Abstract
Background
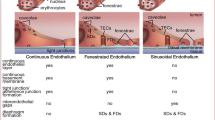
Angptl4 is a secreted protein involved in the regulation of vascular permeability, angiogenesis, and inflammatory responses in different kinds of tissues. Increases of vascular permeability and abnormality changes in angiogenesis contribute to the pathogenesis of tumor metastasis, ischemic-reperfusion injury. Inflammatory response associated with Angptl4 also leads to minimal change glomerulonephritis, wound healing. However, the role of Angptl4 in vascular permeability, angiogenesis, and inflammation is controversy. Hence, an underlying mechanism of Angptl4 in different kind of tissues needs to be further clarified.
Methods
Keywords such as angptl4, vascular permeability, angiogenesis, inflammation, and endothelial cells were used in search tool of PUBMED, and then the literatures associated with Angptl4 were founded and read.
Results
Data have established Angptl4 as the key modulator of both vascular permeability and angiogenesis; furthermore, it may also be related to the progression of metastatic tumors, cardiovascular events, and inflammatory diseases. This view focuses on the recent advances in our understanding of the role of Angptl4 in vascular permeability, angiogenesis, inflammatory signaling and the link between Angptl4 and multiple diseases such as cancer, cardiovascular diseases, diabetic retinopathy, and kidney diseases.
Conclusions
Taken together, Angptl4 modulates vascular permeability, angiogenesis, inflammatory signaling, and associated diseases. The use of Angptl4-modulating agents such as certain drugs, food constituents (such as fatty acids), nuclear factor (such as PPARα), and bacteria may treat associated diseases such as tumor metastasis, ischemic-reperfusion injury, inflammation, and chronic low-grade inflammation. However, the diverse physiological functions of Angptl4 in different tissues can lead to potentially deleterious side effects when used as a therapeutic target. In this regard, a better understanding of the underlying mechanisms for Angptl4 in different tissues is necessary.

Similar content being viewed by others
References
Goddard LM, Iruela-Arispe ML. Cellular and molecular regulation of vascular permeability. Thromb Haemost. 2013;109:407–15.
Armulik A, Genové G, Mäe M, Nisancioglu MH, Wallgard E, Niaudet C, et al. Pericytes regulate the blood-brain barrier. Nature. 2010;468:557–61.
Daneman R, Zhou L, Kebede AA, Barres BA. Pericytes are required for blood-brain barrier integrity during embryogenesis. Nature. 2010;468:562–6.
Mehta D. Signalling mechanisms regulating endothelial permeability. Physiol Rev. 2006;86:279–367.
Dvorak AM, Feng D. The vesiculo-vacuolar organelle (VVO). A new endothelial cell permeability organelle. J Histochem Cytochem. 2001;49:419–32.
Miyawaki-Shimizu K, Predescu D, Shimizu J, Broman M, Predescu S, Malik AB. siRNA-induced caveolin-1 knockdown in mice increases lung vascular permeability via the junctional pathway. Am J Physiol Lung Cell Mol Physiol. 2006;290:L405–13.
Thennes T, Mehta D. Heterotrimeric G proteins, focal adhesion kinase, and endothelial barrier function. Microvasc Res. 2012;83:31–44.
Sun Z, Li X, Massena S, Kutschera S, Padhan N, Gualandi L, et al. VEGFR2 induces c-Src signalling and vascular permeability in vivo via the adaptor protein TSAd. J Exp Med. 2012;209:1363–77.
Thibeault S, Rautureau Y, Oubaha M, Faubert D, Wilkes BC, Delisle C, et al. S-nitrosylation of beta-catenin by eNOS-derived NO promotes VEGF-induced endothelial cell permeability. Mol Cell. 2010;39:468–76.
Mammoto T, Parikh SM, Mammoto A, Gallagher D, Chan B, Mostoslavsky G, et al. Angiopoietin-1 requires p190 RhoGAP to protect against vascular leakage in vivo. J Biol Chem. 2007;282:23910–8.
Yao Q, Shin MK, Jun JC, Hernandez KL, Aggarwal NR, Mock FR, et al. Effect of chronic intermittment hypoxia on triglyceride uptake in different tissues. J Lipid Res. 2013;54:1058–65.
