Abstract
Objective
MicroRNAs are a class of small, non-coding RNAs that play a key role in several biological and molecular processes, including tumorigenesis. We previously identified that MIR452 is upregulated in both colorectal cancer (CRC) and colitis. However, the functional mechanisms of MIR452 and its target genes in CRC and colitis are not well understood. So, we hypothesize that MIR452 can influence CRC and DSS-induced colitis model through the regulation of IL20RA and its downstream JAK-STATs signaling pathway.
Methods
We used a luciferase reporter assay to confirm the effect of MIR452 on IL20RA expression. The protein and mRNA expression of a target gene and its associated molecules were measured by western blot, quantitative RT-PCR, and immunohistochemistry.
Results
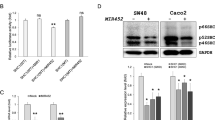
We found that the IL20RA was a direct target gene of MIR452. Overexpression of MIR452 in CRC cell lines significantly decreased IL20RA and its downstream Janus kinase 1 (JAK1), Signal transducer and activator of transcription 1 (STAT1) and STAT3. Knockdown of IL20RA in CRC cell lines by IL20RA gene silencing also decreased the expression of IL20RA, JAK1, and STAT3, but not of STAT1.
Conclusion
Our results suggest that MIR452 regulates STAT3 through the IL20RA-mediated JAK1 pathway, but not STAT1. Overall, MIR452 acts as tumor suppressor in human CRC and in a mouse colitis model. These findings suggest that MIR452 is a promising therapeutic target in the treatment of cancer and colitis.







Similar content being viewed by others
References
Kaplan GG. The global burden of IBD: from 2015 to 2025. Nat Rev Gastroenterol Hepatol. 2015;12(12):720–7.
Center MM, Jemal A, Ward E. International trends in colorectal cancer incidence rates. Cancer Epidemiol Prev Biomark. 2009;18(6):1688–94.
Fiocchi C. Inflammatory bowel disease: etiology and pathogenesis. Gastroenterology. 1998;115(1):182–205.
Nadeem MS, et al. Risk of colorectal cancer in inflammatory bowel diseases. Seminars in cancer biology. Amsterdam: Elsevier; 2019.
Ullman TA, Itzkowitz SH. Intestinal inflammation and cancer. Gastroenterology. 2011;140(6):1807–16.
Hoffmeister M, et al. Male sex and smoking have a larger impact on the prevalence of colorectal neoplasia than family history of colorectal cancer. Clin Gastroenterol Hepatol. 2010;8(10):870–6.
Hassan C, et al. Impact of lifestyle factors on colorectal polyp detection in the screening setting. Dis Colon Rectum. 2010;53(9):1328–33.
Alam KJ, et al. MicroRNA 375 regulates proliferation and migration of colon cancer cells by suppressing the CTGF-EGFR signaling pathway. Int J Cancer. 2017;141(8):1614–29.
Braga EA, et al. Molecular mechanisms in clear cell renal cell carcinoma: role of miRNAs and Hypermethylated miRNA genes in crucial oncogenic pathways and processes. Front Genet. 2019;10:320.
Kasinski AL, Slack FJ. MicroRNAs en route to the clinic: progress in validating and targeting microRNAs for cancer therapy. Nat Rev Cancer. 2011;11(12):849–64.
Mo JS, et al. MicroRNA 452 regulates cell proliferation, cell migration, and angiogenesis in colorectal cancer by suppressing VEGFA expression. Cancers. 2019;11(10):1613.
Lu J, et al. MicroRNA expression profiles classify human cancers. Nature. 2005;435(7043):834–8.
Garzon R, et al. MicroRNA expression and function in cancer. Trends Mol Med. 2006;12(12):580–7.
Mo J-S, et al. MicroRNA 429 regulates mucin gene expression and secretion in murine model of colitis. J Crohns Colitis. 2016;10(7):837–49.
Mo J-S, et al. MicroRNA 196B regulates FAS-mediated apoptosis in colorectal cancer cells. Oncotarget. 2015;6(5):2843.
Rutz S, Wang X, Ouyang W. The IL-20 subfamily of cytokines—from host defence to tissue homeostasis. Nat Rev Immunol. 2014;14(12):783–95.
Lee S-J, et al. Interleukin-20 promotes migration of bladder cancer cells through ERK-mediated MMP-9 expression leading to NF-kB activation by inducing the up-regulation of p21WAF1 expression. J Biol Chem. 2012;288:5539–52.
Mo J-S, et al. MicroRNA 429 regulates the expression of CHMP5 in the inflammatory colitis and colorectal cancer cells. Inflamm Res. 2018;67(11–12):985–96.
Han S-H, et al. Reduced microRNA 375 in colorectal cancer upregulates metadherin-mediated signaling. World J Gastroenterol. 2019;25(44):6495.
Pichler M, Calin GA. MicroRNAs in cancer: from developmental genes in worms to their clinical application in patients. Br J Cancer. 2015;113(4):569–73.
Van Rooij E, Kauppinen S. Development of micro RNA therapeutics is coming of age. EMBO Mol Med. 2014;6(7):851–64.
Qiu T, et al. MiR-145, miR-133a and miR-133b inhibit proliferation, migration, invasion and cell cycle progression via targeting transcription factor Sp1 in gastric cancer. FEBS Lett. 2014;588(7):1168–77.
Zhou W, et al. Cancer-secreted miR-105 destroys vascular endothelial barriers to promote metastasis. Cancer Cell. 2014;25(4):501–15.
