Abstract
An analysis of the scientific literature published in the last 10 years reveals a constant growth of laccase applicative research in several industrial fields followed by the publication of a great number of patents. The Green Chemistry journal devoted the cover of its September 2014 issue to a laccase as greener alternative for chemical oxidation. This indicates that laccase “never-ending story” has found a new promising trend within the constant search for efficient (bio)catalysts able to meet the 12 green chemistry principles. A survey of ancient and cutting-edge uses of laccase in different industrial sectors is offered in this review with the aim both to underline their potential and to provide inspiration for new ones. Applications in textile and food fields have been deeply described, as well as examples concerning polymer synthesis and laccase-catalysed grafting. Recent applications in pharmaceutical and cosmetic industry have also been reviewed.



Similar content being viewed by others
References
Giardina P, Faraco V, Pezzella C, Piscitelli A, Vanhulle S, Sannia G (2010) Laccases: a never-ending story. Cell Mol Life Sci 67(3):369–385
Piscitelli A, Pezzella C, Lettera V, Giardina P, Faraco V, Sannia G (2013) Fungal laccases: structure, function and application. In: Maria de Lourdes TMP and Rai M (eds) Fungal enzymes: progress and prospects. pp 113–151
Jeon JR, Baldrian P, Murugesan K, Chang YS (2012) Laccase-catalysed oxidations of naturally occurring phenols: from in vivo biosynthetic pathways to green synthetic applications. Microb Biotechnol 5(3):318–332
Jeon JR, Chang YS (2013) Laccase-mediated oxidation of small organics: bifunctional roles for versatile applications. Trends Biotechnol 31(6):335–341
Pereira L, Bastos C, Tzanov T, Cavaco-Paulo A, Guebitz GM (2005) Environmentally friendly bleaching of cotton using laccases. Environ Chem Lett 3:66–69
Tzanov T, Basto C, Gübitz GM, Cavaco-Paulo A (2003) Laccases to improve the whiteness in a conventional bleaching of Cotton. Macromol Mater Eng 288:807–810
Ren X, Buschle-Diller G (2007) Oxidoreductases for modification of linen fibers. Colloid Surface A 299:15–21
Basto C, Tzanov T, Cavaco-Paulo A (2007) Combined ultrasound-laccase assisted bleaching of cotton. Ultrason Sonochem 14:350–354
Abou-Okeil A, El-Shafie A, El Zawahry MM (2010) Ecofriendly laccase–hydrogen peroxide/ultrasound-assisted bleaching of linen fabrics and its influence on dyeing efficiency. Ultrason Sonochem 17:383–390
Goncalves I, Herrero-Yniesta V, Perales Arce I, Escrigas Castaneda M, Cavaco-Paulo A, Silva C (2014) Ultrasonic pilot-scale reactor for enzymatic bleaching of cotton fabrics. Ultrason Sonochem 21:1535–1543
Tian L, Branford-White C, Wang W, Nie H, Zhu L (2012) Laccase-mediated system pretreatment to enhance the effect of hydrogen peroxide bleaching of cotton fabric. Int J Biol Macromol 50:782–787
Rodriguez-Couto S (2012) Laccases for denim bleaching: an eco-friendly alternative. Open Textil J 5:1–7
Solís-Oba M, Almendáriz J, Viniegra-González G (2008) Biotechnological treatment for colorless denim and textil wastewater treatment with laccase and ABTS. Rev Int Contam ambient 24:5–11
Montazer M, Sadeghian Maryan A (2010) Influences of different enzymatic treatment on denim garment. Appl Biochem Biotech 160:2114–2128
Pazarlıoğlu NK, Sariişik M, Telefoncu A (2005) Laccase: production by Trametes versicolor and application to denim washing. Process Biochem 40:1673–1678
Hossain KhMG, González MD, Juan AR, Tzanov T (2010) Enzyme-mediated coupling of a bi-functional phenolic compound onto wool to enhance its physical, mechanical and functional properties. Enzyme Microb Tech 46:326–330
Schröder M, Aichernig N, Gübitz GM, Kokol V (2007) Enzymatic coating of lignocellulosic surfaces with polyphenols. Biotechnol J 2:334–341
Hossain KhMG, González MD, Lozano GR, Tzanov T (2009) Multifunctional modification of wool using an enzymatic process in aqueous-organic media. J Biotechnol 141:58–63
Silva C, Matamá T, Kim SY, Padrão J, Nugroho Prasetyo E, Kudanga T, Nyanhongo GS, Guebitz GM, Casal M, Cavaco-Paulo A (2011) Antimicrobial and antioxidant linen via laccase-assisted grafting. React Funct Polym 71:713–720
Kim SY, Cavaco-Paulo A (2012) Laccase-catalysed protein–flavonoid conjugates for flax fibre modification. Appl Microbiol Biotechnol 93:585–600
Yoon MY (1998) Process for improved shrink resistance in wool. WO Patent 98/27264
Lantto R, Schänberg C, Buchert J (2004) Effects of laccase-mediator combinations on wool. Textile Res J 74:713–717
Zhang Y, Dong A, Wang Q, Fan X, Cavaco-Paulo A, Zhang Y (2014) Conductive cotton prepared by polyaniline in situ polymerization using laccase. Appl Biochem Biotech 174:820–831
