Abstract
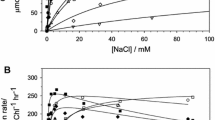
pH-dependent inactivation of Photosystem (PS) II and related quenching of chlorophyll-a-fluorescence have been investigated in isolated thylakoids and PS II-particles and related to calcium release at the donor side of PS II. The capacity of oxygen evolution (measured under light saturation) decreases when the ΔpH is high and the pH in the thylakoid lumen decreases below 5.5. Oxygen evolution recovers upon uncoupling. The pH-response of inactivation can be described by a 1 H+-transition with an apparent pK-value of about 4.7. The yield of variable fluorescence decreases in parallel to the inactivation of oxygen evolution. pH-dependent quenching requires light and can be inhibited by DCMU. In PS II-particles, inactivation is accompanied by a reversible release of Ca2+-ions (one Ca2+ released per 200 Chl). In isolated thylakoids, where a ΔpH was created by ATP-hydrolysis, both inactivation of oxygen evolution (and related fluorescence quenching) by internal acidification and the recovery of that inactivation can be suppressed by calcium-channel blockers. In the presence of the Ca2+-ionophore A23187, recovery of Chl-fluorescence (after relaxation of the ΔpH) is stimulated by external Ca2+ and retarded by EGTA. As shown previously (Krieger and Weis 1993), inactivation of oxygen evolution at low pH is accompanied by an upward shift of the midpoint redox-potential, Em, of QA. Here, we show that in isolated PS II particles the pH-dependent redox-shift (about 160 mV, as measured from redox titration of Chl-fluorescence) is suppressed by Ca2+-channel blockers and DCMU. When a redox potential of −80 to −120mV was established in a suspension of isolated thylakoids, the primary quinone acceptor, QA, was largely reduced in presence of a ΔpH (created by ATP-hydrolysis) but oxidized in presence of an uncoupler. Ca2+-binding at the lumen side seems to control redox processes at the lumen- and stroma-side of PS II. We discuss Ca2+-release to be involved in the physiological process of ‘high energy quenching’.
Similar content being viewed by others
Abbreviations
- Chl:
-
Chlorophyll
- DCMU:
-
3-(3′,4′-dichloro-phenyl)-1,1-dimethylurea
- DMBQ:
-
2,5-dimethylbenzoquinone
- EGTA:
-
ethylengylcol bis(β-aminoethylether)-N,N,N′,N′-tetra-acetate
- HEPES:
-
4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid
- MES:
-
4-morpholino ethanesulfonic acid
- MOPS:
-
4-morpholine-propanesulfonic acid
- P680 :
-
reaction center chlorophyll in Photosystem II
- Pheo:
-
pheophytin
- QA, QB :
-
primary and secondary quinone acceptor of Photosystem II
- tyrZ :
-
redox actie tyrosine (electron donor)
References
Bertold DA, Babcock GT and Yocum CF (1981) A highly resolved, oxygen-evolving Photosystem II preparation from spinach thylakoid membranes. FEBS Lett 134: 231–234
Bilger W, Björkman O and Thayer SS (1989) Light-induced spectral absorbance changes in relation to photosynthesis and the epoxidation state of xanthophyll cycle components in cotton leaves. Plant Physiol 91: 542–551
Blubaugh D, Atamian M, Babcock GT, Goldbeck JH and Cheniae GM (1991) Photoinhibition of hydroxylamin-extracted Photosystem II membranes: Identification of the sites of photodamage. Biochemistry 30: 7586–7597
Boussac A and Rutherford AW (1988) Ca2+-binding to the oxygen evolving enzyme varies with the redox state of the Mn cluster. FEBS Lett 236: 432–436
Boussac A and Rutherford AW (1992) The involvement of Ca2+ in the Ca2+-effect on Photosystem II oxygen evolution. Photosynth Res 33: 208–209
Boussac A, Maison-Peteri B, Etienne A-L and Vernotte C (1985) Reactivation of oxygen evolution of NaCl-washed Photosystem II particles by Ca2+ and/or the 24kDa protein, Biochim Biophys Acta 808: 225–230
Boussac A, Zimmermann J-L, Rutherford AW and Lavergne J (1990) Histidine oxidation in the oxygen-evolving Photosystem II enzme. Nature 347, No 6290: 303–306
Boussac A, Sétif P and Rutherford AW (1992) Inhibition of Tyrosine Z photooxidation after formation of the S3-state in Ca2+-depleted and Cl−-depleted Photosystem II. Biochemistry 31: 1224–1233
Briantais J-M, Vernotte C, Picaud M and Krause GH (1979) A quantitative study of the slow decline of chlorophylla fluorescence in isolated chloroplasts. Biochim Biophys Acta 548: 128–138
Carpentier R and Nakatani HY (1985) Inhibitors affecting the oxidizing side of Photosystem II at the Ca2+- and Cl−-sensitive sites. Biochim Biophys Acta 808: 288–292
Crofts J and Horton P (1991) Dissipation of excess energy by Photosystem II particles at low pH. Biochim Biophys Acta 1058: 187–193
