Abstract
Objective
Recently, Rodgersia podophylla has been reported to exhibit anti-inflammatory activity. However, little is known about the potential mechanisms about its anti-inflammatory activity. We elucidated the anti-inflammatory mechanisms of leaves extracts from Rodgersia podophylla (RP-L) in RAW264.7 cells.
Materials and methods
LPS-induced NO was measured by Griess and mRNA of pro-inflammatory mediators was analyzed by RT-PCR. Cell viability was measured using MTT assay. The protein level was analyzed by Western blot.
Results
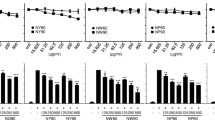
RP-L significantly inhibited the production of the pro-inflammatory mediators such as NO, iNOS, IL-1β and IL-6 in LPS-stimulated RAW264.7 cells. RP-L increased HO-1 expression in RAW264.7 cells, and the inhibition of HO-1 by ZnPP reduced the inhibitory effect of RP-L against LPS-induced NO production in RAW264.7 cells. Inhibition of p38, ROS and GSK3β attenuated RP-L-mediated HO-1 expression. Inhibition of ROS inhibited p38 phosphorylation and GSK3β expression induced by RP-L. In addition, inhibition of GSK3β blocked RP-L-mediated p38 phosphorylation. RP-L induced nuclear accumulation of Nrf2, and inhibition of p38, ROS and GSK3β abolished RP-L-mediated nuclear accumulation of Nrf2. Furthermore, RP-L blocked LPS-induced degradation of IκB-α and nuclear accumulation of p65. RP-L also attenuated LPS-induced phosphorylation of ERK1/2 and p38. In GC/MS analysis of RP-L, pyrogallol was detected as bioactive compound for anti-inflammatory activity of RP-L. Pyrogallol was observed to activate HO-1 expression through ROS/GSK3β/p38/Nrf2/HO-1 signaling.
Conclusions
Our results suggest that RP-L exerts potential anti-inflammatory activity by activating ROS/GSK3β/p38/Nrf2/HO-1 signaling and inhibiting NF-κB and MAPK signaling in RAW264.7 cells. These findings suggest that RP-L may have great potential for the development of anti-inflammatory drug.







Similar content being viewed by others
Abbreviations
- CO:
-
Carbon monoxide
- COX-2:
-
Cyclooxygenase-2
- DMSO:
-
Dimethyl sulfoxide
- ERK1/2:
-
Extracellular signal-regulated kinases 1/2
- GSK3β:
-
Glycogen synthase kinase 3 beta
- HO-1:
-
Heme oxygenase-1
- IKK:
-
IκB kinase
- IL-1β:
-
Interleukin-1β
- IL-6:
-
Interleukin-6
- iNOS:
-
Inducible nitric oxide synthase
- LPS:
-
Lipopolysaccharide
- MTT:
-
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
- JNK:
-
C-Jun N-terminal kinases
- MAPKs:
-
Mitogen-activated protein kinases
- NAC:
-
N-Acetyl-l-cysteine
- NF-κB:
-
Nuclear factor-κB
- NO:
-
Nitric oxide
- Nrf2:
-
NF-E2-related factor 2
- NSAIDs:
-
Nonsteroidal anti-inflammatory drugs
- PI3K:
-
Phosphoinositide 3-kinase
- ROS:
-
Reactive oxygen species
- RP-L:
-
Leave extracts from Rodgersia podophylla
- RT-PCR:
-
Reverse transcriptase-polymerase chain reaction
- ZnPP:
-
Zinc protoporphyrin IX
References
Furst R, Zundorf I. Plant-derived anti-inflammatory compounds: hopes and disappointments regarding the translation of preclinical knowledge into clinical progress. Mediators Inflamm. 2014;2014:146832.
Scrivo R, Vasile M, Bartosiewicz I, Valesini G. Inflammation as “common soil” of the multifactorial diseases. Autoimmun Rev. 2011;10(7):369–74.
Ghasemian M, Owlia S, Owlia MB. Review of anti-inflammatory herbal medicines. Adv Pharmacol Sci. 2016;2016:9130979.
Chin YW, Park EY, Seo SY, Yoon KD, Ahn MJ, Suh YG, Kim SG, Kim J. A novel iNOS and COX-2 inhibitor from the aerial parts of Rodgersia podophylla. Bioorg Med Chem Lett. 2006;16(17):4600–2.
Chin YW, Lim SW, Kim YC, Choi SZ, Lee KR, Kim J. Hepatoprotective flavonol glycosides from the aerial parts of Rodgersia podophylla. Planta Med. 2004;70(6):576–7.
Jang SE, Jeong JJ, Hyam SR, Han MJ, Kim DH. Ursolic acid isolated from the seed of Cornus officinalis ameliorates colitis in mice by inhibiting the binding of lipopolysaccharide to Toll-like receptor 4 on macrophages. J Agric Food Chem. 2014;62(40):9711–21.