Tzanov T, Silva C, Zille A, Oliveira J, Cavaco-Paulo A (2003) Effect of some process parameters in enzymatic dyeing of wool. Appl Biochem Biotech 111:1–14
Hadzhiyska H, Calafell M, Gibert JM, Dagà JM, Tzanov T (2006) Laccase-assisted dyeing of cotton. Biotechnol Lett 28:755–759
Kim SY, Moldes D, Cavaco-Paulo A (2007) Laccase for enzymatic coloration of unbleached cotton. Enzyme Microb Tech 40:1788–1793
Suparno O, Covington AD, Evans CS (2007) Application of diphenols for dyeing. J Soc Leath Tech Ch 91:139–141
Kim SY, Lopez C, Guebitz GM, Cavaco-Paulo A (2008) Biological coloration of flax fabrics with flavonoids using laccase from Trametes hirsuta. Eng Life Sci 8:324–330
Blanco DC, González DM, Monmany DM, Tzanov T (2009) Dyeing properties, synthesis, isolation and characterization of an in situ generated phenolic pigment, covalently bound to cotton. Enzyme Microb Technol 44:380–385
Guimarães C, Kim S, Silva C, Cavaco-Paulo A (2011) In situ laccase-assisted overdyeing of denim using flavonoids. Biotechnol J 6:1272–1279
Kudanga T, Nyanhongo GS, Guebitz GM, Burton S (2011) Potential applications of laccase-mediated coupling and grafting reactions: a review. Enzyme Microb Technol 48:195–208
Polak J, Jarosz-Wilkołazka A (2012) Structure/redox potential relationship of simple organic compounds as potential precursors of dyes for laccase-mediated transformation. Biotechnol Prog 23:93–102
Polak J, Jarosz-Wilkolazka A (2010) Whole-cell fungal transformation of precursors into dyes. Microb Cell Fact 9:51
Ganachaud Ch, Garfagnoli V, Tron T, Iacazio G (2008) Trimerisation of indole through laccase catalysis. Tetrahedron Lett 49:2476–2478
Kim S, Silva C, Evtuguin DV, Gamelas JAF, Cavaco-Paulo A (2011) Polyoxometalate/laccase-mediated oxidative polymerization of catechol for textile dyeing. Appl Microbiol Biotechnol 89:981–987
Herter S, Mikolasch A, Michalik D, Hammer E, Schauer F et al (2011) C–N coupling of 3-methylcatechol with primary amines using native and recombinant laccases from Trametes versicolor and Pycnoporus cinnabarinus. Tetrahedron 67:9311–9321
Forte S, Polak J, Valensin D, Taddei M, Basosi R, Vanhulle S et al (2010) Synthesis and structural characterization of a novel phenoxazinone dye by use of a fungal laccase. J Mol Catal B Enzym 63:116–120
Bruyneel F, Enaud E, Billottet L, Vanhulle S, Marchand-Brynaert J (2008) Regioselective synthesis of 3-hydroxyorthanilic acid and its biotransformation into a novel phenoxazinone dye by use of laccase. Eur J Org Chem 2008:72–79
Bruyneel F, Payen O, Rescigno A, Tinant B, Marchand-Brynaert J (2009) Laccase mediated synthesis of novel substitutes phenoxazine chromophores featuring tuneable water solubility. Chem Eur J 15:8283–8295
Setti L, Giuliani S, Spinozzi G, Pifferi PG (1999) Laccase catalyzed-oxidative coupling of 3-methyl 2-benzothiazoline hydrazone and methoxyphenols. Enzyme Microb Tech 25:285–289
Martorana A, Bernini C, Valensin D, Sinicropi A, Pogni R, Basosi R et al (2011) Insights into the homocoupling reaction of 4-methylamino benzoic acid mediated by Trametes versicolor laccase. Mol BioSyst 7:2967–2969
Enaud E, Trovaslet M, Bruyneel F, Billottet L, Karaaslan R, Sener ME et al (2010) A novel azoanthraquinone dye made through innovative enzymatic process. Dye Pigments 85:99–108
Enaud E, Bols ChM, Casas Infantes A, Groslambert S, Hercher Ch, Iacazio G et al (2010) New azo dyes. WO Patent 2010/003969, 14 Jan 2010
Osma JF, Toca-Herrera JL, Rodriguez-Couto S (2010) Uses of laccases in the food industry. Enzyme Res 2010:918761
Labat E, Morel MH, Rouau X (2000) Wheat gluten phenolic acids: occurrence and fate upon mixing. J Agric Food Chem 48:6280–6283
Selinheimo E, Kruus K, Buchert J, Hopia A, Autio K (2006) Effects of laccase, xylanase and their combination on the rheological properties of wheat doughs. J Cereal Science 43:152–159
Flander L, Holopainen U, Kruus K, Buchert J (2011) Effects of tyrosinase and laccase on oat proteins and quality parameters of gluten-free oat breads. J Agr Food Chem 59:8385–8390
Flander L, Rouau X, Morel MH, Autio K, Seppanen-Laakso T, Kruus K, Buchert J (2008) Effects of laccase and xylanase on the chemical and rheological properties of oat and wheat doughs. J Agr Food Chem 56:5732–5742
Renzetti S, Courtin CM, Delcour JA, Arendt EK (2010) Oxidative and proteolytic enzyme preparations as promising improvers for oat bread formulations: rheological, biochemical and microstructural background. Food Chem 119:1465–1473
Tsuchiya R, Petersen BR, Christensen S (2000) Oxidoreductases for reduction of malodor. US Patent 6074631 A
Petersen BR, Mathiasen TE, Peelen B, Andersen H (1996) Use of laccase for deoxygenation of oil-containing product such as salad dressing. PCT international application WO 9635768 A1
