Abstract
Objectives
The objective of the paper was to determine whether circulating stem-progenitor cells were elevated along with its mobilizing cytokines in neonatal respiratory distress syndrome (RDS).
Subjects and methods
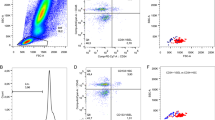
Circulating CD34+ cells were identified by flow cytometry in 41 RDS in comparison with 20 preterm and 14 term controls without diffuse lung diseases. Plasma concentrations of vascular endothelial growth factor, stromal cell-derived factor-1 (SDF-1) and granulocyte–macrophage colony-stimulating factor were determined by immunochemical assays.
Results
The number of CD34+ cells was significantly higher in RDS [25(6–174) cells/μl] than in the preterm controls [15(1–100) cells/μl, P < 0.05]. RDS survivors had higher level of CD34+ cells than non-survivors (P < 0.05), and low CD34+ cell level in RDS was correlated with prolonged duration of ventilation (r = −0.396, P < 0.05). Likewise, the CD34+ cell level was inversely associated with Score for Neonatal Acute Physiology Perinatal Extension II (r = −0.473, P < 0.01) in RDS. Plasma SDF-1 concentration was significantly higher in RDS than in the preterm controls (P < 0.01), and was correlated with the level of CD34+ cells (r = 0.305, P < 0.01).
Conclusions
The level of circulating CD34+ cells was elevated in RDS along with an increase of plasma SDF-1, suggesting CD34+ cells might be involved in reparation of neonatal lung injury.



Similar content being viewed by others
References
Mariani GL, Carlo WA. Ventilatory management in neonates. Science or art? Clin Perinatol. 1998;25:33–48.
Lukkarinen HP, Laine J, Kääpä PO. Lung epithelial cells undergo apoptosis in neonatal respiratory distress syndrome. Pediatr Res. 2003;53:254–9.
Yamada M, Kubo H, Kobayashi S, Ishizawa K, Numasaki M, Ueda S, et al. Bone marrow-derived progenitor cells are important for lung repair after lipopolysaccharide-induced lung injury. J Immunol. 2004;172:1266–72.
Gupta N, Su X, Popov B, Lee JW, Serikov V, Matthay MA. Intrapulmonary delivery of bone marrow-derived mesenchymal stem cells improves survival and attenuates endotoxin-induced acute lung injury in mice. J Immunol. 2007;179:1855–63.
Ishizawa K, Kubo H, Yamada M, Kobayashi S, Numasaki M, Ueda S, et al. Bone marrow-derived cells contribute to lung regeneration after elastase-induced pulmonary emphysema. FEBS Lett. 2004;556:249–52.
Kleeberger W, Versmold A, Rothamel T, Glockner S, Bredt M, Haverich A, et al. Increased chimerism of bronchial and alveolar epithelium in human lung allografts undergoing chronic injury. Am J Pathol. 2003;162:1487–94.
Kotton DN, Ma BY, Cardoso WV, Sanderson EA, Summer RS, Williams MC, et al. Bone marrow-derived cells as progenitors of lung alveolar epithelium. Development. 2001;128:5181–8.
Palange P, Testa U, Huertas A, Calabrò L, Antonucci R, Petrucci E, et al. Circulating haemopoietic and endothelial progenitor cells are decreased in COPD. Eur Respir J. 2006;27:529–41.
Fadini GP, Schiavon M, Cantini M, Baesso I, Facco M, Miorin M, et al. Circulating progenitor cells are reduced in patients with severe lung disease. Stem Cells. 2006;24:1806–13.
Gehling UM, Ergün S, Schumacher U, Wagener C, Pantel K, Otte M, et al. In vitro differentiation of endothelial cells from AC133-positive progenitor cells. Blood. 2000;95:3106–12.
Yamada M, Kubo H, Ishizawa K, Kobayashi S, Shinkawa M, Sasaki H. Increased circulating endothelial progenitor cells in patients with bacterial pneumonia: evidence that bone marrow derived cells contribute to lung repair. Thorax. 2005;60:410–3.
Burnham EL, Taylor WR, Quyyumi AA, Rojas M, Brigham KL, Moss M. Increased circulating endothelial progenitor cells are associated with survival in acute lung injury. Am J Respir Crit Care Med. 2005;172:854–60.
Bizzarro MJ, Bhandari V, Krause DS, Smith BR, Gross I. Circulating stem cells in extremely preterm neonates. Acta Paediatr. 2007;96:521–5.
