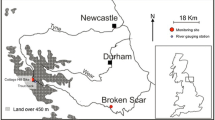
Abstract
Peatlands export significant amounts of dissolved organic carbon (DOC) to freshwaters, but the quantity of DOC reaching marine environments is typically less than the input to the fluvial system due to processing within the water column. Key removal processes include photo-chemical degradation, and heterotrophic bacterial respiration. In this study we examined these processes using 14C-labelled DOC to quantify the extent of DOC breakdown and to determine its fate following irradiation under controlled laboratory conditions. We examined the influence of microbial processes occurring within the water column, the potential role of stream-bed biofilms, and the possible modifying effects of downstream mixing, as DOC in water from the peatland encounters runoff from upland mineral soils (“Mountain”), nutrient-rich runoff from agricultural soils, and seawater in an estuary. Our results demonstrated conservative mixing of DOC from Peatland and Mountain waters but interactive effects when Peatland water was mixed with Agricultural and Estuary waters and exposed to solar radiation. The mixing of Peatland and Agricultural waters led to net DOC production, suggesting that DOC was only partially degraded by solar radiation and that the products of this might have fuelled autotrophic microbial growth in the samples. The mixing of Peatland water with saline estuary water resulted in net DOC loss following irradiation, suggesting a role for sunlight in enhancing the flocculation of DOC to particulate organic carbon (POC) in saline environments.






Similar content being viewed by others
References
Amaral JHF, Suhett AL, Melo S, Farjalla VF (2013) Seasonal variation and interaction of photodegradation and microbial metabolism of DOC in black water Amazonian ecosystems. Aquat Microb Ecol 70:157–168. doi:10.3354/ame01651
Asmala E, Bowers DG, Autio R et al (2014) Qualitative changes of riverine dissolved organic matter at low salinities due to flocculation. J Geophys Res-Biogeosciences 119:1919–1933. doi:10.1002/2014JG002722
Augspurger C, Gleixner G, Kramer C, Kuesel K (2008) Tracking carbon flow in a 2-week-old and 6-week-old stream biofilm food web. Limnol Oceanogr 53:642–650. doi:10.4319/lo.2008.53.2.0642
Austnes K, Evans CD, Eliot-Laize C, Naden PS, Old GH (2009) Effects of storm events on mobilisation and in-stream processing of dissolved organic matter (DOM) in a Welsh peatland catchment. Biogeochemistry 99:157–173
Baldwin DS, Whitworth KL, Hockley CL (2014) Uptake of dissolved organic carbon by biofilms provides insights into the potential impact of loss of large woody debris on the functioning of lowland rivers. Freshw Biol 59:692–702. doi:10.1111/fwb.12296
Battin TJ, Kaplan LA, Findlay S et al (2008) Biophysical controls on organic carbon fluxes in fluvial networks. Nat Geosci 1:95–100. doi:10.1038/ngeo101
Battin TJ, Luyssaert S, Kaplan LA et al (2009) The boundless carbon cycle. Nat Geosci 2:598–600. doi:10.1038/ngeo618
Berggren M, del Giorgio PA (2015) Distinct patterns of microbial metabolism associated to riverine dissolved organic carbon of different source and quality. J Geophys Res Biogeosci. 120:989–999. doi:10.1002/2015JG002963
Bertilsson S, Tranvik L (2000) Photochemical transformation of dissolved organic matter in lakes. Limnol Oceanogr 45:753–762
Billett MF, Deacon CM, Palmer SM et al (2006) Connecting organic carbon in stream water and soils in a peatland catchment. J Geophys Res-Biogeosciences 111:G02010. doi:10.1029/2005JG000065
Bischofberger T, Cha S, Schmitt R et al (1990) The bacterial-flora of noncarbonated, natural mineral water from the springs to reservoir and glass and plastic bottles. Int J Food Microbiol 11:51–72. doi:10.1016/0168-1605(90)90039-8
Brock TD, Clyne J (1984) Significance of algal excretory products for growth of epilimnetic bacteria. Appl Environ Microbiol 47:731–734
Cole JJ, Prairie YT, Caraco NF et al (2007) Plumbing the global carbon cycle: integrating inland waters into the terrestrial carbon budget. Ecosystems 10:171–184. doi:10.1007/s10021-006-9013-8
