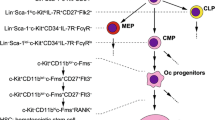
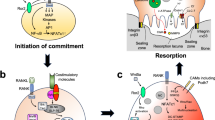
Abstract
Osteoclasts are bone-resorbing cells that play an essential role in bone remodeling. Defects in osteoclasts result in unbalanced bone remodeling and are linked to many bone diseases including osteoporosis, rheumatoid arthritis, primary bone cancer, and skeletal metastases. Receptor activator of NF-kappaB ligand (RANKL) is a classical inducer of osteoclast formation. In the presence of macrophage-colony-stimulating factor, RANKL and co-stimulatory signals synergistically regulate osteoclastogenesis. However, recent discoveries of alternative pathways for RANKL-independent osteoclastogenesis have led to a reassessment of the traditional mechanisms that regulate osteoclast formation. In this review, we provide an overview of signaling pathways and other regulatory elements governing osteoclastogenesis. We also identify how osteoclastogenesis is altered in pathological conditions and discuss therapeutic targets in osteoclasts for the treatment of skeletal diseases.



Similar content being viewed by others
References
Hancox NM (1949) The osteoclast. Biol Rev Camb Philos Soc 24:448–471
Takayanagi H (2007) Osteoimmunology: shared mechanisms and crosstalk between the immune and bone systems. Nat Rev Immunol 7:292–304
Lorenzo J, Horowitz M, Choi Y (2008) Osteoimmunology: interactions of the bone and immune system. Endocr Rev 29:403–440
McInnes IB, Schett G (2011) The pathogenesis of rheumatoid arthritis. N Engl J Med 365:2205–2219
Novack DV, Teitelbaum SL (2008) The osteoclast: friend or foe? Annu Rev Pathol 3:457–484
Weilbaecher KN, Guise TA, McCauley LK (2011) Cancer to bone: a fatal attraction. Nat Rev Cancer 11:411–425
Wong BR, Josien R, Lee SY, Sauter B, Li HL, Steinman RM et al (1997) TRANCE (tumor necrosis factor [TNF]-related activation-induced cytokine), a new TNF family member predominantly expressed in T cells, is a dendritic cell-specific survival factor. J Exp Med 186:2075–2080
Wong BR, Rho J, Arron J, Robinson E, Orlinick J, Chao M et al (1997) TRANCE is a novel ligand of the tumor necrosis factor receptor family that activates c-Jun N-terminal kinase in T cells. J Biol Chem 272:25190–25194
Lacey DL, Timms E, Tan HL, Kelley MJ, Dunstan CR, Burgess T et al (1998) Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell 93:165–176
Yasuda H, Shima N, Nakagawa N, Yamaguchi K, Kinosaki M, Mochizuki S et al (1998) Osteoclast differentiation factor is a ligand for osteoprotegerin/osteoclastogenesis-inhibitory factor and is identical to TRANCE/RANKL. Proc Natl Acad Sci USA 95:3597–3602
Kong YY, Yoshida H, Sarosi I, Tan HL, Timms E, Capparelli C et al (1999) OPGL is a key regulator of osteoclastogenesis, lymphocyte development and lymph-node organogenesis. Nature 397:315–323
Bachmann MF, Wong BR, Josien R, Steinman RM, Oxenius A, Choi Y (1999) TRANCE, a tumor necrosis factor family member critical for CD40 ligand-independent T helper cell activation. J Exp Med 189:1025–1031
Fata JE, Kong YY, Li J, Sasaki T, Irie-Sasaki J, Moorehead RA et al (2000) The osteoclast differentiation factor osteoprotegerin-ligand is essential for mammary gland development. Cell 103:41–50
Hanada R, Leibbrandt A, Hanada T, Kitaoka S, Furuyashiki T, Fujihara H et al (2009) Central control of fever and female body temperature by RANKL/RANK. Nature 462:505–509