Zhu PC, GOH YY, Alison HF. Angiopoietin-like 4: a decade of research. Biosci Rep. 2011;32:211–9.
Valenzuela DM, Griffiths JA, Rojas J, Aldrich TH, Jones PF, Zhou H, et al. Angiopoietins 3 and 4: diverging gene counterparts in mice and humans. Proc Natl Acad Sci USA. 1996;96:1904–9.
Wiesner G, Morash BA, Ur E, Wilkinson M. Food restriction regulates adipose-specific cytokines in pituitary gland but not in hypothalamus. J Endocrinol. 2004;180:R1–6.
Koliwad SK, Kuo T, Shipp LE, Gray NE, Backhed F, Wang JC, et al. Angiopoietin-like 4(ANGPTL4, fasting-induced adipose factor) is a direct glucocorticoid receptor target and participates in glucocorticoid-regulated triglyceride metabolism. J. BiolChem. 2009;284:25593–601.
Kersten S, Mandard S, Tan NS, Escher P, Metzger D, Chambon P, et al. Characterization of the fasting-induced adipose factor FIAF, a novel peroxisome proliferators-activated receptor target gene. J Biol Chem. 2000;275:28488–93.
Kersten S, Lichtenstein L, Steenbergen E, Mudde K, Hendriks HJ, Hesselink MK, et al. Caloric restriction and exercises increase plasma ANGPTL4 levels in humans via elevated free fatty acids. Arterioscler Thromb Vasc Biol. 2009;29:969–74.
Padua D, Zhang XHF, Wang Q, Nadal C, Gerald WL, Gomis RR, et al. TGFbeta primes breast tumors for lung metastasis seeding through angiopoietin-like 4. Cell. 2008;133:66–77.
Clement LC, Avila-Casado C, Mace C, Soria E, Bakker WW, Kersten S, et al. Podocyte-secreted angiopoietin-like-4 mediates proteinuria in glucocorticoid-sensitive nephrotic syndrome. Nat Med. 2011;17:117–22.
Galaup A, Cazes A, Le Jan S, Phlippe J, Connault E, CozE Le, et al. Angiopoietin-like 4 prevents metastasis through inhibition of vascular permeability and tumor cell motility and invasiveness. Proc Natl Acad Sci USA. 2006;103:18721–6.
Huang RL, Teo Z, Chong HC, Zhu P, Tan MJ, Tan CK, et al. ANGPTL4 modulates vascular junction integrity by integrin signaling and disruption of intercellular VE-cadherin and claudin-5 clusters. Blood. 2011;118:3990–4002.
Katanasaka Y, Kodera Y, Kitamura Y, Morimoto T, Tamura T, Koizumi F. Epidermal growth factor receptor variant type III markedly accelerates angiogenesis and tumor growth via inducing c-myc mediated angiopoietin-like 4 expression in malignant glioma. Mol Cancer. 2013;25:12–31.
Perdiguero EG, Galaup A, Durand M, Teillon J, Phillippe J, Valenzuela DM, et al. Alteration of developmental and pathological retinal angiogenesis in angptl4-deficient mice. J Biol Chem. 2011;286:36841–51.
Mandard S, Zandbergen F, Tan NS, Escher P, Patsouris D, Koenig W, et al. The direct peroxisome proliferator-activated receptor target fasting-induced adipose factor (FIAF/PGAR/ANGPTL4) is present in blood plasma as a truncated protein that is increased by fenofibrate treatment. J Biol Chem. 2004;279:34411–20.
Ge H, Yang G, Huang L, Motola DL, Pourbahrami T, Li C. Oligomerization and regulated proteolytic processing of angiopoietin-like protein 4. J Biol Chem. 2004;279:2038–45.
Lei X, Shi F, Basu D, Huq A, Routhier S, Day R, et al. Proteolytic processing of angiopoietin-like protein 4 by proprotein convertases modulates its inhibitory effects on lipoprotein lipase activity. J Biol Chem. 2011;286:15747–56.
