Abstract
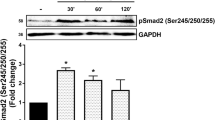
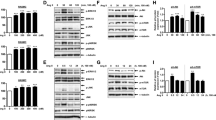
Transforming growth factor (TGF)-β treatment of human vascular smooth-muscle cells increases the expression of biglycan and causes marked elongation of its glycosaminoglycan (GAG) chains. We investigated the role of MAP kinases and Smad transcription factors in this response. TGF-β-stimulated phosphorylation of p38, ERK, and JNK as well as Smad2 at both its carboxy terminal (phospho-Smad2C) and in the linker region (phospho-Smad2L). Pharmacological inhibition of ERK and p38 blocked TGF-β-mediated GAG elongation and expression of biglycan whereas inhibition of JNK had no effect. Inhibition of ERK and p38 but not JNK attenuated the effect of TGF-β to increase phospho-Smad2L. High levels of phospho-Smad2L were detected in a nuclear fraction of TGF-β treated cells. Thus, MAP kinase signaling through ERK and p38 and via phosphorylation of the linker region of Smad2 mediates the effects of TGF-β on biglycan synthesis in vascular smooth-muscle cells.






Similar content being viewed by others
References
Wight TN (1989) Cell biology of arterial proteoglycans. Arteriosclerosis 9:1–20
Hascall VC, Heinegard DK, Wight TN (1991) Proteoglycans: metabolism and pathology. In: Hay ED (ed) Cell biology of extracellular matrix. Plenum Press, New York, pp 149–175
Wight T (1996) The vascular extracellular matrix. In: Fuster V, Ross R, Topol EJ (eds) Atherosclerosis and coronary artery disease. Lippincott-Raven, Philadelphia, pp 421–440
Schonherr E, Jarvelainen HT, Kinsella MG, Sandell LJ, Wight TN (1993) Platelet-derived growth factor and transforming growth factor-beta 1 differentially affect the synthesis of biglycan and decorin by monkey arterial smooth muscle cells. Arterioscler Thromb 13:1026–1036
Dadlani H, Ballinger ML, Osman N, Getachew R, Little PJ (2008) Smad and p38 MAP kinase-mediated signaling of proteoglycan synthesis in vascular smooth muscle. J Biol Chem 283:7844–7852
Ivey ME, Little PJ (2008) Thrombin regulates vascular smooth muscle cell proteoglycan synthesis via PAR-1 and multiple downstream signalling pathways. Thromb Res 123:288–297
Ballinger ML, Ivey ME, Osman N, Thomas WG, Little PJ (2009) Endothelin-1 activates ETA receptors on human vascular smooth muscle cells to yield proteoglycans with increased binding to LDL. Atherosclerosis 205:451–457
Yang SN, Burch ML, Getachew R, Ballinger ML, Osman N, Little PJ (2009) Growth factor-mediated hyper-elongation of glycosaminoglycan chains on biglycan requires transcription and translation. Arch Physiol Biochem 115:147–154
Camejo G (1982) The interaction of lipids and lipoproteins with the intercellular matrix of arterial tissue: its possible role in atherogenesis. Adv Lipid Res 19:1–53
Little PJ, Tannock L, Olin KL, Chait A, Wight TN (2002) Proteoglycans synthesized by arterial smooth muscle cells in the presence of transforming growth factor-beta1 exhibit increased binding to LDLs. Arterioscler Thromb Vasc Biol 22:55–60
Ballinger ML, Nigro J, Frontanilla KV, Dart AM, Little PJ (2004) Regulation of glycosaminoglycan structure and atherogenesis. Cell Mol Life Sci 61:1296–1306
Skalen K, Gustafsson M, Rydberg EK, Hulten LM, Wiklund O, Innerarity TL, Boren J (2002) Subendothelial retention of atherogenic lipoproteins in early atherosclerosis. Nature 417:750–754