Ikeda T, Kasai M, Utsuyama M, Hirokawa K (2001) Determination of three isoforms of the receptor activator of nuclear factor-kappaB ligand and their differential expression in bone and thymus. Endocrinology 142:1419–1426
Li J, Sarosi I, Yan XQ, Morony S, Capparelli C, Tan HL et al (2000) RANK is the intrinsic hematopoietic cell surface receptor that controls osteoclastogenesis and regulation of bone mass and calcium metabolism. Proc Natl Acad Sci USA 97:1566–1571
Dougall WC, Glaccum M, Charrier K, Rohrbach K, Brasel K, De Smedt T et al (1999) RANK is essential for osteoclast and lymph node development. Genes Dev 13:2412–2424
Darnay BG, Ni J, Moore PA, Aggarwal BB (1999) Activation of NF-kappaB by RANK requires tumor necrosis factor receptor-associated factor (TRAF) 6 and NF-kappaB-inducing kinase. Identification of a novel TRAF6 interaction motif. J Biol Chem 274:7724–7731
Lomaga MA, Yeh WC, Sarosi I, Duncan GS, Furlonger C, Ho A et al (1999) TRAF6 deficiency results in osteopetrosis and defective interleukin-1, CD40, and LPS signaling. Genes Dev 13:1015–1024
Naito A, Azuma S, Tanaka S, Miyazaki T, Takaki S, Takatsu K et al (1999) Severe osteopetrosis, defective interleukin-1 signalling and lymph node organogenesis in TRAF6-deficient mice. Genes Cells 4:353–362
Mizukami J, Takaesu G, Akatsuka H, Sakurai H, Ninomiya-Tsuji J, Matsumoto K et al (2002) Receptor activator of NF-kappaB ligand (RANKL) activates TAK1 mitogen-activated protein kinase kinase kinase through a signaling complex containing RANK, TAB 2, and TRAF6. Mol Cell Biol 22:992–1000
Huang H, Ryu J, Ha J, Chang EJ, Kim HJ, Kim HM et al (2006) Osteoclast differentiation requires TAK1 and MKK6 for NFATc1 induction and NF-kappaB transactivation by RANKL. Cell Death Differ 13:1879–1891
Wada T, Nakashima T, Oliveira-dos-Santos AJ, Gasser J, Hara H, Schett G et al (2005) The molecular scaffold Gab2 is a crucial component of RANK signaling and osteoclastogenesis. Nat Med 11:394–399
Tanaka S, Nakamura K, Takahasi N, Suda T (2005) Role of RANKL in physiological and pathological bone resorption and therapeutics targeting the RANKL–RANK signaling system. Immunol Rev 208:30–49
Grigoriadis AE, Wang ZQ, Cecchini MG, Hofstetter W, Felix R, Fleisch HA et al (1994) c-Fos: a key regulator of osteoclast-macrophage lineage determination and bone remodeling. Science 266:443–448
Takayanagi H (2007) The role of NFAT in osteoclast formation. Ann N Y Acad Sci 1116:227–237
Takayanagi H, Kim S, Koga T, Nishina H, Isshiki M, Yoshida H et al (2002) Induction and activation of the transcription factor NFATc1 (NFAT2) integrate RANKL signaling in terminal differentiation of osteoclasts. Dev Cell 3:889–901
Aliprantis AO, Ueki Y, Sulyanto R, Park A, Sigrist KS, Sharma SM et al (2008) NFATc1 in mice represses osteoprotegerin during osteoclastogenesis and dissociates systemic osteopenia from inflammation in cherubism. J Clin Invest 118:3775–3789
Mocsai A, Humphrey MB, Van Ziffle JA, Hu Y, Burghardt A, Spusta SC et al (2004) The immunomodulatory adapter proteins DAP12 and Fc receptor gamma-chain (FcRgamma) regulate development of functional osteoclasts through the Syk tyrosine kinase. Proc Natl Acad Sci USA 101:6158–6163
Koga T, Inui M, Inoue K, Kim S, Suematsu A, Kobayashi E et al (2004) Costimulatory signals mediated by the ITAM motif cooperate with RANKL for bone homeostasis. Nature 428:758–763
Shinohara M, Koga T, Okamoto K, Sakaguchi S, Arai K, Yasuda H et al (2008) Tyrosine kinases Btk and Tec regulate osteoclast differentiation by linking RANK and ITAM signals. Cell 132:794–806