Mandard S, Zandbergen F, Tan NS, Escher P, Patsouris D, Koenig W, et al. The direct peroxisome proliferator-activated receptor target fasting-induced adipose factor (FIAF/PGAR/ANGPTL4) is present in blood plasma as a truncated protein that is increased by fenofibrate treatment. J Biol Chem. 2004;279:34411–20.
Stapleton CM, Joo JH, Kim Y-S, Liao G, Panettieri RA Jr, Jetten AM. Induction of ANGPTL4 expression in human airway smooth muscle cells by PMA through activation of PKC and MAPK pathways. Exp Cell Res. 2010;316:507–16.
Yang YH, Wang Y, Lam KS, Yau MH, Cheng KK, Zhang J, et al. Supression of the Raf/MEK/ERK signaling cascade and inhibition of angiogenesis by the carboxyl terminus of angiopoietin-like protein 4. Arterioscler Thromb Vasc Biol. 2008;28:835–40.
Ito Y, Olike Y, Yasunaga K, Hamada K, Miyata K, Matsumoto SI, et al. Inhibition of angiogenesis and vascular leakiness by angiopoietin-related protein 4. Cancer Res. 2003;63:6651–7.
Hutchings H, Ortega N, Plouet J. Extracellular matrix-bound vascular endothelial growth factor promotes endothelial cell adhesion, migration, and survival through integrin ligation. FASEB J. 2003;17:1520–2.
Okochi-Takada E, Hattori N, Tsukamoto T, Miyamoto K, Ando T, Ito S, et al. ANGPTL4 is a secreted tumor suppressor that inhibits angiogenesis. Oncogene. 2013; 174 [Epub ahead of print].
Le Jan S, Amy C, Cazes A, Monnot C, Lamande N, Favier J, et al. Angiopoietin-like 4 is a proangiogenic factor produced during ischemia and in conventional renal cell carcinoma. Am J Pathol. 2003;162:1521–8.
Goh YY, Pal M, Chong HC, Zhu P, Tan MJ, Punugu L, et al. Angiopoietin-like 4 interacts with matrix proteins to modulate wound healing. J Biol Chem. 2010;285:32999–3009.
Galaup A, Gomez E, Souktani R, Durand M, Cazes A, Monnot C, et al. Protection aganist myocardial infarction and no-reflow through preservation of vascular integrity by angptl4. Circulation. 2012;125:140–9.
Quintero P, Gonzalez-Muniesa P, Martinez JA. Influence of different oxygen supply on metabolic markers and gene response in murine adipocytes. J Biol Requl Homeost Agents. 2012;26:379–88.
Lichtenstein L, Mattijssen F, de Wi NJ, Georgiadi A, Hooiveld GJ, van der Meer R, et al. ANGPTL4 protects against severe proinflammatory effects of saturated fat by inhibiting fatty acid uptake into mesenteric lymph node macrophages. Cell Metab. 2010;12:580–92.
Brown R, Imran SA, Wilkinson M. Lipopolysaccharide (LPS) stimulates adipokine and socs3 gene expression in mouse brain and pituitary gland in vivo, and in N-1 hypothalamic neurons in vitro. J Neuroimmunol. 2009;209:96–103.
Lu B, Moser A, Shigenaga JK, Grunfeld C, Feingold KR. The acute phase response stimulates the expression of angiopoietin like protein 4. Biochem Biophys Res Commun. 2010;391:1737–41.
Rummel C, Inoue W, Sachot C, Poole S, Hubschle T, Luheshi GN. Selective contribution of interleukin-6 and leptin to brain inflammatory signals induced by systemic LPS injection in mice. J Comp Neurol. 2008;511:373–95.
Zhu P, Tan MJ, Huang RL, Tan CK, Chong HC, Pal M, et al. Angiopoietin-like 4 protein elevates the prosurvival intracellular O2 −:H2O2 ratio and confers anoikis resistance to tumors. Cancer Cell. 2011;19:401–15.
Nakayama T, Hirakawa H, Shibata K, Nazneen A, Abe K, Naqayasu T, et al. Expression of angiopoietin-like 4(ANGPTL4) in human colorectal cancer: ANGPTL4 promotes venous invasion and distant metastasis. Oncol Rep. 2011;25:929–35.