Balasubramaniam V, Mervis CF, Maxey AM, Markham NE, Abman SH. Hyperoxia reduces bone marrow, circulating, and lung endothelial progenitor cells in the developing lung: implications for the pathogenesis of bronchopulmonary dysplasia. Am J Physiol Lung Cell Mol Physiol. 2007;292:L1073–84.
Asahara T, Takahashi T, Masuda H, Kalka C, Chen D, Iwaguro H, et al. VEGF contributes to postnatal neovascularization by mobilizing bone marrow-derived endothelial progenitor cells. EMBO J. 1999;18:3964–72.
Askari AT, Unzek S, Popovic ZB, Goldman CK, Forudi F, Kiedrowski M, et al. Effect of stromal-cell-derived factor 1 on stem-cell homing and tissue regeneration in ischaemic cardiomyopathy. Lancet. 2003;362:697–703.
Aicher A, Zeiher AM, Dimmeler S. Mobilizing endothelial progenitor cells. Hypertension. 2005;45:321–5.
Jobe AH, Bancalari E. Bronchopulmonary dysplasia. Am J Respir Crit Care Med. 2001;163:1723–9.
Barnett D, Janossy G, Lubenko A, Matutes E, Newland A, Reilly JT. Guideline for the flow cytometric enumeration of CD34+ haematopoietic stem cells. Clin Lab Haematol. 1999;21:301–8.
Loebinger MR, Janes SM. Stem cells for lung disease. Chest. 2007;132:279–85.
Krause DS. Bone marrow-derived lung epithelial cells. Proc Am Thorac Soc. 2008;5:699–702.
Rafat N, Hanusch C, Brinkkoetter PT, Schulte J, Brade J, Zijlstra JG, et al. Increased circulating endothelial progenitor cells in septic patients: correlation with survival. Crit Care Med. 2007;35:1677–84.
Borghesi A, Massa M, Campanelli R, Bollani L, Tzialla C, Figar TA, et al. Circulating endothelial progenitor cells in preterm infants with bronchopulmonary dysplasia. Am J Respir Crit Care Med. 2009;180:540–6.
Baker CD, Ryan SL, Ingram DA, Seedorf GJ, Abman SH, Balasubramaniam V. Endothelial colony-forming cells from preterm infants are increased and more susceptible to hyperoxia. Am J Respir Crit Care Med. 2009;180:454–61.
Abe S, Boyer C, Liu X, Wen FQ, Kobayashi T, Fang Q, et al. Cells derived from the circulation contribute to the repair of lung injury. Am J Respir Crit Care Med. 2004;170:1158–63.
Kähler CM, Wechselberger J, Hilbe W, Gschwendtner A, Colleselli D, Niederegger H, et al. Peripheral infusion of rat bone marrow derived endothelial progenitor cells leads to homing in acute lung injury. Respir Res. 2007;8:50.
Lapidot T, Petit I. Current understanding of stem cell mobilization: the roles of chemokines, proteolytic enzymes, adhesion molecules, cytokines, and stromal cells. Exp Hematol. 2002;30:973–81.
Aiuti A, Webb IJ, Bleul C, Springer T, Gutierrez-Ramos JC. The chemokine SDF-1 is a chemoattractant for human CD34+ hematopoietic progenitor cells and provides a new mechanism to explain the mobilization of CD34+ progenitors to peripheral blood. J Exp Med. 1997;185:111–20.
Abbott JD, Huang Y, Liu D, Hickey R, Krause DS, Giordano FJ. Stromal cell-derived factor-1alpha plays a critical role in stem cell recruitment to the heart after myocardial infarction but is not sufficient to induce homing in the absence of injury. Circulation. 2004;110:3300–5.
De Falco E, Porcelli D, Torella AR, Straino S, Iachininoto MG, Orlandi A, et al. SDF-1 involvement in endothelial phenotype and ischemia-induced recruitment of bone marrow progenitor cells. Blood. 2004;104:3472–82.
Acknowledgments
We thank Mrs. Qian Zhang and Ying Wang for the flow cytometry analysis, and assistance by staff from the Department of Neonatology is highly appreciated. This study was supported by the National Natural Science Foundation (No. 30600687) of China.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: A. Bauhofer.
Rights and permissions
About this article
Cite this article
Qi, Y., Qian, L., Sun, B. et al. Circulating CD34+ cells are elevated in neonates with respiratory distress syndrome. Inflamm. Res. 59, 889–895 (2010). https://doi.org/10.1007/s00011-010-0201-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-010-0201-9