Lowe C, Yoneda T, Boyce BF, Chen H, Mundy GR, Soriano P (1993) Osteopetrosis in Src-deficient mice is due to an autonomous defect of osteoclasts. Proc Natl Acad Sci USA 90:4485–4489
Soriano P, Montgomery C, Geske R, Bradley A (1991) Targeted disruption of the c-src proto-oncogene leads to osteopetrosis in mice. Cell 64:693–702
Zou W, Reeve JL, Liu Y, Teitelbaum SL, Ross FP (2008) DAP12 couples c-Fms activation to the osteoclast cytoskeleton by recruitment of Syk. Mol Cell 31:422–431
Kim HS, Kim DK, Kim AR, Mun SH, Lee SK, Kim JH et al (2012) Fyn positively regulates the activation of DAP12 and FcRgamma-mediated costimulatory signals by RANKL during osteoclastogenesis. Cell Signal 24:1306–1314
Mao D, Epple H, Uthgenannt B, Novack DV, Faccio R (2006) PLCgamma2 regulates osteoclastogenesis via its interaction with ITAM proteins and GAB2. J Clin Invest 116:2869–2879
Choi HK, Kang HR, Jung E, Kim TE, Lin JJ, Lee SY (2013) Early estrogen-induced gene 1, a novel RANK signaling component, is essential for osteoclastogenesis. Cell Res 23:524–536
Zhou Z, Immel D, Xi CX, Bierhaus A, Feng X, Mei L et al (2006) Regulation of osteoclast function and bone mass by RAGE. J Exp Med 203:1067–1080
Kaifu T, Nakahara J, Inui M, Mishima K, Momiyama T, Kaji M et al (2003) Osteopetrosis and thalamic hypomyelinosis with synaptic degeneration in DAP12-deficient mice. J Clin Invest 111:323–332
Paloneva J, Mandelin J, Kiialainen A, Bohling T, Prudlo J, Hakola P et al (2003) DAP12/TREM2 deficiency results in impaired osteoclast differentiation and osteoporotic features. J Exp Med 198:669–675
Humphrey MB, Daws MR, Spusta SC, Niemi EC, Torchia JA, Lanier LL et al (2006) TREM2, a DAP12-associated receptor, regulates osteoclast differentiation and function. J Bone Miner Res 21:237–245
Park-Min KH, Ji JD, Antoniv T, Reid AC, Silver RB, Humphrey MB et al (2009) IL-10 suppresses calcium-mediated costimulation of receptor activator NF-kappa B signaling during human osteoclast differentiation by inhibiting TREM-2 expression. J Immunol 183:2444–2455
Otero K, Shinohara M, Zhao H, Cella M, Gilfillan S, Colucci A et al (2012) TREM2 and beta-catenin regulate bone homeostasis by controlling the rate of osteoclastogenesis. J Immunol 188:2612–2621
Negishi-Koga T, Gober HJ, Sumiya E, Komatsu N, Okamoto K, Sawa S et al (2015) Immune complexes regulate bone metabolism through FcRgamma signalling. Nat Commun 6:6637
Lee MJ, Lim E, Mun S, Bae S, Murata K, Ivashkiv LB et al (2016) Intravenous immunoglobulin (IVIG) attenuates TNF-induced pathologic bone resorption and suppresses osteoclastogenesis by inducing A20 expression. J Cell Physiol 231:449–458
Simonet WS, Lacey DL, Dunstan CR, Kelley M, Chang MS, Luthy R et al (1997) Osteoprotegerin: a novel secreted protein involved in the regulation of bone density. Cell 89:309–319
Bucay N, Sarosi I, Dunstan CR, Morony S, Tarpley J, Capparelli C et al (1998) osteoprotegerin-deficient mice develop early onset osteoporosis and arterial calcification. Genes Dev 12:1260–1268
Takayanagi H, Kim S, Matsuo K, Suzuki H, Suzuki T, Sato K et al (2002) RANKL maintains bone homeostasis through c-Fos-dependent induction of interferon-beta. Nature 416:744–749
Luo J, Yang Z, Ma Y, Yue Z, Lin H, Qu G et al (2016) LGR4 is a receptor for RANKL and negatively regulates osteoclast differentiation and bone resorption. Nat Med 22:539–546
