Abstract
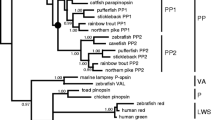
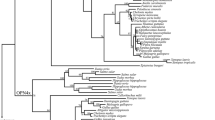
Melanopsin (OPN4) is an opsin photopigment that, in mammals, confers photosensitivity to retinal ganglion cells and regulates circadian entrainment and pupil constriction. In non-mammalian species, two forms of opn4 exist, and are classified into mammalian-like (m) and non-mammalian-like (x) clades. However, far less is understood of the function of this photopigment family. Here we identify in zebrafish five melanopsins (opn4m-1, opn4m-2, opn4m-3, opn4x-1 and opn4x-2), each encoding a full-length opsin G protein. All five genes are expressed in the adult retina in a largely non-overlapping pattern, as revealed by RNA in situ hybridisation and immunocytochemistry, with at least one melanopsin form present in all neuronal cell types, including cone photoreceptors. This raises the possibility that the teleost retina is globally light sensitive. Electrophysiological and spectrophotometric studies demonstrate that all five zebrafish melanopsins encode a functional photopigment with peak spectral sensitivities that range from 470 to 484 nm, with opn4m-1 and opn4m-3 displaying invertebrate-like bistability, where the retinal chromophore interchanges between cis- and trans-isomers in a light-dependent manner and remains within the opsin binding pocket. In contrast, opn4m-2, opn4x-1 and opn4x-2 are monostable and function more like classical vertebrate-like photopigments, where the chromophore is converted from 11-cis to all-trans retinal upon absorption of a photon, hydrolysed and exits from the binding pocket of the opsin. It is thought that all melanopsins exhibit an invertebrate-like bistability biochemistry. Our novel findings, however, reveal the presence of both invertebrate-like and vertebrate-like forms of melanopsin in the teleost retina, and indicate that photopigment bistability is not a universal property of the melanopsin family. The functional diversity of these teleost melanopsins, together with their widespread expression pattern within the retina, suggests that melanopsins confer global photosensitivity to the teleost retina and might allow for direct “fine-tuning” of retinal circuitry and physiology in the dynamic light environments found in aquatic habitats.







Similar content being viewed by others
References
Hattar S, Liao HW, Takao M, Berson DM, Yau KW (2002) Melanopsin-containing retinal ganglion cells: architecture, projections, and intrinsic photosensitivity. Science 295:1065–1070
Rollag MD, Berson DM, Provencio I (2003) Melanopsin, ganglion-cell photoreceptors, and mammalian photoentrainment. J Biol Rhythms 18:227–234
Hankins MW, Peirson SN, Foster RG (2008) Melanopsin: an exciting photopigment. Trends Neurosci 31:27–36
Bailes HJ, Lucas RJ (2010) Melanopsin and inner retinal photoreception. Cell Mol Life Sci 67:99–111
Muller LP, Do MT, Yau KW, He S, Baldridge WH (2010) Tracer coupling of intrinsically photosensitive retinal ganglion cells to amacrine cells in the mouse retina. J Comp Neurol 518:4813–4824
Gamlin PD, McDougal DH, Pokorny J, Smith VC, Yau KW, Dacey DM (2007) Human and macaque pupil responses driven by melanopsin-containing retinal ganglion cells. Vision Res 47:946–954
Hatori M, Panda S (2010) The emerging roles of melanopsin in behavioral adaptation to light. Trends Mol Med 16:435–446
Melyan Z, Tarttelin EE, Bellingham J, Lucas RJ, Hankins MW (2005) Addition of human melanopsin renders mammalian cells photoresponsive. Nature 433:741–745
Koyanagi M, Kubokawa K, Tsukamoto H, Shichida Y, Terakita A (2005) Cephalochordate melanopsin: evolutionary linkage between invertebrate visual cells and vertebrate photosensitive retinal ganglion cells. Curr Biol 15:1065–1069
Walker MT, Brown RL, Cronin TW, Robinson PR (2008) Photochemistry of retinal chromophore in mouse melanopsin. Proc Natl Acad Sci USA 105:8861–8865
Mure LS, Cornut PL, Rieux C, Drouyer E, Denis P, Gronfier C, Cooper HM (2009) Melanopsin bistability: a fly’s eye technology in the human retina. PLoS One 4:e5991
