Abstract
Objective
This study aimed to investigate the effects of FK506 on experimental sepsis immunopathology. It investigated the effect of FK506 on leukocyte recruitment to the site of infection, systemic cytokine production, and organ injury in mice with sepsis.
Methods
Using a murine cecal ligation and puncture (CLP) peritonitis model, the experiments were performed with wild-type (WT) mice and mice deficient in the gene Nfat1 (Nfat1−/−) in the C57BL/6 background. Animals were treated with 2.0 mg/kg of FK506, subcutaneously, 1 h before the sepsis model, twice a day (12 h/12 h). The number of bacteria colony forming units (CFU) was manually counted. The number of neutrophils in the lungs was estimated by the myeloperoxidase (MPO) assay. The expression of CXCR2 in neutrophils was determined using flow cytometry analysis. The expression of inflammatory cytokines in macrophage was determined using ELISA. The direct effect of FK506 on CXCR2 internalization was evaluated using HEK-293T cells after CXCL2 stimulation by the BRET method.
Results
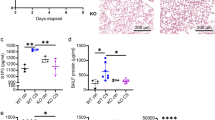
FK506 treatment potentiated the failure of neutrophil migration into the peritoneal cavity, resulting in bacteremia and an exacerbated systemic inflammatory response, which led to higher organ damage and mortality rates. Failed neutrophil migration was associated with elevated CXCL2 chemokine plasma levels and lower expression of the CXCR2 receptor on circulating neutrophils compared with non-treated CLP-induced septic mice. FK506 did not directly affect CXCL2-induced CXCR2 internalization by transfected HEK-293 cells or mice neutrophils, despite increasing CXCL2 release by LPS-treated macrophages. Finally, the CLP-induced response of Nfat1−/− mice was similar to those observed in the Nfat1+/+ genotype, suggesting that the FK506 effect is not dependent on the NFAT1 pathway.
Conclusion
Our data indicate that the increased susceptibility to infection of FK506-treated mice is associated with failed neutrophil migration due to the reduced membrane availability of CXCR2 receptors in response to exacerbated levels of circulating CXCL2.




Similar content being viewed by others
References
Singer M, Deutschman CS, Seymour C, Shankar-Hari M, Annane D, Bauer M, et al. The third international consensus definitions for sepsis and septic shock (sepsis-3). J Am Med Assoc. 2016;315:801–10.
Florescu DF, Sandkovsky U, Kalil AC. Sepsis and challenging infections in the immunosuppressed patient in the intensive care unit. Infect Dis Clin North Am. 2017;31:415–34.
Kalil AC, Sandkovsky U, Florescu DF. Severe infections in critically ill solid organ transplant recipients. Clin Microbiol Infect. 2018;24:1257–63. https://doi.org/10.1016/j.cmi.2018.04.022.
Kalil AC, Dakroub AGFH. HIV and solid organ transplantation. Hopkins HIV Rep. 2003;15:5–8.
Syu SH, Lin YW, Lin KH, Lee LM, Hsiao CH, Wen YC. Risk factors for complications and graft failure in kidney transplant patients with sepsis. Bosn J Basic Med Sci. 2019;19:304–11.
Creamer TP. Calcineurin. 2020;4:1–12
Clipstone NA, Fiorentino DF, Crabtree GR. Molecular analysis of the interaction of calcineurin with drug- immunophilin complexes. J Biol Chem [Internet]. © 1994 ASBMB. Currently published by Elsevier Inc; originally published by American Society for Biochemistry and Molecular Biology. 1994;269:26431–7. Available from: https://doi.org/10.1016/S0021-9258(18)47212-2
Shaw KTY, Ho AM, Raghavan A, Kim J, Jain J, Park J, et al. Immunosuppressive drugs prevent a rapid dephosphorylation of transcription factor NFAT1 in stimulated immune cells. Proc Natl Acad Sci USA. 1995;92:11205–9.
Kiani A, Rao A, Aramburu J. Manipulating immune responses with immunosuppressive agents that target NFAT. Immunity. 2000;12:359–72.
Healy JI, Dolmetsch RE, Timmerman LA, Cyster JG, Thomas ML, Crabtree GR, et al. Different nuclear signals are activated by the B cell receptor during positive versus negative signaling. Immunity. 1997;6:419–28.