Ballinger ML, Osman N, Hashimura K, de Hann J, Jandeleit-Dahm K, Allen TJ, Tannock LR, Rutledge JC, Little PJ (2009) Imatinib inhibits vascular smooth muscle proteoglycan synthesis and reduces LDL binding in vitro and aortic lipid deposition in vivo. J Cell Mol Med. doi:10.1111/j.1582-4934.2009.00902.x)
Nakashima Y, Fujii H, Sumiyoshi S, Wight TN, Sueishi K (2007) Early human atherosclerosis: accumulation of lipid and proteoglycans in intimal thickenings followed by macrophage infiltration. Arterioscler Thromb Vasc Biol 27:1159–1165
Little PJ, Osman N, O’Brien KD (2008) Hyperelongated biglycan: the surreptitious initiator of atherosclerosis. Curr Opin Lipidol 19:448–454
Little PJ, Ballinger ML, Osman N (2007) Vascular wall proteoglycan synthesis and structure as a target for the prevention of atherosclerosis. Vasc Health Risk Manag 3:1–8
Bobik A, Agrotis A, Kanellakis P, Dilley R, Krushinsky A, Smirnov V, Tararak E, Condron M, Kostolias G (1999) Distinct patterns of transforming growth factor-beta isoform and receptor expression in human atherosclerotic lesions. Colocalization implicates TGF-beta in fibrofatty lesion development. Circulation 99:2883–2891
Derynck R, Zhang YE (2003) Smad-dependent and Smad-independent pathways in TGF-beta family signalling. Nature 425:577–584
Massague J (1998) TGF-beta signal transduction. Annu Rev Biochem 67:753–791
Kretzschmar M, Doody J, Timokhina I, Massague J (1999) A mechanism of repression of TGFbeta/Smad signaling by oncogenic Ras. Genes Dev 13:804–816
Moustakas A, Heldin CH (2005) Non-Smad TGF-beta signals. J Cell Sci 118:3573–3584
Ungefroren H, Lenschow W, Chen WB, Faendrich F, Kalthoff H (2003) Regulation of biglycan gene expression by transforming growth factor-beta requires MKK6-p38 mitogen-activated protein Kinase signaling downstream of Smad signaling. J Biol Chem 278:11041–11049
Ungefroren H, Groth S, Ruhnke M, Kalthoff H, Fandrich F (2005) Transforming growth factor-beta (TGF-beta) type I receptor/ALK5-dependent activation of the GADD45beta gene mediates the induction of biglycan expression by TGF-beta. J Biol Chem 280:2644–2652
Neylon CB, Little PJ, Cragoe EJ Jr, Bobik A (1990) Intracellular pH in human arterial smooth muscle. Regulation by Na+/H+ exchange and a novel 5-(N-ethyl-N-isopropyl)amiloride-sensitive Na(+)- and HCO3(−)-dependent mechanism. Circ Res 67:814–825
Chang Y, Yanagishita M, Hascall VC, Wight TN (1983) Proteoglycans synthesized by smooth muscle cells derived from monkey (Macaca nemestrina) aorta. J Biol Chem 258:5679–5688
Nigro J, Dilley RJ, Little PJ (2002) Differential effects of gemfibrozil on migration, proliferation and proteoglycan production in human vascular smooth muscle cells. Atherosclerosis 162:119–129
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Inman GJ, Nicolas FJ, Callahan JF, Harling JD, Gaster LM, Reith AD, Laping NJ, Hill CS (2002) SB-431542 is a potent and specific inhibitor of transforming growth factor-beta superfamily type I activin receptor-like kinase (ALK) receptors ALK4, ALK5, and ALK7. Mol Pharmacol 62:65–74
Jiang J, Song CP (2008) MEK1/2 and p38-like MAP kinase successively mediate H(2)O(2) signaling in Vicia guard cell. Plant Signal Behav 3:996–998
Zhang JY, Jiang H, Gao W, Wu J, Peng K, Shi YF, Zhang XJ (2008) The JNK/AP1/ATF2 pathway is involved in H2O2-induced acetylcholinesterase expression during apoptosis. Cell Mol Life Sci 65:1435–1445
Laping NJ, Grygielko E, Mathur A, Butter S, Bomberger J, Tweed C, Martin W, Fornwald J, Lehr R, Harling J, Gaster L, Callahan JF, Olson BA (2002) Inhibition of transforming growth factor (TGF)-beta1-induced extracellular matrix with a novel inhibitor of the TGF-beta type I receptor kinase activity: SB-431542. Mol Pharmacol 62:58–64