Takayanagi H, Ogasawara K, Hida S, Chiba T, Murata S, Sato K et al (2000) T-cell-mediated regulation of osteoclastogenesis by signalling cross-talk between RANKL and IFN-gamma. Nature 408:600–605
Ji JD, Park-Min KH, Shen Z, Fajardo RJ, Goldring SR, McHugh KP et al (2009) Inhibition of RANK expression and osteoclastogenesis by TLRs and IFN-gamma in human osteoclast precursors. J Immunol 183:7223–7233
Hayashi M, Nakashima T, Taniguchi M, Kodama T, Kumanogoh A, Takayanagi H (2012) Osteoprotection by semaphorin 3A. Nature 485:69–74
Ivashkiv LB, Zhao B, Park-Min KH, Takami M (2011) Feedback inhibition of osteoclastogenesis during inflammation by IL-10, M-CSF receptor shedding, and induction of IRF8. Ann N Y Acad Sci 1237:88–94
Ikeda K, Takeshita S (2016) The role of osteoclast differentiation and function in skeletal homeostasis. J Biochem 159:1–8
Indo Y, Takeshita S, Ishii KA, Hoshii T, Aburatani H, Hirao A et al (2013) Metabolic regulation of osteoclast differentiation and function. J Bone Miner Res 28:2392–2399
Jin Z, Wei W, Yang M, Du Y, Wan Y (2014) Mitochondrial complex I activity suppresses inflammation and enhances bone resorption by shifting macrophage-osteoclast polarization. Cell Metab 20:483–498
Zeng R, Faccio R, Novack DV (2015) Alternative NF-kappaB regulates RANKL-induced osteoclast differentiation and mitochondrial biogenesis via independent mechanisms. J Bone Miner Res 30(12):2287–2299
Ishii KA, Fumoto T, Iwai K, Takeshita S, Ito M, Shimohata N et al (2009) Coordination of PGC-1beta and iron uptake in mitochondrial biogenesis and osteoclast activation. Nat Med 15:259–266
Wei W, Wang X, Yang M, Smith LC, Dechow PC, Sonoda J et al (2010) PGC1beta mediates PPARgamma activation of osteoclastogenesis and rosiglitazone-induced bone loss. Cell Metab 11:503–516
Wan Y (2010) PPARgamma in bone homeostasis. Trends Endocrinol Metab 21:722–728
Wei W, Schwaid AG, Wang X, Wang X, Chen S, Chu Q et al (2016) Ligand activation of ERRalpha by cholesterol mediates statin and bisphosphonate effects. Cell Metab 23(3):479–491
Bae S, Lee MJ, Mun SH, Giannopoulou EG, Yong-Gonzalez V, Cross JR et al (2017) MYC-dependent oxidative metabolism regulates osteoclastogenesis via nuclear receptor ERRalpha. J Clin Invest 127:2555–2568
Nishikawa K, Iwamoto Y, Kobayashi Y, Katsuoka F, Kawaguchi S, Tsujita T et al (2015) DNA methyltransferase 3a regulates osteoclast differentiation by coupling to an S-adenosylmethionine-producing metabolic pathway. Nat Med 21:281–287
Cox TR, Rumney RMH, Schoof EM, Perryman L, Hoye AM, Agrawal A et al (2015) The hypoxic cancer secretome induces pre-metastatic bone lesions through lysyl oxidase. Nature 522:106–110
Tsukasaki M, Hamada K, Okamoto K, Nagashima K, Terashima A, Komatsu N et al (2017) LOX fails to substitute for RANKL in osteoclastogenesis. J Bone Miner Res 32:434–439
Schett G, Gravallese E (2012) Bone erosion in rheumatoid arthritis: mechanisms, diagnosis and treatment. Nat Rev Rheumatol 8:656–664
Kitaura H, Zhou P, Kim HJ, Novack DV, Ross FP, Teitelbaum SL (2005) M-CSF mediates TNF-induced inflammatory osteolysis. J Clin Invest 115:3418–3427
Danks L, Komatsu N, Guerrini MM, Sawa S, Armaka M, Kollias G et al (2016) RANKL expressed on synovial fibroblasts is primarily responsible for bone erosions during joint inflammation. Ann Rheum Dis 75:1187–1195
Kobayashi K, Takahashi N, Jimi E, Udagawa N, Takami M, Kotake S et al (2000) Tumor necrosis factor alpha stimulates osteoclast differentiation by a mechanism independent of the ODF/RANKL–RANK interaction. J Exp Med 191:275–286