Bouwens EM, Trivedi K, Van Vliet C, Winkel C (1999) Method of enhancing color in a tea based foodstuff. US Patent 5879730 A
Takemori T, Ito Y, Ito M, Yoshama M (1992) Flavor and taste improvement of cacao nib by enzymatic treatment. Japan Kokai Tokkyo Koho JP 04126037 A2
Mensah CA, Adamafio NA, Amaning-Kwarteng K, Rodrigues FK (2012) Reduced tannin content of laccase-treated cocoa (Theobroma cacao) pod husk. Int J Biol Chem 6:31–36
Tanriöven D, Ekşi A (2005) Phenolic compounds in pear juice from different cultivars. Food Chem 93:89–93
Minussi RC, Pastore GM, Durán N (2002) Potential applications of laccase in the food industry. Trends Food Sci Tech 13:205–216
Minussi RC, Rossi M, Bologna L, Rotilio D, Pastore GM, Durán N (2007) Phenols removal in musts: strategy for wine stabilization by laccase. J Mol Catal B-Enzym 45:102–107
Conrad LS, Sponholz WR, Berker O (2000) Treatment of cork with a phenol oxidizing enzyme. US Patent 6152966
Mathiasen TE (1995) Laccase and beer storage. PCT international application, WO 9521240 A2
Sammartino M, Piacquadio P, De Stefano G, Sciancalepore V (1998) Apple juice stabilization by conventional and innovative methods. Ind. Bevande 27:367–369
Giovanelli G, Ravasini G (1993) Apple juice stabilization by combined enzyme-membrane filtration process. Lebensm Wiss Technol 26:1–7
Gökmen V, Borneman Z, Nijhuis HH (1998) Improved ultrafiltration for color reduction and stabilization of apple juice. J Food Sci 63:504–507
Ritter G, Maier G, Schoepplein E, Dietrich H (1992) The application of polyphenoloxidase in the processing of apple juice. Bulletin de Liaison-Groupe Polyphenols 16:209–212
Cantarelli C, Giovanelli G (1990) Stabilization of pome and grape juice against phenolic deterioration by enzymic treatments. Inst Fruchtsaft Union Wiss Tech Komm 21:35–57
Neifar M, Ellouze-Ghorbel R, Kamoun A, Baklouti S, Mokni A, Jaouani A, Ellouze-Chaabouni S (2011) Effective clarification of pomegranate juice using laccase treatment optimized by response surface methodology followed by ultrafiltration. J Food Process Eng 34:1199–1219
Gassara-Chatti F, Brar SK, Ajila CM, Verma M, Tyagi RD, Valero JR (2013) Encapsulation of ligninolytic enzymes and its application in clarification of juice. Food Chem 137:18–24
Norsker M, Jensen M, Adler-Nissen J (2000) Enzymatic gelation of sugar beet pectin in food products. Food Hydrocolloid 14:237–243
Kuuva T, Lantto R, Reinikainen T, Buchert J, Autio K (2003) Rheological properties of laccase-induced sugar beet pectin gels. Food Hydrocolloid 17:679–684
Littoz F, McClements DJ (2008) Bio-mimetic approach to improving emulsion stability: cross-linking adsorbed beet pectin layers using laccase. Food Hydrocolloid 22:1203–1211
Zeeb B, Gibis M, Fischer L, Weiss J (2012) Crosslinking of interfacial layers in multilayered oil-in-water emulsions using laccase: characterization and pH-stability. Food Hydrocolloid 27:126–136
Jung J, Wicker L (2012) Laccase mediated conjugation of heat treated beta-lactoglobulin and sugar beet pectin. Carbohyd Polym 89:1244–1249
Ma H, Forssell P, Partanen R, Buchert J, Boer H (2011) Improving laccase catalyzed cross-linking of whey protein isolate and their application as emulsifiers. J Agric Food Chem 59:1406–1414
Gazme B, Madadlou A (2014) Fabrication of whey protein–pectin conjugate particles through laccase-induced gelation of microemulsified nanodroplets. Food Hydrocolloid 40:189–195
Mattinen ML, Kruus K, Buchert J, Nielsen JH, Andersen HJ, Steffensen CL (2005) Laccase-catalysed polymerization of tyrosine-containing peptides. FEBS J 272:3640–3650
Stanic D, Monogioudi E, Dilek E, Radosavljevic J, Atanaskovic-Markovic M, Vuckovic O, Raija L, Mattinen M, Buchert J, Cirkovic Velickovic T (2010) Digestibility and allergenicity assessment of enzymatically crosslinked beta-casein. Mol Nutr Food Res 54:1273–1284
Tantoush Z, Stanic D, Stojadinovic M, Ognjenovic J, Mihajlovic L, Atanaskovic-Markovic M, Cirkovic T (2011) Digestibility and allergenicity of b-lactoglobulin following laccase-mediated cross-linking in the presence of sour cherry phenolics. Food Chem 125:84–91
Kontopidis G, Holt C, Sawyer L (2004) Beta-lactoglobulin: binding properties, structure, and function. J Dairy Sci 87:785–796
Ghindilis AL, Gavrilova VP, Yaropolov AI (1992) Laccase-based biosensor for determination of polyphenols: determination of catechols in tea. Biosens Bioelectron 7:127–131
Di Fusco M, Tortolini C, Deriu D, Mazzei F (2010) Laccase-based biosensor for the determination of polyphenol index in wine. Talanta 81:235–240
Ibarra-Escutia P, Gomez JJ, Calas-Blanchard C, Marty JL, Ramirez-Silva MT (2010) Amperometric biosensor based on a high resolution photopolymer deposited onto a screen-printed electrode for phenolic compounds monitoring in tea infusions. Talanta 81:1636–1642