Cooper DM, Evans CD, Norris D, Thacker S, Pereira MG (2014) Application of a simple multiplicative stream water quality model to the river Conwy, North Wales. Environ Sci Process Impacts 16:1600–1607
Cory RM, Ward CP, Crump BC, Kling GW (2014) Sunlight controls water column processing of carbon in arctic fresh waters. Science 345:925–928. doi:10.1126/science.1253119
Dehaan H (1993) Solar UV-light penetration and photodegradation of humic substances in peaty lake water. Limnol Oceanogr 38:1072–1076
Del Giorgio PA, Duarte CM (2002) Respiration in the open ocean. Nature 420:379–384. doi:10.1038/nature01165
Evans CD, Renou-Wilson F, Strack M (2015) The role of waterborne carbon in the greenhouse gas balance of drained and re-wetted peatlands. Aquatic Sci (in press)
Fedotova AV, Serkebaeva YM, Sorokin VV, Dedysh SN (2013) Filterable microbial forms in the Rybinsk water reservoir. Microbiology 82:728–734. doi:10.1134/S0026261713060052
Findlay S, Sinsabaugh RL, Fischer DT, Franchini P (1998) Sources of dissolved organic carbon supporting planktonic bacterial production in the tidal freshwater Hudson River. Ecosystems 1:227–239. doi:10.1007/s100219900018
Freeman C, Lock MA (1995) The biofilm polysaccharide matrix: a buffer against changing organic substrate supply? Limnol Oceanogr 40:273–278. doi:10.4319/lo.1995.40.2.0273
Goldman J, Dennett M (1985) Susceptibility of some marine-phytoplankton species to cell breakage during filtration and post-filtration rinsing. J Exp Mar Biol Ecol 86:47–58. doi:10.1016/0022-0981(85)90041-3
Graneli W, Lindell M, De Faria BM, Esteves FD (1998) Photoproduction of dissolved inorganic carbon in temperate and tropical lakes—dependence on wavelength band and dissolved organic carbon concentration. Biogeochemistry 43:175–195. doi:10.1023/A:1006042629565
Hammes F, Vital M, Egli T (2010) Critical evaluation of the volumetric “Bottle Effect” on microbial batch growth. Appl Environ Microbiol 76:1278–1281. doi:10.1128/AEM.01914-09
Hedges JI, Keil RG, Benner R (1997) What happens to terrestrial organic matter in the ocean? Org Geochem 27:195–212. doi:10.1016/S0146-6380(97)00066-1
Hellebust JA (1965) Excretion of some organic compounds by marine phytoplankton. Limnol Oceanogr 10:192–206
Helms JR, Mao J, Schmidt-Rohr K et al (2013) Photochemical flocculation of terrestrial dissolved organic matter and iron. Geochim Cosmochim Acta 121:398–413. doi:10.1016/j.gca.2013.07.025
Hill PW, Marshall C, Williams GG et al (2007) The fate of photosynthetically-fixed carbon in Lolium perenne grassland as modified by elevated CO2 and sward management. New Phytol 173:766–777. doi:10.1111/j.1469-8137.2007.01966.x
Hill PW, Farrar JF, Jones DL (2008) Decoupling of microbial glucose uptake and minerlization in soil. Soil Biol Biochem 40:616–624. doi:10.1016/j.soilbio.2007.09.008
Jack J, Sellers T, Bukaveckas PA (2002) Algal production and trihalomethane formation potential: an experimental assessment and inter-river comparison. Can J Fish Aquat Sci 59:1482–1491. doi:10.1139/F02-121
Koehler B, Landelius T, Weyhenmeyer GA et al (2014) Sunlight-induced carbon dioxide emissions from inland waters. Glob Biogeochem Cycles. doi:10.1002/2014GB004850
Lee C, Henrichs SM (1993) How the nature of dissolved organic matter might affect the analysis of dissolved organic carbon. Mar Chem 41:105–120. doi:10.1016/0304-4203(93)90109-2
Malinsky-Rushansky NZ, Legrand C (1996) Excretion of dissolved organic carbon by phytoplankton of different sizes and subsequent bacterial uptake. Mar Ecol Prog Ser 132:249–255. doi:10.3354/meps132249
Meyer J, Edwards R, Risley R (1987) Bacterial-growth on dissolved organic-carbon from a blackwater river. Microb Ecol 13:13–29. doi:10.1007/BF02014960
Molot LA, Hudson JJ, Dillon PJ, Miller SA (2005) Effect of pH on photo-oxidation of dissolved organic carbon by hydroxyl radicals in a coloured, softwater stream. Aquat Sci 67:189–195. doi:10.1007/s00027-005-0754-9
Moody CS, Worrall F, Evans CD, Jones TG (2013) The rate of loss of dissolved organic carbon (DOC) through a catchment. J Hydrol 492:139–150. doi:10.1016/j.jhydrol.2013.03.016