Pires SS, Hughes S, Turton M, Melyan Z, Peirson SN, Zheng L, Kosmaoglou M, Bellingham J, Cheetham ME, Lucas RJ, Foster RG, Hankins MW, Halford S (2009) Differential expression of two distinct functional isoforms of melanopsin (Opn4) in the mammalian retina. J Neurosci 29:12332–12342
Davies WL, Hankins MW, Foster RG (2010) Vertebrate ancient opsin and melanopsin: divergent irradiance detectors. Photochem Photobiol Sci 9:1444–1457
Torii M, Kojima D, Okano T, Nakamura A, Terakita A, Shichida Y, Wada A, Fukada Y (2007) Two isoforms of chicken melanopsins show blue light sensitivity. FEBS Lett 581:5327–5331
Bellingham J, Chaurasia SS, Melyan Z, Liu C, Cameron MA, Tarttelin EE, Iuvone PM, Hankins MW, Tosini G, Lucas RJ (2006) Evolution of melanopsin photoreceptors: discovery and characterization of a new melanopsin in nonmammalian vertebrates. PLoS Biol 4:e254
Drivenes O, Soviknes AM, Ebbesson LO, Fjose A, Seo HC, Helvik JV (2003) Isolation and characterization of two teleost melanopsin genes and their differential expression within the inner retina and brain. J Comp Neurol 456:84–93
Bailey MJ, Cassone VM (2005) Melanopsin expression in the chick retina and pineal gland. Brain Res Mol Brain Res 134:345–348
Chaurasia SS, Rollag MD, Jiang G, Hayes WP, Haque R, Natesan A, Zatz M, Tosini G, Liu C, Korf HW, Iuvone PM, Provencio I (2005) Molecular cloning, localization and circadian expression of chicken melanopsin (Opn4): differential regulation of expression in pineal and retinal cell types. J Neurochem 92:158–170
Jenkins A, Munoz M, Tarttelin EE, Bellingham J, Foster RG, Hankins MW (2003) VA opsin, melanopsin, and an inherent light response within retinal interneurons. Curr Biol 13:1269–1278
Provencio I, Jiang G, De Grip WJ, Hayes WP, Rollag MD (1998) Melanopsin: an opsin in melanophores, brain, and eye. Proc Natl Acad Sci USA 95:340–345
Bellingham J, Whitmore D, Philp AR, Wells DJ, Foster RG (2002) Zebrafish melanopsin: isolation, tissue localisation and phylogenetic position. Brain Res Mol Brain Res 107:128–136
Cheng N, Tsunenari T, Yau KW (2009) Intrinsic light response of retinal horizontal cells of teleosts. Nature 460:899–903
Grone BP, Sheng Z, Chen CC, Fernald RD (2007) Localization and diurnal expression of melanopsin, vertebrate ancient opsin, and pituitary adenylate cyclase-activating peptide mRNA in a teleost retina. J Biol Rhythms 22:558–561
Tomonari S, Takagi A, Noji S, Ohuchi H (2007) Expression pattern of the melanopsin-like (cOpn4m) and VA opsin-like genes in the developing chicken retina and neural tissues. Gene Expr Patterns 7:746–753
Chomczynski P, Sacchi N (1987) Single-step method of RNA isolation by acid guanidinium thiocyanate–phenol–chloroform extraction. Anal Biochem 162:156–159
Higgins DG, Thompson JD, Gibson TJ (1996) Using CLUSTAL for multiple sequence alignments. Methods Enzymol 266:383–402
Tamura K, Dudley J, Nei M, Kumar S (2007) MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0. Mol Biol Evol 24:1596–1599
Kimura M (1980) A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16:111–120
Whitmore D, Foulkes NS, Sassone-Corsi P (2000) Light acts directly on organs and cells in culture to set the vertebrate circadian clock. Nature 404:87–91
Vandesompele J, De Preter K, Pattyn F, Poppe B, Van Roy N, De Paepe A, Speleman F (2002) Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol 3:RESEARCH0034
Carleton KL, Kocher TD (2001) Cone opsin genes of African Cichlid fishes: tuning spectral sensitivity by differential gene expression. Mol Biol Evol 18:1540–1550
Lo K, Smale ST (1996) Generality of a functional initiator consensus sequence. Gene 182:13–22
Cross SH, Bird AP (1995) CpG islands and genes. Curr Opin Genet Dev 5:309–314
Phillips JE, Corces VG (2009) CTCF: master weaver of the genome. Cell 137:1194–1211
Amouyal M (2010) Gene insulation. Part II: natural strategies in vertebrates. Biochem Cell Biol 88:885–898
Ishihara K, Sasaki H (2002) An evolutionarily conserved putative insulator element near the 3′ boundary of the imprinted Igf2/H19 domain. Hum Mol Genet 11:1627–1636
Juo ZS, Chiu TK, Leiberman PM, Baikalov I, Berk AJ, Dickerson RE (1996) How proteins recognize the TATA box. J Mol Biol 261:239–254