Zang Y, Zhou X, Wang Q, Li X, Huang H. LncRNA FIRRE/NF-kB feedback loop contributes to OGD/R injury of cerebral microglial cells. Biochem Biophy Res Commun. 2018;501(1):131–8.
Ren J, Li L, Wang Y, Zhai J, Chen G, Hu K. Gambogic acid induces heme oxygenase-1 through Nrf2 signaling pathway and inhibits NF-κB and MAPK activation to reduce inflammation in LPS-activated RAW264.7 cells. Biomed Pharmacother. 2019;109:555–62.
Sun J, Ramnath RD, Zhi L, Tamizhselvi R, Bhatia M. Substance P enhances NF-kappaB transactivation and chemokine response in murine macrophages via ERK1/2 and p38 MAPK signaling pathways. Am J Physiol Cell Physiol. 2008;294(6):C1586–1596.
Bi CL, Wang H, Wang YJ, Sun J, Dong JS, Meng X, Li JJ. Selenium inhibits Staphylococcus aureus-induced inflammation by suppressing the activation of the NF-kappaB and MAPK signalling pathways in RAW264.7 macrophages. Eur J Pharmacol. 2016;780:159–65.
Otterbein LE, Choi AM. Heme oxygenase: colors of defense against cellular stress. Am J Physiol Lung Cell Mol Physiol. 2000;279(6):L1029–1037.
Dijkstra G, Blokzijl H, Bok L, Homan M, van Goor H, Faber KN, Jansen PL, Moshage H. Opposite effect of oxidative stress on inducible nitric oxide synthase and haem oxygenase-1 expression in intestinal inflammation: anti-inflammatory effect of carbon monoxide. J Pathol. 2004;204(3):296–303.
Wang WW, Smith DL, Zucker SD. Bilirubin inhibits iNOS expression and NO production in response to endotoxin in rats. Hepatology. 2004;40(2):424–33.
Yachie A, Niida Y, Wada T, Igarashi N, Kaneda H, Toma T, Ohta K, Kasahara Y, Koizumi S. Oxidative stress causes enhanced endothelial cell injury in human heme oxygenase-1 deficiency. J Clin Invest. 1999;103(1):129–35.
Poss KD, Tonegawa S. Reduced stress defense in heme oxygenase 1-deficient cells. Proc Natl Acad Sci USA. 1997;94(20):10925–30.
Choi AM, Alam J. Heme oxygenase-1: function, regulation, and implication of a novel stress-inducible protein in oxidant-induced lung injury. Am J Respir Cell Mol Biol. 1996;15(1):9–19.
Lin CY, Hsiao WC, Huang CJ, Kao CF, Hsu GS. Heme oxygenase-1 induction by the ROS-JNK pathway plays a role in aluminium-induced anemia. J Inorg Biochem. 2013;128:221–8.
Jin CH, Lee HJ, Park YD, Choi DS, Kim DS, Kang SY, Seo KI, Jeong IY. Isoegomaketone inhibits lipopolysaccharide-induced nitric oxide production in RAW 264.7 macrophages through the heme oxygenase-1 induction and inhibition of the interferon-beta-STAT-1 pathway. J Agric Food Chem. 2010;58(2):860–7.
Hu CM, Liu YH, Cheah KP, Li JS, Lam CS, Yu WY, Choy CS. Heme oxygenase-1 mediates the inhibitory actions of brazilin in RAW264.7 macrophages stimulated with lipopolysaccharide. J Ethnopharmacol. 2009;121(1):79–85.
Jun MS, Ha YM, Kim HS, Jang HJ, Kim YM, Lee YS, Kim HJ, Seo HG, Lee JH, Lee SH, Chang KC. Anti-inflammatory action of methanol extract of Carthamus tinctorius involves in heme oxygenase-1 induction. J Ethnopharmacol. 2011;133(2):524–30.
Lee DS, Jeong GS, Li B, Park H, Kim YC. Anti-inflammatory effects of sulfuretin from Rhus verniciflua Stokes via the induction of heme oxygenase-1 expression in murine macrophages. Int Immunopharmacol. 2010;10(8):850–8.
Dong L, Yin L, Chen R, Zhang Y, Hua S, Quan H, Fu X. Anti-inflammatory effect of calycosin glycoside on lipopolysaccharide-induced inflammatory responses in RAW 264.7 cells. Gene. 2018;675:94–101.
Shen Y, Sun Z, Guo X. Citral inhibits lipopolysaccharide-induced acute lung injury by activating PPAR-gamma. Eur J Pharmacol. 2015;747:45–51.
Zou J, Guo P, Lv N, Huang D. Lipopolysaccharide-induced tumor necrosis factor-alpha factor enhances inflammation and is associated with cancer (Review). Mol Med Rep. 2015;12(5):6399–404.