Hutchinson LE, McCloskey MA. FcεRI-mediated induction of nuclear factor of activated T-cells. J Biol Chem. 1995;270:16333–8.
Aramburu J, Azzoni L, Rao A, Perussia B. Activation and expression of the nuclear factors of activated T cells, NFATp and NFATc, in human natural killer cells: regulation upon CD16 ligand binding. J Exp Med. 1995;182:801–10.
Liu Z, Dronadula N, Rao GN. A novel role for nuclear factor of activated T cells in receptor tyrosine kinase and G protein-coupled receptor agonist-induced vascular smooth muscle cell motility. J Biol Chem. 2004;279:41218–26.
Ichida M, Finkel T. Ras regulates NFAT3 activity in cardiac myocytes. J Biol Chem. 2001;276:3524–30.
Goodridge HS, Simmons RM, Underhill DM. Dectin-1 stimulation by Candida albicans yeast or zymosan triggers NFAT activation in macrophages and dendritic cells. J Immunol. 2007;178:3107–15.
Calderon-Sanchez E, Rodriguez-Moyano M, Smani T. Immunophilins and cardiovascular complications. Curr Med Chem. 2011;18:5408–13.
Nauseef WM, Borregaard N. Neutrophils at work. Nat Immunol. 2014;15:602–11.
Sônego F, Castanheira FVeS, Ferreira RG, Kanashiro A, Leite CAVG, Nascimento DC, et al. Paradoxical roles of the neutrophil in sepsis: Protective and deleterious. Front Immunol. 2016;7:1–7.
Farias Benjamim C, Santana Silva J, Bruno Fortes Z, Aparecida Oliveira M, Henrique Ferreira S, Queiroz CF. Inhibition of leukocyte rolling by nitric oxide during sepsis leads to reduced migration of active microbicidal neutrophils. Infect Immun. 2002;70:3602–10.
Torres-Dueñas D, Benjamim CF, Ferreira SH, Cunha FQ. Failure of neutrophil migration to infectious focus and cardiovascular changes on sepsis in rats: effects of the inhibition of nitric oxide production, removal of infectious focus, and antimicrobial treatment. Shock. 2006;25:267–76.
Freitas A, Alves-Filho JC, Trevelin SC, Spiller F, Suavinha MM, Nascimento DC, et al. Divergent role of heme oxygenase inhibition in the pathogenesis of sepsis. Shock. 2011;35:550–9.
Rios-Santos F, Alves-Filho JC, Souto FO, Spiller F, Freitas A, Lotufo CMC, et al. Down-regulation of CXCR2 on neutrophils in severe sepsis is mediated by inducible nitric oxide synthase-derived nitric oxide. Am J Respir Crit Care Med. 2007;175:490–7.
Alves-Filho JC, Freitas A, Souto FO, Spiller F, Paula-Neto H, Silva JS, et al. Regulation of chemokine receptor by Toll-like receptor 2 is critical to neutrophil migration and resistance to polymicrobial sepsis. Proc Natl Acad Sci USA. 2009;106:4018–23.
Arraes SMA, Freitas MS, Da Silva SV, De Paula Neto HA, Alves-Filho JC, Martins MA, et al. Impaired neutrophil chemotaxis in sepsis associates with GRK expression and inhibition of actin assembly and tyrosine phosphorylation. Blood. 2006;108:2906–13.
Kurowska-Stolarska M, Stolarski B, Kewin P, Murphy G, Corrigan CJ, Ying S, et al. IL-33 amplifies the polarization of alternatively activated macrophages that contribute to airway inflammation. J Immunol. 2009;183:6469–77.
Souto FO, Alves-Filho JC, Turato WM, Auxiliadora-Martins M, Basile-Filho A, Cunha FQ. Essential role of CCR2 in neutrophil tissue infiltration and multiple organ dysfunction in sepsis. Am J Respir Crit Care Med. 2011;183:234–42.
Namkung Y, Le Gouill C, Lukashova V, Kobayashi H, Hogue M, Khoury E, et al. Monitoring G protein-coupled receptor and β-arrestin trafficking in live cells using enhanced bystander BRET. Nat Commun. 2016;7:1–12.