Eremia SA, Vasilescu I, Radoi A, Litescu SC, Radu GL (2013) Disposable biosensor based on platinum nanoparticles-reduced graphene oxide-laccase biocomposite for the determination of total polyphenolic content. Talanta 110:164–170
Medina-Plaza C, de Saja JA, Rodriguez-Mendez ML (2014) Bioelectronic tongue based on lipidic nanostructured layers containing phenol oxidases and lutetium bisphthalocyanine for the analysis of grapes. Biosens Bioelectron 57:276–283
Kobayashi S, Makino A (2009) Enzymatic polymer synthesis: an opportunity for green polymer chemistry. Chem Rev 109:5288–5353
Hollmann F, Arends IWCE (2012) Enzyme initiated radical polymerizations. Polymers 4:759–793
Mikolasch A, Schauer F (2009) Fungal laccases as tools for the synthesis of new hybrid molecules and biomaterials. Appl Microbiol Biotechnol 82:605–624
Hollmann F, Gumulya Y, Toelle C, Liese A, Thum O (2008) Evaluation of the laccase from Myceliophthora thermophila as industrial biocatalyst for polymerization reactions. Macromolecules 41:8520–8524
Walde P, Guo ZW (2011) Enzyme-catalyzed chemical structure-controlling template polymerization. Soft Matter 7:316–331
Mita N, Tawaki S, Uyama H, Kobayashi S (2002) Structural control in enzymatic oxidative polymerization of phenols with varying the solvent and substituent nature. Chem Lett 31:402–403
Ikeda R, Sugihara J, Uyama H, Kobayashi S (1998) Enzymatic oxidative polymerization of 4-hydroxybenzoic acid derivatives to poly(phenylene oxide)s. Polym Int 47:295–301
Mita N, Tawaki S, Uyama H, Kobayashi S (2004) Precise structure control of enzymatically synthesized polyphenols. Bull Chem Soc Jpn 77:1523–1527
Mita N, Tawaki SI, Uyama H, Kobayashi S (2003) Laccase-catalyzed oxidative polymerization of phenols. Macromol Biosci 3:253–257
Witayakran S, Ragauskas AJ (2009) Synthetic applications of laccase in green chemistry. Adv Synth Catal 35:1187–1209
Piscitelli A, Amore A, Faraco V (2012) Last advances in synthesis of added value compounds by laccase-mediated biocatalysis. Curr Org Chem 16:2508–2524
Uyama H (2007) Artificial polymeric flavonoids: synthesis and applications. Macromol Biosci 7:410–422
Es-Safi NE, Ghidouche S, Ducrot PH (2007) Flavonoids: hemisynthesis, reactivity, characterization and free radical scavenging activity. Molecules 12:2228–2258
Hagerman AE, Riedl KM, Jones GA, Sovik KN, Ritchard NT, Hartzfeld PW, Riechel TL (1998) High molecular weight plant polyphenolics (tannins) as biological antioxidants. J Agr Food Chem 46:1887–1892
Kurisawa M, Chung JE, Uyama H, Kobayashi S (2003) Laccase-catalyzed synthesis and antioxidant property of poly(catechin). Macromol Biosci 3:758–764
Jadhav SB, Singhal RS (2014) Laccase–gum Arabic conjugate for preparation of water-soluble oligomer of catechin with enhanced antioxidant activity. Food Chem 150:9–16
Desentis-Mendoza RM, Hernandez-Sanchez H, Moreno A, Emilio RDC, Chel-Guerrero L, Tamariz J, Jaramillo-Flores ME (2006) Enzymatic polymerization of phenolic compounds using laccase and tyrosinase from Ustilago maydis. Biomacromol 7:1845–1854
Kobayashi S, Uyama H, Ikeda R (2001) Artificial urushi. Chem Eur J 7:4755–4760
Kobayashi S, Ikeda R, Oyabu H, Tanaka H (2000) Artificial Urushi: design, synthesis, and enzymatic curing of new urushiol analogues. Chem Lett 29:1214–1215
Ikeda R, Tanaka H, Oyabu H, Uyama H, Kobayashi S (2001) Preparation of artificial urushi via an environmentally benign process. Bull Chem Soc Japan 74:1067–1073
Otrokhov GV, Morozova OV, Vasil’eva IS, Shumakovich GP, Zaitseva EA, Khlupova ME, Yaropolov AI (2013) Biocatalytic synthesis of conducting polymers and prospects for its application. Biochemistry (Moscow) 78:1539–1553
Karamyshev AV, Shleev SV, Koroleva OV, Yaropolov AI, Sakharov IY (2003) Laccase-catalyzed synthesis of conducting polyaniline. Enzyme Microb Technol 33:556–564
Vasil’eva IS, Morozova OV, Shumakovich GP, Yaropolov AI (2009) Synthesis of electroconductive polyaniline using immobilized laccase. Appl Biochem Microbiol 45:27–30
Shumakovich GP, Vasil’eva IS, Morozova OV, Khomenkov VG, Staroverova IN, Budashov IA, Kurochkin IN, Boyeva JA, Sergeyev VG, Yaropolov AI (2010) A comparative study of water dispersible polyaniline nanocomposites prepared by laccase-catalyzed and chemical methods. J Appl Polym Sci 117:1544–1550
Streltsov AV, Shumakovich GP, Morozova OV, Gorbacheva MA, Yaropolov AI (2008) Micellar laccase-catalyzed synthesis of electroconductive polyaniline. Appl Biochem Microb 44:264–270
Streltsov AV, Morozova OV, Arkharova NA, Klechkovskaya VV, Staroverova IN, Shumakovich GP, Yaropolov AI (2009) Synthesis and characterization of conducting polyaniline prepared by laccase-catalyzed method in sodium dodecylbenzenesulfonate micellar solutions. J Appl Pol Sci 114:928–934
