscarinic Activity of Derivatives of 2-Substituted-1,3-Dioxolanes. Med Chem Res 14, 274–296 (2005). https://doi.org/10.1007/s00044-005-0139-6
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s00044-005-0139-6