Herbst S, Shah A, Carby M, Chusney G, Kikkeri N, Dorling A, et al. A new and clinically relevant murine model of solid-organ transplant aspergillosis. DMM Dis Model Mech. 2013;6:643–51.
Parreiras-E-Silva LT, Vargas-Pinilla P, Duarte DA, Longo D, Espinoza Pardo GV, Finkler AD, et al. Functional New World monkey oxytocin forms elicit an altered signaling profile and promotes parental care in rats. Proc Natl Acad Sci USA. 2017;114:9044–9.
Shiratori M, Tozaki-Saitoh H, Yoshitake M, Tsuda M, Inoue K. P2X7 receptor activation induces CXCL2 production in microglia through NFAT and PKC/MAPK pathways. J Neurochem. 2010;114:810–9.
Zelante T, Wong AYW, Mencarelli A, Foo S, Zolezzi F, Lee B, et al. Impaired calcineurin signaling in myeloid cells results in downregulation of pentraxin-3 and increased susceptibility to aspergillosis. Mucosal Immunol. 2017;10:470–80. https://doi.org/10.1038/mi.2016.52.
Bendickova K, Tidu F, Fric J. Calcineurin– NFAT signalling in myeloid leucocytes: new prospects and pitfalls in immunosuppressive therapy. EMBO Mol Med. 2017;9:990–9.
Martin KR, Layton D, Seach N, Corlett A, Barallobre MJ, Arbonés ML, et al. Upregulation of RCAN1 causes down syndrome-like immune dysfunction. J Med Genet. 2013;50:444–54.
Junkins RD, MacNeil AJ, Wu Z, McCormick C, Lin T-J. Regulator of calcineurin 1 suppresses inflammation during respiratory tract infections. J Immunol. 2013;190:5178–86.
Zhang S, Rahman M, Zhang S, Qi Z, Thorlacius H. Simvastatin antagonizes CD40L secretion, CXC chemokine formation, and pulmonary infiltration of neutrophils in abdominal sepsis. J Leukoc Biol. 2011;89:735–42.
Hwaiz R, Rahman M, Syk I, Zhang E, Thorlacius H. Rac1-dependent secretion of platelet-derived CCL5 regulates neutrophil recruitment via activation of alveolar macrophages in septic lung injury. J Leukoc Biol. 2015;97:975–84.
Hack C, De Groot E, Felt-Bersma R, Nuijens J, Strack Van Schijndel R, Eerenberg-Belmer A, et al. Increased plasma levels of interleukin-6 in sepsis. Blood. 1989;74:1704–10
Song J, Park DW, Moon S, Cho HJ, Park JH, Seok H, et al. Diagnostic and prognostic value of interleukin-6, pentraxin 3, and procalcitonin levels among sepsis and septic shock patients: a prospective controlled study according to the sepsis-3 definitions. BMC Infect Dis BMC Infect Dis. 2019;19:1–11.
Nemzek JA, Elisa I-. Six at six : interleukin-6 measured 6 H after the initiation of sepsis predicts mortality over 3 days. Daniel G. Remick,* Gerald R. Bolgos,* Javed Siddiqui,* Jungsoon Shin, † and. Crit Care Med. 2002;17:463–7.
Remick DG, Bolgos G, Copeland S, Siddiqui J. Role of interleukin-6 in mortality from and physiologic response to sepsis. Infect Immun. 2005;73:2751–7.
Murayama K, Sawamura M, Murakami H, Tamura J, Naruse T, Murakami H, et al. FK506 and cyclosporin A enhance IL-6 production in monocytes: a single-cell assay. Mediators Inflamm. 1994;3:375–80.
Ermert D, Zychlinsky A, Urban C. Fungal and bacterial killing by neutrophils. Methods Mol Biol. 2008;470:293–312.
Zweemer AJM, Toraskar J, Heitman LH, Ijzerman AP. Bias in chemokine receptor signalling. Trends Immunol. 2014;35:243–52. https://doi.org/10.1016/j.it.2014.02.004.
Rajagopal S, Bassoni DL, Campbell JJ, Gerard NP, Gerard C, Wehrman TS. Biased agonism as a mechanism for differential signaling by chemokine receptors. J Biol Chem. 2013;288:35039–48.