Shibata K, Nakayama T, Hirakama H, Hidaka S, Naqayasu T. Clinicopathological significance of angiopoietin-like protein 4 expression in oesophageal squamous cell carcinoma. J Clin Pathol. 2010;63:1054–8.
Ma T, Jham BC, Hu J, Friedman ER, Basile JR, Molinolo A, et al. Viral G protein-coupled receptor up-regulates angiopoietin-like 4 promoting angiogenesis and vascular permeability in Kaposi’s sarcoma. PNAS. 2010;107:14363–8.
Reiser J. Filtering new facts about kidney disease. Nat Med. 2011;17:44–5.
Werner S, Grose R. Regulation of wound healing by growth factors and cytokines. Physiol Rev. 2003;83:835–70.
Goh YY, Pal M, Chong HC, Zhu P, Tan MJ, Punugu L, et al. Angiopoietin-like 4 interacts with integrins beta1 and beta5 to modulate keratinocyte migration. Am J Pathol. 2010;177:2791–803.
Musso G, Gambino R, Cassader M. Interactions between gut microbiota and host metabolism predisposing to obesity and diabetes. Annu Rev Med. 2011;62:361–80.
Lichtenstein L, Berbée JF, van Dijk SJ, van Dijk KW, Bensadoun A, Kema IP, et al. Angptl4 upregulates cholesterol synthesis in liver via inhibition of LPL- and HL-dependent hepatic cholesterol uptake. Arterioscler Thromb Vasc Biol. 2007;27:2420–7.
Adachi H, Fujiwara Y, Kondo T, Nishikawa T, Ogawa R. Angptl 4 deficiency improves lipid metabolism, suppresses foam cell formation and protects against atherosclerosis. Biochem Biophys Res Commun. 2009;379:806–11.
Romeo S, Pennacchio LA, Fu Y, Boerwinkle E, Tybjaerg-Hansen A, Hobbs HH, et al. Population-based resequencing of ANGPTL4 uncovers variations that reduce triglycerides and increase HDL. Nat Genet. 2007;39:513–6.
Folsom AR, Peacock JM, Demerath E, Boerwinkle E. Variation in ANGPTL4 and risk of coronary heart disease: the atherosclerosis risk in communities study. Metabolism. 2008;57:1591–6.
Jonker JT, Smit JW, Hammer S, Snel M, van der Meer RW, Lamb HJ, et al. Dietary modulation of plasma angiopoietin-like protein 4 concentrations in healthy volunteers and in patients with type 2 diabetes. Am J Clin Nutr. 2013;97:255–60.
Xu A, Lam MC, Chan KW, Wang Y, Zhang J. Angiopoietin-like protein 4 decreases blood glucose and improves glucose tolerance but induces hyperlipidemia and hepatic steatosis in mice. Proc Natl Acad Sci USA. 2005;102:6086–91.
Yokouchi H, Eto K, Nishimura W, Takeda N, Kaburagi Y, Yamamoto S, et al. Angiopoietin-like protein 4 (ANGPTL4) is induced by high glucose in retinal pigment epithelial cells and exhibits potent angiogenic activity on retinal endothelial cells. Acta Ophthalmol. 2013;91:e289–97.
Bouleti C, Mathivet T, Coqueran B, Serfaty JM, Lesaqe M, Berland E, et al. Protective effects of angiopoietin-like 4 on cerebrovascular and functional damages in ischaemic stroke. Eur Heart J. 2013; [Epub ahead of print].
Acknowledgments
This work was supported by the Pudong New Area Health Bureau Foundation of Shanghai PWRd2012-07, the National Natural Science Foundation of China (NO. 81270130) and the National Natural Science Foundation of China(NO.81070056).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Responsible Editor: Ikuo Morita.
L. Guo and S.-Y. Li contributed equally to this work.
Rights and permissions
About this article
Cite this article
Guo, L., Li, SY., Ji, FY. et al. Role of Angptl4 in vascular permeability and inflammation. Inflamm. Res. 63, 13–22 (2014). https://doi.org/10.1007/s00011-013-0678-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-013-0678-0