Baumann M, Pontiller J, Ernst W (2010) Structure and basal transcription complex of RNA polymerase II core promoters in the mammalian genome: an overview. Mol Biotechnol 45:241–247
Juven-Gershon T, Kadonaga JT (2010) Regulation of gene expression via the core promoter and the basal transcriptional machinery. Dev Biol 339:225–229
Benoist C, O’Hare K, Breathnach R, Chambon P (1980) The ovalbumin gene-sequence of putative control regions. Nucleic Acids Res 8:127–142
Burke TW, Kadonaga JT (1997) The downstream core promoter element, DPE, is conserved from Drosophila to humans and is recognized by TAFII60 of Drosophila. Genes Dev 11:3020–3031
Lim CY, Santoso B, Boulay T, Dong E, Ohler U, Kadonaga JT (2004) The MTE, a new core promoter element for transcription by RNA polymerase II. Genes Dev 18:1606–1617
Lagrange T, Kapanidis AN, Tang H, Reinberg D, Ebright RH (1998) New core promoter element in RNA polymerase II-dependent transcription: sequence-specific DNA binding by transcription factor IIB. Genes Dev 12:34–44
Briggs MR, Kadonaga JT, Bell SP, Tjian R (1986) Purification and biochemical characterization of the promoter-specific transcription factor, Sp1. Science 234:47–52
Lee W, Mitchell P, Tjian R (1987) Purified transcription factor AP-1 interacts with TPA-inducible enhancer elements. Cell 49:741–752
Swaroop A, Kim D, Forrest D (2010) Transcriptional regulation of photoreceptor development and homeostasis in the mammalian retina. Nat Rev Neurosci 11:563–576
Cvekl A, Wang WL (2009) Retinoic acid signaling in mammalian eye development. Exp Eye Res 89:280–291
Akimoto M (2005) Transcriptional factors involved in photoreceptor differentiation. Semin Ophthalmol 20:25–30
Smyth VA, Di Lorenzo D, Kennedy BN (2008) A novel, evolutionarily conserved enhancer of cone photoreceptor-specific expression. J Biol Chem 283:10881–10891
Rehemtulla A, Warwar R, Kumar R, Ji X, Zack DJ, Swaroop A (1996) The basic motif-leucine zipper transcription factor Nrl can positively regulate rhodopsin gene expression. Proc Natl Acad Sci USA 93:191–195
Kataoka K, Noda M, Nishizawa M (1994) Maf nuclear oncoprotein recognizes sequences related to an AP-1 site and forms heterodimers with both Fos and Jun. Mol Cell Biol 14:700–712
Kerppola TK, Curran T (1994) Maf and Nrl can bind to AP-1 sites and form heterodimers with Fos and Jun. Oncogene 9:675–684
Chen J, Rattner A, Nathans J (2005) The rod photoreceptor-specific nuclear receptor Nr2e3 represses transcription of multiple cone-specific genes. J Neurosci 25:118–129
Jetten AM, Kurebayashi S, Ueda E (2001) The ROR nuclear orphan receptor subfamily: critical regulators of multiple biological processes. Prog Nucleic Acid Res Mol Biol 69:205–247
Ueda HR, Hayashi S, Chen W, Sano M, Machida M, Shigeyoshi Y, Iino M, Hashimoto S (2005) System-level identification of transcriptional circuits underlying mammalian circadian clocks. Nat Genet 37:187–192
Furukawa T, Morrow EM, Cepko CL (1997) Crx, a novel otx-like homeobox gene, shows photoreceptor-specific expression and regulates photoreceptor differentiation. Cell 91:531–541
Kelley CG, Lavorgna G, Clark ME, Boncinelli E, Mellon PL (2000) The Otx2 homeoprotein regulates expression from the gonadotropin-releasing hormone proximal promoter. Mol Endocrinol 14:1246–1256
Kimura A, Singh D, Wawrousek EF, Kikuchi M, Nakamura M, Shinohara T (2000) Both PCE-1/RX and OTX/CRX interactions are necessary for photoreceptor-specific gene expression. J Biol Chem 275:1152–1160
Chen S, Zack DJ (1996) Ret 4, a positive acting rhodopsin regulatory element identified using a bovine retina in vitro transcription system. J Biol Chem 271:28549–28557
Moses K, Rubin GM (1991) Glass encodes a site-specific DNA-binding protein that is regulated in response to positional signals in the developing Drosophila eye. Genes Dev 5:583–593
Dorval KM, Bobechko BP, Fujieda H, Chen S, Zack DJ, Bremner R (2006) CHX10 targets a subset of photoreceptor genes. J Biol Chem 281:744–751
Zhang J, Dong X, Fujimoto Y, Okamura H (2004) Molecular signals of mammalian circadian clock. Kobe J Med Sci 50:101–109
Govardovskii VI, Fyhrquist N, Reuter T, Kuzmin DG, Donner K (2000) In search of the visual pigment template. Vis Neurosci 17:509–528