Emal D, Rampanelli E, Claessen N, Bemelman FJ, Leemans JC, Florquin S, et al. Calcineurin inhibitor tacrolimus impairs host immune response against urinary tract infection. Sci Rep. 2019;9:1–11. https://doi.org/10.1038/s41598-018-37482-x.
MacMillan D, Currie S, Bradley KN, Muir TC, McCarron JG. In smooth muscle, FK506-binding protein modulates IP3 receptor-evoked Ca2+ release by mTOR and calcineurin. J Cell Sci. 2005;118:5443–51.
Buckley C, Wilson C, McCarron JG. FK506 regulates Ca2+ release evoked by inositol 1,4,5-trisphosphate independently of FK-binding protein in endothelial cells. Br J Pharmacol. 2020;177:1131–49.
Tang WX, Chen YF, Zou AP, Campbell WB, Li PL. Role of FKBP12.6 in cADPR-induced activation of reconstituted ryanodine receptors from arterial smooth muscle. Am J Physiol Hear Circ Physiol. 2002;282:1304–10.
MacMillan D, Currie S, McCarron JG. FK506-binding protein (FKBP12) regulates ryanodine receptor-evoked Ca2+ release in colonic but not aortic smooth muscle. Cell Calcium. 2008;43:539–49.
MacMillan D, McCarron JG. Regulation by FK506 and rapamycin of Ca2+ release from the sarcoplasmic reticulum in vascular smooth muscle: the role of FK506 binding proteins and mTOR. Br J Pharmacol. 2009;158:1112–20.
Leon-Aparicio D, Chavez-Reyes J, Guerrero-Hernandez A. Activation of endoplasmic reticulum calcium leak by 2-APB depends on the luminal calcium concentration. Cell Calcium. 2017;65:80–90. https://doi.org/10.1016/j.ceca.2017.01.013.
Zhang S, Zhang S, Garcia-Vaz E, Herwald H, Gomez MF, Thorlacius H. Streptococcal M1 protein triggers chemokine formation, neutrophil infiltration, and lung injury in an NFAT-dependent manner. J Leukoc Biol. 2015;97:1003–10.
Zhang S, Luo L, Wang Y, Gomez MF, Thorlaciusa H. Nuclear factor of activated T cells regulates neutrophil recruitment, systemic inflammation, and T-cell dysfunction in abdominal sepsis. Infect Immun. 2014;82:3275–88.
Awla D, Zetterqvist AV, Abdulla A, Camello C, Berglund LM, Spégel P, et al. NFATc3 regulates trypsinogen activation, neutrophil recruitment, and tissue damage in acute pancreatitis in mice. Gastroenterology. 2012;143:1352–1360.e7. https://doi.org/10.1053/j.gastro.2012.07.098.
Tourneur E, Ben Mkaddem S, Chassin C, Bens M, Goujon JM, Charles N, et al. Cyclosporine A impairs nucleotide binding oligomerization domain (Nod1)-mediated innate antibacterial renal defenses in mice and human transplant recipients. PLoS Pathog. 2013;9:1–16.
Wang D, Chen X, Fu M, Xu H, Li Z. Tacrolimus increases the expression level of the chemokine receptor CXCR2 to promote renal fibrosis progression. Int J Mol Med. 2019;44:2181–8.
Acknowledgements
This study was supported by the São Paulo Research Foundation (FAPESP) (grants: 13/08216-2) and the National Council of Technological and Scientific Development (CNPq–Brazil). VFB and LSG hold a PhD scholarship from FAPESP (grants: 17/18264-5 and 20/08109-5).
Author information
Authors and Affiliations
Contributions
VFB, LSG, AK, and FQC designed the study. VFB, LSG, AK, FVSC, VVSM, DAD, FCR, AHS, GCMC, CMSS, and MHFL performed the mouse experiments. VFB, LSG, AK, and FQC wrote the manuscript. VFB, LSG, AK, FVSC, VVSM, AHS, GCMC, CMSS, MHFL, JPBV, TMC, CMCN, JCFAF, ASP, and FQC reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Responsible Editor: John Di Battista.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Borges, V., Galant, L.S., Kanashiro, A. et al. FK506 impairs neutrophil migration that results in increased polymicrobial sepsis susceptibility. Inflamm. Res. 72, 203–215 (2023). https://doi.org/10.1007/s00011-022-01669-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-022-01669-w