Moran MA, Zepp RG (1997) Role of photoreactions in the formation of biologically labile compounds from dissolved organic matter. Limnol Oceanogr 42:1307–1316
Osburn CL, Morris DP, Thorn KA, Moeller RE (2001) Chemical and optical changes in freshwater dissolved organic matter exposed to solar radiation. Biogeochemistry 54:251–278. doi:10.1023/A:1010657428418
Palmer SM, Evans CD, Chapman PJ et al (2015) Sporadic hotspots for physico-chemical retention of aquatic organic carbon: from peatland headwater source to sea. Aquat Sci. doi:10.1007/s00027-015-0448-x
Roulet NT, Lafleur PM, Richard PJH et al (2007) Contemporary carbon balance and late Holocene carbon accumulation in a northern peatland. Global Change Biol 13:397–411. doi:10.1111/j.1365-2486.2006.01292.x
Spencer R, Ahad J, Baker A et al (2007) The estuarine mixing behaviour of peatland derived dissolved organic carbon and its relationship to chromophoric dissolved organic matter in two North Sea estuaries (U.K.). Estuar Coast Shelf Sci 74:131–144. doi:10.1016/j.ecss.2007.03.032
Tank JL, Dodds WK (2003) Nutrient limitation of epilithic and epixylic biofilms in ten North American streams. Freshw Biol 48:1031–1049. doi:10.1046/j.1365-2427.2003.01067.x
Teira E, Pazo MJ, Serret P, Fernandez E (2001) Dissolved organic carbon production by microbial populations in the Atlantic Ocean. Limnol Oceanogr 46:1370–1377
Tranvik LJ, Downing JA, Cotner JB et al (2009) Lakes and reservoirs as regulators of carbon cycling and climate. Limnol Oceanogr 54:2298–2314. doi:10.4319/lo.2009.54.6_part_2.2298
Uselman SM, Qualls RG, Lilienfein J (2007) Contribution of root vs. leaf litter to dissolved organic carbon leaching through soil. Soil Sci Soc Am J 71:1555–1563. doi:10.2136/sssaj2006.0386
Vähätalo AV, Salonen K, Munster U et al (2003) Photochemical transformation of allochthonous organic matter provides bioavailable nutrients in a humic lake. Arch Hydrobiol 156:287–314. doi:10.1127/0003-9136/2003/0156-0287
Volk C, Wood L, Johnson B et al (2002) Monitoring dissolved organic carbon in surface and drinking waters. J Environ Monit 4:43–47. doi:10.1039/b107768f
Wang Y, Hammes F, Boon N, Egli T (2007) Quantification of the filterability of freshwater bacteria through 0.45, 0.22, and 0.1 mu m pore size filters and shape-dependent enrichment of filterable bacterial communities. Environ Sci Technol 41:7080–7086. doi:10.1021/es0707198
Wetzel RG, Hatcher PG, Bianchi TS (1995) Natural photolysis by ultraviolet irradiance of recalcitrant dissolved organic matter to simple substrates for rapid bacterial metabolism. Limnol Oceanogr 40:1369–1380
Wu FC, Mills RB, Cai YR et al (2005) Photodegradation-induced changes in dissolved organic matter in acidic waters. Can J Fish Aquat Sci 62:1019–1027. doi:10.1139/F05-009
Ye L, Shi X, Wu X, Zhang M, Yu Y, Li D, Kong F (2011) Dynamics of dissolved organic carbon after a cyanobacterial bloom in hypereutrophic Lake Taihu (China). Limnol Ecol Manag Inland Waters 41(4):382–388
Zepp RG (1988) Environmental photoprocesses involving natural organic matter. In: Frimmel FH, Christmas RF (eds) Humic substances and their role in the environment. Wiley, New York, pp 193–214
Zepp RG, Faust BC, Hoigne J (1992) Hydroxyl radical formation in aqueous reactions (pH 3-8) of iron(II) with hydrogen peroxide: the photo-Fenton reaction. Environ Sci Technol 26:313–319. doi:10.1021/es00026a011
Ziegler SE, Lyon DR (2010) Factors regulating epilithic biofilm carbon cycling and release with nutrient enrichment in headwater streams. Hydrobiologia 657:71–88. doi:10.1007/s10750-010-0296-6
Zlotnik I, Dubinsky Z (1989) The effect of light and temperature on DOC excretion by Phytoplankton. Limnol Oceanogr 34:831–839
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the special issue ‘Carbon Cycling in Aquatic Ecosystems’.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jones, T.G., Evans, C.D., Jones, D.L. et al. Transformations in DOC along a source to sea continuum; impacts of photo-degradation, biological processes and mixing. Aquat Sci 78, 433–446 (2016). https://doi.org/10.1007/s00027-015-0461-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00027-015-0461-0