Shumakovich G, Streltsov A, Gorshina E, Rusinova T, Kurova V, Vasil’eva I, Otrokhov G, Morozova O, Yaropolov A (2011) Laccase-catalyzed oxidative polymerization of aniline dimer (N-phenyl-1,4-phenylenediamine) in aqueous micellar solution of sodium dodecylbenzenesulfonate. J Mol Catal B Enzym 69:83–88
Shumakovich GP, Kurova V, Vasil’eva IS, Pankratov D, Otrokhov G, Morozova O, Yaropolov A (2012) Laccase-mediated synthesis of conducting polyaniline. J Mol Catal B Enzym 77:105–110
Junker K, Kissner R, Rakvin B, Guo Z, Willeke M, Busato S, Weber T, Walde P (2014) The use of Trametes versicolor laccase for the polymerization of aniline in the presence of vesicles as templates. Enzyme Microb Technol 55:72–84
Vasil’eva IS, Morozova OV, Shumakovich GP, Shleev SV, Sakharov IY, Yaropolov AI (2007) Laccase-catalyzed synthesis of optically active polyaniline. Synth Met 157:684–689
Shumakovich GP, Otrokhov G, Vasil’eva IS, Pankratov D, Morozova O, Yaropolov A (2012) Laccase-mediated polymerization of 3,4-ethylenedioxythiophene (EDOT). J Mol Catal B Enzym 81:66–68
George PM, Lyckman AW, LaVan DA, Hegde A, Leung Y, Avasare R, Testa C, Alexander PM, Langer R, Sur M (2005) Fabrication and biocompatibility of polypyrrole implants suitable for neural prosthetics. Biomaterials 26:3511–3519
Song YK, Palmore GTR (2005) Conductive Polypyrrole via enzyme catalysis. J Phys Chem B 109:19278–19287
López J, Alonso-Omlin EM, Hernández-Alcántara JM, Bárzana E, Gimeno M (2014) Novel photoluminescent material by laccase-mediated polymerization of 4-fluoroguaiacol throughout defluorination. J Mol Catal B Enzymatic 109:70–75
Nyanhongo GS, Kudanga T, Nugroho Prasetyo E, Gübitz GM (2011) Enzymatic polymer functionalisation: advances in laccase and peroxidase derived lignocellulose functional polymers. Adv Biochem Eng Biotechnol 125:47–68
Chandra RP, Ragauskas AJ (2001) Laccase: the renegade of fiber modification. In: Tappi pulping conference. pp 1041–1051
Chandra RP, Ragauskas AJ (2002) Elucidating the effects of laccase on the physical properties of high-kappa kraft pulps. Prog Biotechnol 21:165–172
Chandra RP, Wolfaardt F, Ragauskas AJ (2003) Biografting of celestine blue onto a high kappa kraft pulp. ACS Sym Ser 855:66–80
Chandra RP, Lehtonen LK, Ragauskas AJ (2004) Modification of high lignin content kraft pulps with laccase to improve paper strength properties. Laccase treatment in the presence of gallic acid. Biotechnol Prog 20:255–261
Chandra RP, Felby C, Ragauskas AJ (2004) Improving laccase-facilitated grafting of 4-hydroxybenzoic acid to high-kappa kraft pulps. J Wood Chem Technol 24:69–81
Witayakran S, Ragauskas AJ (2009) Modification of high-lignin softwood kraft pulp with laccase and amino acids. Enzyme Microb Technol 44:176–181
Fillat A, Colom JF, Vidal T (2010) A new approach to the biobleaching of flax pulp with laccase using natural mediators. Bioresour Technol 101:4104–4110
Aracri E, Fillat A, Colom JF, Gutiérrez A, Del Río JC, Martínez AT, Vidal T (2010) Enzymatic grafting of simple phenols on flax and sisal pulp fibres using laccases. Bioresour Technol 101:8211–8216
Aracri E, Roncero MB, Vidal T (2011) Studying the effects of laccase-catalysed grafting of ferulic acid on sisal pulp fibers. Bioresour Technol 102:7555–7560
Aracri E, Vidal T, Ragauskas AJ (2011) Wet strength development in sisal cellulose fibers by effect of a laccase–TEMPO treatment. Carbohyd Polym 84:1384–1390
Aracri E, Valls C, Vidal T (2012) Paper strength improvement by oxidative modification of sisal cellulose fibers with laccase–TEMPO system: influence of the process variables. Carbohyd Polym 88:830–837
Elegir G, Kindl A, Sadocco P, Orlandi M (2008) Development of antimicrobial cellulose packaging through laccase-mediated grafting of phenolic compounds. Enzyme Microb Technol 43:84–92
Fillat A, Gallardo O, Vidal T, Pastor FIJ, Díaz P, Roncero MB (2012) Enzymatic grafting of natural phenols to flax fibres: development of antimicrobial properties. Carbohyd Polym 87:146–152
Widsten P, Heathcote C, Kandelbauer A, Guebitz G, Nyanhongo GS, Nugroho Prasetyo E, Kudanga T (2010) Enzymatic surface functionalisation of lignocellulosic materials with tannins for enhancing antibacterial properties. Proc Biochem 45:1072–1081
Fackler K, Kuncinger T, Ters T, Srebotnik E (2008) Laccase-catalyzed functionalization with 4-hydroxy-3-methoxybenzylurea significantly improves internal bond of particle boards. Holzforschung 62:223–229
Kudanga T, Nugroho Prasetyo E, Sipilä J, Nyanhongo GS, Guebitz G (2010) Enzymatic grafting of functional molecules to the lignin model dibenzodioxocin and lignocellulose material. Enzyme Microb Technol 46:272–280