Fang Y, et al. MicroRNA-7 inhibits tumor growth and metastasis by targeting the phosphoinositide 3-kinase/Akt pathway in hepatocellular carcinoma. Hepatology. 2012;55(6):1852–62.
Itzkowitz SH, Yio X. Inflammation and cancer IV Colorectal cancer in inflammatory bowel disease: the role of inflammation. Am J Physiol Gastrointest Liver Physiol. 2004;287(1):7–17.
Benderska N, et al. miRNA-26b overexpression in ulcerative colitis-associated carcinogenesis. Inflamm Bowel Dis. 2015;21(9):2039–51.
Li W, et al. Tumor-suppressive microRNA-452 inhibits migration and invasion of breast cancer cells by directly targeting RAB11A. Oncol Lett. 2017;14(2):2559–65.
Liu L, et al. Downregulation of miR-452 promotes stem-like traits and tumorigenicity of gliomas. Clin Cancer Res. 2013;19(13):3429–38.
Goto Y, et al. Regulation of E3 ubiquitin ligase-1 (WWP1) by microRNA-452 inhibits cancer cell migration and invasion in prostate cancer. Br J Cancer. 2016;114(10):1135–44.
Li R-Z, Wang L-M. RETRACTED ARTICLE: decreased microRNA-452 expression and its prognostic significance in human osteosarcoma. World J Surg Oncol. 2016;14(1):1–8.
Zheng Q, et al. MicroRNA-452 promotes tumorigenesis in hepatocellular carcinoma by targeting cyclin-dependent kinase inhibitor 1B. Mol Cell Biochem. 2014;389(1–2):187–95.
Liu S-G, et al. Differential expression of miRNAs in esophageal cancer tissue. Oncol Lett. 2013;5(5):1639–42.
Veerla S, et al. MiRNA expression in urothelial carcinomas: important roles of miR-10a, miR-222, miR-125b, miR-7 and miR-452 for tumor stage and metastasis, and frequent homozygous losses of miR-31. Int J Cancer. 2009;124(9):2236–42.
Chen J, Caspi RR, Chong WP. IL-20 receptor cytokines in autoimmune diseases. J Leukoc Biol. 2018;104(5):953–9.
Shabgah AG, et al. Interleukin-22 in human inflammatory diseases and viral infections. Autoimmun Rev. 2017;16(12):1209–18.
Kunz S, et al. Interleukin (IL)-19, IL-20 and IL-24 are produced by and act on keratinocytes and are distinct from classical ILs. Exp Dermatol. 2006;15(12):991–1004.
Fonseca-Camarillo G, et al. Expression of interleukin (IL)-19 and IL-24 in inflammatory bowel disease patients: a cross-sectional study. Clin Exp Immunol. 2014;177(1):64–75.
Fonseca-Camarillo G, et al. IL-10—and IL-20—expressing epithelial and inflammatory cells are increased in patients with ulcerative colitis. J Clin Immunol. 2013;33(3):640–8.
Andoh A, et al. Expression of IL-24, an activator of the JAK1/STAT3/SOCS3 cascade, is enhanced in inflammatory bowel disease. J Immunol. 2009;183(1):687–95.
Kiesler P, Fuss IJ, Strober W. Experimental models of inflammatory bowel diseases. Cell Mol Gastroenterol Hepatol. 2015;1(2):154–70.
Morris R, Kershaw NJ, Babon JJ. The molecular details of cytokine signaling via the JAK/STAT pathway. Protein Sci. 2018;27(12):1984–2009.
Nivarthi H, et al. The ratio of STAT1 to STAT3 expression is a determinant of colorectal cancer growth. Oncotarget. 2016;7(32):51096.
Nicholas C, Lesinski GB. The Jak-STAT signal transduction pathway in melanoma, breakthroughs in melanoma research. In: Tanaka Y (Ed.). London: InTech; 2011. pp. 283–306.
Becker C, et al. IL-6 signaling promotes tumor growth in colorectal cancer. Cell Cycle. 2005;4(2):220–3.
Corvinus FM, et al. Persistent STAT3 activation in colon cancer is associated with enhanced cell proliferation and tumor growth. Neoplasia (New York, NY). 2005;7(6):545.
Tsareva SA, et al. Signal transducer and activator of transcription 3 activation promotes invasive growth of colon carcinomas through matrix metal loproteinase induction. Neoplasia. 2007;9(4):279–91.
Acknowledgements
The biospecimens for this study were provided by the Biobank of Wonkwang University Hospital, a member of the National Biobank of Korea, which is supported by the Ministry of Health and Welfare. This research was supported by Wonkwang University in 2020.
Author information
Authors and Affiliations
Contributions
The authors have made the following declarations about their contributions: Conceived and designed the experiments: SCC. Performed the experiments, analyzed the data and discussion: SL, JSM, GS, and SCC. Contributed to draft and revision of the manuscript: SL, TYC and SCC.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving humans were in accordance with the Ethical Standards set by the Committee of Ethical Standards of Wonkwang University, Republic of Korea (WKIRB-201703-BR-011). The animal studies were approved by the Animal Care Committee of the Wonkwang University (WKU17-53).
Additional information
Responsible Editor: John Di Battista.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lamichhane, S., Mo, JS., Sharma, G. et al. MicroRNA 452 regulates IL20RA-mediated JAK1/STAT3 pathway in inflammatory colitis and colorectal cancer. Inflamm. Res. 70, 903–914 (2021). https://doi.org/10.1007/s00011-021-01486-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-021-01486-7