Tsoyi K, Kim HJ, Shin JS, Kim DH, Cho HJ, Lee SS, Ahn SK, Yun-Choi HS, Lee JH, Seo HG, Chang KC. HO-1 and JAK-2/STAT-1 signals are involved in preferential inhibition of iNOS over COX-2 gene expression by newly synthesized tetrahydroisoquinoline alkaloid, CKD712, in cells activated with lipopolysaccharide. Cell Signal. 2008;20(10):1839–47.
Hyung JH, Ahn CB, Il Kim B, Kim K, Je JY. Involvement of Nrf2-mediated heme oxygenase-1 expression in anti-inflammatory action of chitosan oligosaccharides through MAPK activation in murine macrophages. Eur J Pharmacol. 2016;793:43–8.
Jin GH, Park SY, Kim E, Ryu EY, Kim YH, Park G, Lee SJ. Anti-inflammatory activity of Bambusae Caulis in Taeniam through heme oxygenase-1 expression via Nrf-2 and p38 MAPK signaling in macrophages. Environ Toxicol Pharmacol. 2012;34(2):315–23.
Kim KC, Kang KA, Zhang R, Piao MJ, Kim GY, Kang MY, Lee SJ, Lee NH, Surh YJ, Hyun JW. Up-regulation of Nrf2-mediated heme oxygenase-1 expression by eckol, a phlorotannin compound, through activation of Erk and PI3K/Akt. Int J Biochem Cell Biol. 2010;42:297–305.
Park SY, Park DJ, Kim YH, Kim Y, Kim SG, Shon KJ, Choi YW, Lee SJ. Upregulation of heme oxygenase-1 via PI3K/Akt and Nrf-2 signaling pathways mediates the anti-inflammatory activity of schisandrin in Porphyromonas gingivalis LPS-stimulated macrophages. Immunol Lett. 2011;139(1–2):93–101.
Ryter SW, Xi S, Hartsfield CL, Choi AM. Mitogen activated protein kinase (MAPK) pathway regulates heme oxygenase-1 gene expression by hypoxia in vascular cells. Antioxid Redox Signal. 2002;4(4):587–92.
Jin CH, So YK, Han SN, Kim JB. Isoegomaketone upregulates heme oxygenase-1 in RAW264.7 cells via ROS/p38 MAPK/Nrf2 pathway. Biomol Ther. 2016;24(5):510–6.
McNally SJ, Harrison EM, Ross JA, Garden OJ, Wigmore SJ. Curcumin induces heme oxygenase 1 through generation of reactive oxygen species, p38 activation and phosphatase inhibition. Int J Mol Med. 2007;19(1):165–72.
Vene R, Cardinali B, Arena G, Ferrari N, Benelli R, Minghelli S, Poggi A, Noonan DM, Albini A, Tosetti F. Glycogen synthase kinase 3 regulates cell death and survival signaling in tumor cells under redox stress. Neoplasia. 2014;16(9):710–22.
Ko SH, da Rho J, Jeon JI, Kim YJ, Woo HA, Lee YK, Kim JM. Bacteroides fragilis enterotoxin upregulates heme oxygenase-1 in intestinal epithelial cells via a mitogen-activated protein kinase- and NF-kappaB-dependent pathway, leading to modulation of apoptosis. Infect Immun. 2016;84(9):2541–54.
Kansanen E, Kuosmanen SM, Leinonen H, Levonen AL. The Keap1-Nrf2 pathway: mechanisms of activation and dysregulation in cancer. Redox Biol. 2013;1:45–9.
Chin YW, Kim J. Three new flavonol glycosides from the aerial parts of Rodgersia podophylla. Chem Pharm Bull. 2006;54(2):234–6.
Nicolis E, Lampronti I, Dechecchi MC, Borgatti M, Tamanini A, Bianchi N, Bezzerri V, Mancini I, Giri MG, Rizzotti P, Gambari R, Cabrini G. Pyrogallol, an active compound from the medicinal plant Emblica officinalis, regulates expression of pro-inflammatory genes in bronchial epithelial cells. Int Immunopharmacol. 2008;8:1672–80.
Yin XJ, Ma JYC, Antonini JM, Castranova V, Ma JKH. Roles of reactive oxygen species and heme oxygenase-1 in modulation of alveolar macrophage-mediated pulmonary immune responses to Listeria monocytogenes by disel exhaust particles. Toxicol Sci. 2004;82:143–53.
Acknowledgements
This work was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2016R1D1A3B03931713 and NRF-2018R1A6A1A03024862).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Responsible Editor: John Di Battista.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kim, H.N., Kim, J.D., Park, S.B. et al. Anti-inflammatory activity of the extracts from Rodgersia podophylla leaves through activation of Nrf2/HO-1 pathway, and inhibition of NF-κB and MAPKs pathway in mouse macrophage cells. Inflamm. Res. 69, 233–244 (2020). https://doi.org/10.1007/s00011-019-01311-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-019-01311-2