Kudanga T, Nugroho Prasetyo E, Widsten P, Kandelbauer A, Jury S, Heathcote C, Sipilä J, Weber H, Nyanhongo GS, Guebitz GM (2010) Laccase-catalyzed covalent coupling of fluorophenols increases lignocellulose surface hydrophobicity. Bioresour Technol 101:2793–2799
Kudanga T, Nugroho Prasetyo E, Sipilä J, Guebitz GM, Nyanhongo GS (2010) Reactivity of long chain alkylamines to lignin moieties: implications on hydrophobicity of lignocellulose materials. J Biotechnol 149:81–87
Widsten P, Kandelbauer A (2008) Laccase applications in the forest products industry: a review. Enzyme Microb Technol 42:293–307
Felby C, Pedersen LS, Nielsen BR (1997) Enhanced auto-adhesion of wood fibers using phenol oxidases. Holzforschung 51:281–286
Widsten P, Laine JE, Tuominen S, Qvintus-Leino P (2003) Effect of high defibration temperature on the properties of medium-density fiberboard (MDF) made from laccase-treated hardwood fibers. J Adhes Sci Technol 17:67–78
Felby C, Thygesen LG, Sanadi A, Barsberg S (2004) Native lignin for bonding of fiber boards-evaluation of bonding mechanisms in boards made from laccase-treated fibers of beech (Fagus sylvatica). Ind Crop Prod 20:181–189
Kharazipour A, Hüttermann A, Kühne G, Rong M (1993) Process for glueing wood chips and articles produced by this process. Eur Pat Appl EP0565109
Kharazipour A, Mai C, Hüttermann A (1998) Polyphenols for compounded materials. Polym Degrad Stabil 59:237–243
Qvintus-Leino P, Widsten P, Tuominen S, Laine J, Kunnas J (2003) Method of producing compressed layered structures such as fiberboard or similar wood-based product. Int Pat Appl WO03047826
Hüttermann A, Mai C, Kharazipour A (2001) Modification of lignin for the production of new compounded materials. Appl Microbiol Biotechnol 55:387–394
Tsapikouni TS, Missirlis YF (2008) Protein–material interactions: from micro-to-nano scale. Mater Sci Eng B 152:2–7
Nady N, Schroën K, Franssen MCR, van Lagen B, Murali S, Boom RM, Mohy Eldin MS, Zuilhof H (2011) Mild and highly flexible enzyme-catalyzed modification of poly(ethersulfone) membranes. ACS Appl Mater Interfaces 3:801–810
Nady N, Schroën K, Franssen MCR, Fokkink R, Mohy Eldin MS, Zuilhof H, Boom RM (2012) Enzyme-catalyzed modification of PES surfaces: reduction in adsorption of BSA, dextrin and tannin. J Colloid Interface Sci 378:191–200
Mikolasch A, Niedermeyer THJ, Lalk M, Witt S, Seefeldt S, Hammer E, Schauer F, Gesell M, Hessel s Julich WD, Lindequist U (2006) Novel penicillins synthesized by biotransformation using laccase from Trametes spec. Chem Pharm Bull 54(5):632–638
Mikolasch A, Niedermeyer THJ, Lalk M, Witt S, Seefeldt S, Hammer E, Schauer F, Salazar MG, Hessel S, Julich WD, Lindequist U (2007) Novel cephalosporins synthesized by amination of 2,5-dihydroxybenzoic acid derivatives using fungal laccases II. Chem Pharm Bull 55(3):412–416
Mikolasch A, Wurster M, Lalk M, Witt S, Seefeldt S, Hammer E, Shauer F, Julich WD, Lindequist U (2008) Novel b-lactam antibiotics synthesized by amination of catechols using fungal laccase. Chem Pharm Bull 56(7):902–907
Mikolasch A, Manda K, Schlüter R, Lalk M, Witt S, Seefeldt S, Hammer E, Schauer F, Hammer E, Jülich WD, Lindequist U (2012) Comparative analyses of laccase-catalyzed amination reactions for production of novel β-lactam antibiotics. Biotechnol Appl Bioc 59:295–306
Yang Z, Pattamana K, Molino BF, Haydar SN, Cao Y, Bois F, Maeng JH, Hemenway MS, Rich JO, Khmelnitsky YL, Friedrich TD, Peace D, Michels PC (2009) Novel Oxidation of cyclosporin A: preparation of cyclosporin methyl vinyl ketone (Cs-MVK). Synlett 18:2935–2938. doi:10.1055/s-0029-1218011
Fontana G, Baldelli E, Riva S, Danieli B (2009) Process for the preparation of bisindole alkaloid derivatives WO 2009153025 A1
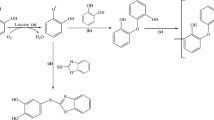
Sagui F, Chirivì C, Fontana G, Nicotra S, Passarella D, Riva S, Danieli B (2009) Laccase-catalyzed coupling of catharanthine and vindoline: an efficient approach to the bis indole alkaloid anhydrovinblastine. Tetrahedron 65:312–317
Herter S, Michalik D, Mikolasch A, Schmidt M, Wohlgemuth R, Bornscheuer, Schauer F (2013) Laccase-mediated synthesis of 2-methoxy-3-methyl-5-(alkylamino)-and 3-methyl-2,5-bis(alkylamino)-[1,4]-benzoquinones. J Mol Catal B Enzym 90:91–97
Wellington KW, Steenkamp P, Brady D (2010) Diamination by N-coupling using a commercial laccase. Bioorgan Med Chem 18(4):1406–1414
Wellington KW, Kolesnikova NI (2012) A laccase-catalysed one-pot synthesis of aminonaphthoquinones and their anticancer activity. Bioorgan Med Chem 20(14):4472–4481
Ravikumar G, Gimathi DG, Kalaiselvi M, Devaki K, Uma C (2013) Antioxidant property and anti-proliferative activity of recombinant laccase from Hypsizygus ulmarius against breast cancer cell line (MCF7). Int J Res Pharm Biomed Sci 4:1148–1152