Shiraki T, Kojima D, Fukada Y (2010) Light-induced body color change in developing zebrafish. Photochem Photobiol Sci 9:1498–1504
Fitzgibbon J, Hope A, Slobodyanyuk SJ, Bellingham J, Bowmaker JK, Hunt DM (1995) The rhodopsin-encoding gene of bony fish lacks introns. Gene 164:273–277
Bellingham J, Tarttelin EE, Foster RG, Wells DJ (2003) Structure and evolution of the teleost extraretinal rod-like opsin (errlo) and ocular rod opsin (rho) genes: is teleost rho a retrogene? J Exp Zool B Mol Dev Evol 297:1–10
Allison WT, Barthel LK, Skebo KM, Takechi M, Kawamura S, Raymond PA (2010) Ontogeny of cone photoreceptor mosaics in zebrafish. J Comp Neurol 518:4182–4195
Farhat FP, Martins CB, De Lima LH, Isoldi MC, Castrucci AM (2009) Melanopsin and clock genes: regulation by light and endothelin in the zebrafish ZEM-2S cell line. Chronobiol Int 26:1090–1119
Qiu X, Kumbalasiri T, Carlson SM, Wong KY, Krishna V, Provencio I, Berson DM (2005) Induction of photosensitivity by heterologous expression of melanopsin. Nature 433:745–749
Panda S, Nayak SK, Campo B, Walker JR, Hogenesch JB, Jegla T (2005) Illumination of the melanopsin signaling pathway. Science 307:600–604
Tsukamoto H, Terakita A, Shichida Y (2005) A rhodopsin exhibiting binding ability to agonist all-trans-retinal. Proc Natl Acad Sci USA 102:6303–6308
Hillman P, Hochstein S, Minke B (1983) Transduction in invertebrate photoreceptors: role of pigment bistability. Physiol Rev 63:668–772
Wald G (1968) The molecular basis of visual excitation. Nature 219:800–807
Farrens DL, Khorana HG (1995) Structure and function in rhodopsin. Measurement of the rate of metarhodopsin II decay by fluorescence spectroscopy. J Biol Chem 270:5073–5076
Lamb TD (2009) Evolution of vertebrate retinal photoreception. Philos Trans R Soc Lond B Biol Sci 364:2911–2924
Arendt D, Hausen H, Purschke G (2009) The ‘division of labour’ model of eye evolution. Philos Trans R Soc Lond B Biol Sci 364:2809–2817
Lamb TD, Pugh EN Jr (2004) Dark adaptation and the retinoid cycle of vision. Prog Retin Eye Res 23:307–380
Terakita A, Tsukamoto H, Koyanagi M, Sugahara M, Yamashita T, Shichida Y (2008) Expression and comparative characterization of Gq-coupled invertebrate visual pigments and melanopsin. J Neurochem 105:883–890
Kirsch M, Wagner HJ, Douglas RH (1989) Rods trigger light adaptive retinomotor movements in all spectral cone types of a teleost fish. Vision Res 29:389–396
Brockerhoff SE, Rieke F, Matthews HR, Taylor MR, Kennedy B, Ankoudinova I, Niemi GA, Tucker CL, Xiao M, Cilluffo MC, Fain GL, Hurley JB (2003) Light stimulates a transducin-independent increase of cytoplasmic Ca2+ and suppression of current in cones from the zebrafish mutant nof. J Neurosci 23:470–480
Sekaran S, Foster RG, Lucas RJ, Hankins MW (2003) Calcium imaging reveals a network of intrinsically light-sensitive inner-retinal neurons. Curr Biol 13:1290–1298
Acknowledgments
We thank Dr. Rosalie Crouch and Dr. Jill Cowing for the generous provision of 11-cis retinal and 1D4 antibody; Dr. Stephen Price and Dr. Sofia Godinho for help with RNA in situ hybridisation experiments; Dr. Kara Cerveny for microscopy and photography advice; Dr. Stuart Peirson for his assistance on UV-vis template fitting and useful discussions; and University College London Fish Facility for adult zebrafish. We thank Prof. Shaun Collin for his advice on photoreceptor morphology. This work was supported by a grant awarded by the UK Biotechnology and Biological Research Council (BBSRC) to MWH.
Conflict of interest
The authors declare no competing financial interests.
Author information
Authors and Affiliations
Corresponding author
Additional information
W. I. L. Davies and L. Zheng contributed equally to this research.
Supplementary material is linked to the online version of the paper at http://www.springer.com/birkhauser/biosciences/journal/18.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Davies, W.I.L., Zheng, L., Hughes, S. et al. Functional diversity of melanopsins and their global expression in the teleost retina. Cell. Mol. Life Sci. 68, 4115–4132 (2011). https://doi.org/10.1007/s00018-011-0785-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-011-0785-4