Li F, Zeng B, Chai Y, Cai P, Fan C, Cheng T (2009) The linker region of Smad2 mediates TGF-beta-dependent ERK2-induced collagen synthesis. Biochem Biophys Res Commun 386:289–293
Mori S, Matsuzaki K, Yoshida K, Furukawa F, Tahashi Y, Yamagata H, Sekimoto G, Seki T, Matsui H, Nishizawa M, Fujisawa J, Okazaki K (2004) TGF-beta and HGF transmit the signals through JNK-dependent Smad2/3 phosphorylation at the linker regions. Oncogene 23:7416–7429
Agrotis A, Kalinina N, Bobik A (2005) Transforming growth factor-beta, cell signaling and cardiovascular disorders. Curr Vasc Pharmacol 3:55–61
Mulder KM, Morris SL (1992) Activation of p21ras by transforming growth factor beta in epithelial cells. J Biol Chem 267:5029–5031
Yamaguchi K, Shirakabe K, Shibuya H, Irie K, Oishi I, Ueno N, Taniguchi T, Nishida E, Matsumoto K (1995) Identification of a member of the MAPKKK family as a potential mediator of TGF-beta signal transduction. Science 270:2008–2011
Massague J (2003) Integration of Smad and MAPK pathways: a link and a linker revisited. Genes Dev 17:2993–2997
Verrecchia F, Tacheau C, Wagner EF, Mauviel A (2003) A central role for the JNK pathway in mediating the antagonistic activity of pro-inflammatory cytokines against transforming growth factor-beta-driven SMAD3/4-specific gene expression. J Biol Chem 278:1585–1593
Victor XV, Nguyen TK, Ethirajan M, Tran VM, Nguyen KV, Kuberan B (2009) Investigating the elusive mechanism of glycosaminoglycan biosynthesis. J Biol Chem 284:25842–25853
Gotoh M, Yada T, Sato T, Akashima T, Iwasaki H, Mochizuki H, Inaba N, Togayachi A, Kudo T, Watanabe H, Kimata K, Narimatsu H (2002) Molecular cloning and characterization of a novel chondroitin sulfate glucuronyltransferase that transfers glucuronic acid to N-acetylgalactosamine. J Biol Chem 277:38179–38188
Little PJ, Ballinger ML, Burch ML, Osman N (2008) Biosynthesis of natural and hyperelongated chondroitin sulfate glycosaminoglycans: new insights into an elusive process. Open Biochem J 2:135–142
Tabas I, Williams KJ, Boren J (2007) Subendothelial lipoprotein retention as the initiating process in atherosclerosis: update and therapeutic implications. Circulation 116:1832–1844
Finn AV, Kramer MC, Vorpahl M, Kolodgie FD, Virmani R (2009) Pharmacotherapy of coronary atherosclerosis. Expert Opin Pharmacother 10:1587–1603
Acknowledgments
This work was supported by research grants from the National Health and Medical Research Council of Australia Project Grants (#268928 and #472611) and Development Grant #418934) (PJL), project grant from the Diabetes Australia Research Trust (NO) and a Grant-in-Aid from the National Heart Foundation of Australia (PJL). The PhD Program of MLB generously received support through a National Heart Foundation of Australia post-graduate scholarship and a post-graduate supervisor award from GlaxoSmithKline Australia.
Author information
Authors and Affiliations
Corresponding author
Additional information
M. L. Burch and S. N. Y. Yang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Burch, M.L., Yang, S.N.Y., Ballinger, M.L. et al. TGF-β stimulates biglycan synthesis via p38 and ERK phosphorylation of the linker region of Smad2. Cell. Mol. Life Sci. 67, 2077–2090 (2010). https://doi.org/10.1007/s00018-010-0315-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-010-0315-9