Zhang GQ, Tian T, Liu YP, Wang HX, Chen QJ (2011) A laccase with anti-proliferative activity against tumor cells from a white root fungus Abortiporus biennis. Process Biochem 46:2336–2340
Hu DD, Zhang RY, Zhang GQ, Wang HX, Ng TB (2011) A laccase with antiproliferative activity against tumor cells from an edible mushroom, white common Agrocybe cylindracea. Phytomedicine 18:374–379
Othman AM, Elshafei AM, Hassan MM, Haroun AM, Elsayed MA, Farrag AA (2014) Purification, biochemical characterization and applications of Pleurotus ostreatus ARC 280 laccase. Br Microbiol Res J 4(12):1418–1439
Wong JH, Ng TB, Jiang Y, Liu F, Sze SC, Zhang KY (2010) Purification and characterization of a laccase with inhibitory activity toward HIV-1 reverse transcriptase and tumor cells from an edible mushroom (Pleurotus cornucopiae). Protein Peptide Lett 17(8):1040–1047
Wang HX, Ng TB (2004) Purification of a novel low-molecular-mass laccase with HIV-1 reverse transcriptase inhibitory activity from the mushroom Trichoderma gigantum. Biochem Bioph Res Co 315:450–454
Zhang GQ, Chen QJ, Wang HX, Ng TB (2013) A laccase with inhibitory activity against HIV-1 reverse transcriptase from mycorrhizal fungus Lepiota ventrisospora. J Mol Catal B Enzym 85–86:31–36
Sun J, Wang H, Ng TB (2011) Isolation of a laccase with HIV-1 reverse transcriptase inhibitory activity from fresh fruiting bodies of the Lentinus edodes (Shiitake mushroom). Indian J Biochem Bio 48:88–94
Sun J, Chen QJ, Cao QQ, Wu YY, Xu LJ, Zhu MJ, Ng TB, Wang HX, Zhang GQ (2012) A laccase with antiproliferative and HIV-I reverse transcriptase inhibitory activities from the mycorrhizal fungus Agaricus placomyces. J Biomed Biotechnol. doi:10.1155/2012/736472
Wu X, Huang C, Chen Q, Wang H, Zhang J (2014) A novel laccase with inhibitory activity towards HIV-I reverse transcriptase and antiproliferative effects on tumor cells from the fermentation broth of mushroom Pleurotus cornucopiae. Biomed Chromatogr 28(4):548–553
Zhao S, Rong CB, Kong C, Liu Y, Xu F, Miao DJ, Wang SX, Wang HX, Zhang GQ (2014) A novel laccase with potent antiproliferative and HIV-1 reverse transcriptase inhibitory activities from mycelia of mushroom Coprinus comatus. J Biomed Biotechnol. doi:10.1155/2014/417461
Xu L, Wang HX, Ng T (2012) A laccase with HIV-1 reverse transcriptase inhibitor activity from the broth of mycelial culture of the mushroom Lentinus tigrinus. J Biomed Biotechnol. doi:10.1155/2012/536725
Zou YJ, Wang HX, Ng TB, Huang CY, Zhang JX (2012) Purification and characterization of a novel laccase from the edible mushroom Hericium coralloides. J Microbiol 50(1):72–78
Harris ZL, Davis-Kaplan SR, Gitlin JD, Kaplan J (2004) A fungal multicopper oxidase restores iron homeostasis in aceruloplasminemia. Blood 103:4672–4673
Kulys J, Bratkovskaja I, Vidziunaite R (2005) Laccase-catalysed iodide oxidation in presence of methyl syringate. Biotechnol Bioeng 92(1):124–128
Xu F (1998) Method of producing iodine by use of a copper containing oxidase enzyme. US 5766896 A
Ihssen J, Schubert M, Thony-Meyer L, Richter M (2013) Laccase Catalyzed Synthesis of Iodonated Phenolic Compounds with Antifungal Activity. PLoS One 9(3):e89924. doi:10.1371/journal.pone.0089924
Christensen BE, Danielsen S, Oestergaard LH (2007) Methods and compositions for killing spores. WO 2006094975 A3
Eggert C (1997) Laccase-catalyzed formation of cinnabarinic acid is responsible for antibacterial activity of Pycnoporus cinnabarinus. Microbiol Res 152:315–318
Grover N, Borkar IV, Dinu CZ, Kane RS, Dordick JS (2012) Laccase- and chloroperoxidase-nanotube paint composites with bactericidal and sporicidal activity. Enzyme Microb Tech 50:271–279
Chen SC, Chen CH, Chern CL, Hsu LS, Huang YC, Chung KT, Chye SM (2006) p-Phenylenediamine induces p53-mediated apoptosis in Mardin-Darby canine kidney cells. Toxicol In Vitro 20:801–807
Huang YC, Hung WC, Kang WY, Chen WT, Chai CY (2007) p-Phenylenediamine induced DNA damage in SV-40 immortalized human uroepithelial cells and expression of mutant p53 and COX-2 proteins. Toxicol Lett 170:116–123
Araujo R, Fernandes M, Cavaco-Paulo A, Gomes A (2011) Biology of human hair: know your hair to control it. Adv Biochem Eng Biot 13:121–143
Takada K, Nakamura A, Matsuo N, Inoue A, Someya K, Shimogaki H (2003) Influence of oxidative and/or reductive treatment on human hair (I): analysis of hair-damage after oxidative and/or reductive treatment. J Oleo Sci 52:541–548
Fu J, Nyanhongo GS, Gubitz GM, Cavaco Paulo A, Kim S (2012) Enzymatic colouration with laccase and peroxidases: recent progress. Biocatal Biotransfor 30(1):125–140
Lavanya C, Dhankar R, Chhikara S, Sheoran S (2014) Degradation of toxic dyes: a review. Int J Curr Microbiol App Sci 3(6):189–199
Salame TM, Yarden O, Hadar Y (2010) Pleurotus ostreatus manganese-dependent peroxidase silencing impairs decolourization of Orange II. Microb Biotechnol 3:93–106
