Abstract.
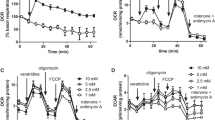
The role of some serine/threonine kinases in the regulation of mitochondrial physiology is now well established, but little is known about mitochondrial tyrosine kinases. We showed that tyrosine phosphorylation of rat brain mitochondrial proteins was increased by in vitro addition of ATP and H2O2, and also during in situ ATP production at state 3, and maximal reactive oxygen species production. The Src kinase inhibitor PP2 decreased tyrosine phosphorylation and respiratory rates at state 3. We found that the 39-kDa subunit of complex I was tyrosine phosphorylated, and we identified putative tyrosine-phosphorylated subunits for the other complexes. We also have strong evidence that the FoF1-ATP synthase α chain is probably tyrosine-phosphorylated, but demonstrated that the β chain is not. The tyrosine phosphatase PTP 1B was found in brain but not in muscle, heart or liver mitochondria. Our results suggest that tyrosine kinases and phosphatases are involved in the regulation of oxidative phosphorylation.
Similar content being viewed by others
Author information
Authors and Affiliations
Corresponding author
Additional information
Received 7 January 2005; received after revision 19 April 2005; accepted 22 April 2005
Rights and permissions
About this article
Cite this article
Augereau, O., Claverol, S., Boudes, N. et al. Identification of tyrosine-phosphorylated proteins of the mitochondrial oxidative phosphorylation machinery. CMLS, Cell. Mol. Life Sci. 62, 1478–1488 (2005). https://doi.org/10.1007/s00018-005-5005-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-005-5005-7