Azuma Y, Kaji K, Katogi R, Takeshita S, Kudo A (2000) Tumor necrosis factor-alpha induces differentiation of and bone resorption by osteoclasts. J Biol Chem 275:4858–4864
Lam J, Takeshita S, Barker JE, Kanagawa O, Ross FP, Teitelbaum SL (2000) TNF-alpha induces osteoclastogenesis by direct stimulation of macrophages exposed to permissive levels of RANK ligand. J Clin Invest 106:1481–1488
Kim N, Kadono Y, Takami M, Lee J, Lee SH, Okada F et al (2005) Osteoclast differentiation independent of the TRANCE–RANK–TRAF6 axis. J Exp Med 202:589–595
Yokota K, Sato K, Miyazaki T, Kitaura H, Kayama H, Miyoshi F et al (2014) Combination of tumor necrosis factor alpha and interleukin-6 induces mouse osteoclast-like cells with bone resorption activity both in vitro and in vivo. Arthritis Rheumatol 66:121–129
O’Brien W, Fissel BM, Maeda Y, Yan J, Ge X, Gravallese EM et al (2016) RANK-independent osteoclast formation and bone erosion in inflammatory arthritis. Arthritis Rheumatol 68:2889–2900
Cohen SB, Dore RK, Lane NE, Ory PA, Peterfy CG, Sharp JT et al (2008) Denosumab treatment effects on structural damage, bone mineral density, and bone turnover in rheumatoid arthritis: a twelve-month, multicenter, randomized, double-blind, placebo-controlled, phase II clinical trial. Arthritis Rheum 58:1299–1309
Chiu YG, Ritchlin CT (2017) Denosumab: targeting the RANKL pathway to treat rheumatoid arthritis. Expert Opin Biol Ther 17:119–128
Usategui-Martin R, Calero-Paniagua I, Garcia-Aparicio J, Corral-Gudino L, Del Pino Montes J, Gonzalez Sarmiento R (2016) VAV3 gene polymorphism is associated with Paget’s disease of bone. Genet Test Mol Biomark 20:335–337
Faccio R, Teitelbaum SL, Fujikawa K, Chappel J, Zallone A, Tybulewicz VL et al (2005) Vav3 regulates osteoclast function and bone mass. Nat Med 11:284–290
Susani L, Pangrazio A, Sobacchi C, Taranta A, Mortier G, Savarirayan R et al (2004) TCIRG1-dependent recessive osteopetrosis: mutation analysis, functional identification of the splicing defects, and in vitro rescue by U1 snRNA. Hum Mutat 24:225–235
Frattini A, Pangrazio A, Susani L, Sobacchi C, Mirolo M, Abinun M et al (2003) Chloride channel ClCN7 mutations are responsible for severe recessive, dominant, and intermediate osteopetrosis. J Bone Miner Res 18:1740–1747
Sobacchi C, Schulz A, Coxon FP, Villa A, Helfrich MH (2013) Osteopetrosis: genetics, treatment and new insights into osteoclast function. Nat Rev Endocrinol 9:522–536
Verkman AS, Galietta LJ (2009) Chloride channels as drug targets. Nat Rev Drug Discov 8:153–171
Lacey DL, Boyle WJ, Simonet WS, Kostenuik PJ, Dougall WC, Sullivan JK et al (2012) Bench to bedside: elucidation of the OPG–RANK–RANKL pathway and the development of denosumab. Nat Rev Drug Discov 11:401–419
Toulis KA, Anastasilakis AD (2010) Increased risk of serious infections in women with osteopenia or osteoporosis treated with denosumab. Osteoporos Int 21:1963–1964
Acknowledgements
This work is supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health under Award numbers R01 AR069562 and AR073156. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park-Min, KH. Mechanisms involved in normal and pathological osteoclastogenesis. Cell. Mol. Life Sci. 75, 2519–2528 (2018). https://doi.org/10.1007/s00018-018-2817-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-018-2817-9