Jeon JR, Kim EJ, Murugesan K, Park HK, Kim YM, Kwon JH, Kim WG, Lee JY, Chang YS (2010) Laccase-catalysed polymeric dye synthesis from plant-derived phenols for potential application in hair dyeing: enzymatic colourations driven by homo- or hetero-polymer synthesis. Microb Biotechnol 3:324–335
Otsuka Saito K, Ikeda R, Endo K, Tsujino Y, Takagi M, Tamiya E (2012) Isolation of a novel alkaline-induced laccase from Flammulina velutipes and its application for hair coloring. J Biosci Bioeng 113(5):575–579
Chen CY, Huang YC, Wei CM, Meng M, Liu WH, Yang CH (2013) Properties of the newly isolated extracellular thermo-alkali-stable laccase from thermophilic actinomycetes, Thermobifida fusca and its application in dye intermediates oxidation. AMB Express 3:49
Fang Z, Zhou P, Chang F, Yin Q, Fang W, Yuan J, Zhang X, Xiao Y (2014) Structure-based rational design to enhance the solubility and thermostability of a bacterial laccase Lac15. PLoS One 9(7):e102423. doi:10.1371/journal.pone.0102423
Golz-Berner K, Walzel B, Zastrow L, Doucet O (2004) Cosmetic or dermatological preparation with skin-lightening proteins. WO2004017931
Kunamneni A, Plou FJ, Ballesteros A, Alcalde M (2008) Laccases and their applications: a patent review. Recent Pat Biotechnol 2(1):10–24
Nagai M, Kawata M, Watanabe H, Ogawa M, Saito K, Takesawa T, Kanda K, Sato T (2003) Important role of fungal intracellular laccase for melanin synthesis:purification and characterization of an intracellular laccase from Lentinula edodes fruit body. Microbiology 149:2455–2462
Madhavi V, Lele SS (2009) Laccase: properties and applications. BioResources 4:4
Takase T, Narise A, Sakurai K (2011) Deodorant composition. WO2011105042 A1
Brinch DS, Pederson PB (2002) Toxicological studies on laccase from Myceliophthora thermophila expressed in Aspergillus oryzae. Regul Toxicol Pharm 35(3):296–307
Mano N, Durand F (2013) Laccase of Podospora anserina and uses of same. WO2013175399 A1
Bhogal RK, Casey J, Ganguli S, Hunter KJ, Koek JH, Redfern SP (2013) Hair colouring composition. WO2013189966 A2
Doucet O, Golz-Berner K, Walzel B, Zastrow L (2004) Cosmetic or dermatological preparation with skin-lightening proteins. WO 2004017931:A1
Koike K (2002). Multiple agent type hair dye. JP2002255764
Lang G, Cotteret J (2002) Ready to use hair dye containing oxidation coloring agent, cationic polymers and enzyme; hydrogen peroxide free; homogeneous, intense; nondegrading or decolouring of keratinous fibers. US 6471730 B1
Lang G, Cotteret J (2004) Composition for the oxidation dyeing of keratinous fibres containing a laccase and dyeing method using this composition. US2004255401 A1
Nakajima M, Fujita H, Kikuchi Y, Kobayashi I (2002) Cosmetic mixture for the oxidation tinting of keratin fibres, containing in a support material suitable for tinting keratin fibres (a) at least one laccase-type enzyme; (b) at least one polymer thickener selected from polymers. US 20020043731 A1
Onuki T, Nogucji M, Mitamura J (2000) Oxidative hair dye com-position containing laccase. WO0037030
Pereira R, Burgaud H (2005) Producing tetraazapentamethine compounds comprises reacting an azine compound with an oxidizing agent, useful for dyeing keratinic fibers, e.g. hair. FR2863487 A1
PLoS G (2005) Oxidation dyeing method using N-acetyclysteine as a reducing agent and laccase as an oxidising agent. US 6840964 B1
PLoS G (2001) Composition for oxidative dyeing of keratinous fibres and dyeing process using same. EP1138318 A2
Pruche F, Saint LP, Bernards B (2000) Hydroxystilbene compounds of formula (I) used as components in oxidation dye systems especially for dyeing hair. EP1013260
Shichiri S, Morita K, Koike K (2003) Hair cosmetic. JP2003055175 A2
Riva S (2006) Laccases blue enzymes for green chemistry. Trends Biotechnol 24:219–226
Sorensen NH (2001) Method for dyeing dry hair. WO2001068042 A1
Tsuji K, Yoshino T, Asai Y (2002) Composition for bleaching melamine. JP2002012535 A2
Acknowledgments
This work was supported by grants from the European project “Optimized oxidoreductases for medium and large scale industrial biotransformations, INDOX” (KBBE-2013-7-613549) and from the Italian MIUR (Ministero dell’Università e della Ricerca Scientifica Progetti di Rilevante Interesse Nazionale) PRIN 2009STNWX3. Cinzia Pezzella is recipient of a fellowship funded by POR Campania FSE 2007/2013 “Campania Research in Experimental Medicine, Project CREME” (CUP B25B09000050007).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pezzella, C., Guarino, L. & Piscitelli, A. How to enjoy laccases. Cell. Mol. Life Sci. 72, 923–940 (2015). https://doi.org/10.1007/s00018-014-1823-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-014-1823-9