Debus JD (1992) The manganese and calcium ions of photosynthetic oxygen evolution. Biochim Biophys Acta 1102: 269–352
Demmig-Adams B (1990) Carotenoids and photoprotection in plants: A role of the xanthophyll zeaxanthin. Biochim Biophys Acta 1020: 1–24
Dietz KJ, Schreiber U and Heber U (1985) The relationship between the redox state of QA and photosynthesis in leaves at various carbon dioxide, oxygen and light regimes. Planta 166: 219–226
Diner BA and Delosme R (1983) Oxidation-reduction properties of the electron acceptors of Photosystem II, II. Redox titrations at various pH values of the flash-induced formation of C550 inChlamydomonas Photosystem II particles lacking the secondary quinone electron acceptor. Biochim Biophys Acta 722: 452–459
Eckert H-J, Geiken B, Bernarding J, Napiwotzki A, Eichler H-J and Renger G (1991) Two sites of photoinhibition of the electron transfer in oxygen evolving and Tris-treated PS II membrane fragments from spinach. Photosynth Res 27: 97–108
Genty B, Briantais J-M and Baker NR (1989) The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochim Biophys Acta 990: 87–92
Gilmore AU and Yamamoto HY (1992) Dark induction of zeaxanthin-dependent nonphotochemical quenching mediated by ATP. Proc Natl Acad Sci USA 89: 1899–1903
Grynkiewicz G, Poenie M and Tsien RY (1985) A new generation of Ca2+ indicators with greatly improved fluorescence properties. J Biol Chem 260(6): 3440–3450
Hager A (1969) Lichtbedingte pH-Erniedrigung in einem Chloroplasten Kompartiment als Ursache der enzymatischen Violaxanthin → Zeaxanthin-Umwandlung: Beziehung zur Photophosphorylierung. Planta 89: 224–243
Heber U, Neimanis S, Siebke K, Schönknecht G and Katona E (1992) Chloroplast energization and oxidation of P700/ plastocyanin in illuminated leaves at reduced levels of CO2 or oxygen. Photosynth Res 34: 433–447
Homann PH (1988) The chloride and calcium requirement of photosynthetic water oxidation: Effects of pH. Biochim Biophys Acta 934: 1–13
Horton P and Hague A (1988) Studies on the induction of chlorophyll fluorescence in isolated barley protoplasts. IV. Resolution of non-photochemical quenching. Biochim Biophys Acta 932: 107–115
Horton P, Oxborough K, Rees D and Scholes JD (1988) Regulation of the photochemical efficiency of Photosystem II; consequences for the light response of field photosynthesis. Plant Physiol Biochim 26: 453–460
Horton P, Ruban AV, Rees D, Pascal AA, Noctor G and Young AJ (1991) Control of the light-harvesting function of chloroplast membranes by aggregation of the LHC II chlorophyll-protein complex. FEBS Lett 292: 1–4
Jegerschöld C, Virgin I and Styring S (1990) Light-dependent degradation of the D1-protein in Photosystem II is accelerated after inhibition of the water splitting reaction. Biochemistry 29: 6170–6186
Krause GH and Weis E (1991) Chlorophyll fluorescence and photosynthesis: The basics. Ann Rev Plant Physiol Plant Mol Biol 42: 313–349
Krause GH, Laasch H and Weis E (1988) Regulation of thermal dissipation of absorbed light energy in chloroplasts indicated by energy-dependent fluorescence quenching. Plant Physiol Biochem 26: 445–452
Krieger A and Weis E (1993) Energy-dependent quenching of chlorophyll-a-fluorescence: The involvement of protoncalcium exchange at Photosystem II. Photosynthetica 27(1–2): 89–98
Krieger A, Moya I and Weis E (1992) Energy-dependent quenching of chlorophyll-a-fluorescence: Effect of pH on stationary fluorescence and picosecond-relaxation kinetics in thylakoid membranes and Photosystem II preparations. Biochim Biophys Acta 1102: 167–176
Krieger A, Weis E and Demeter S (1993) Low pH-induced Ca ion release in the water-splitting system is accompanied with a shift in the midpoint redox potential of the primary quinone aceptor, QA. Biochim Biophys Acta (in press)
Laasch H (1987) Non-photochemical quenching of Chlorophyll-a-fluorescence in isolated chloroplasts under conditions of stressed photosynthesis. Planta 171: 220–226
Laisk A, Kiirats O, Oja V, Weis E and Heber U (1992) Analysis of oxygen evolution during photosynthetic induction and in multiple-turnover flashes in sunflower leaves. Planta 186: 434–441
Onishi ST (1987) Characterization of the murexid method: Dual-wavelength spectrometry of cations under physiological conditions. Analyt Biochem 85: 165–179
Ono T and Inoue Y (1988) Discrete extraction of Ca atom functional for O2-evolution in higher plant Photosystem II by a simple low pH treatment. FEBS Lett 227: 147–152
Ono T and Inoue Y (1989) Removal of Ca by pH3.0 treatment inhibits S2 to S3 transition in photosynthetic oxygen evolution system. Biochim Biophys Acta 973: 443–449
Oxborough K and Horton P (1987) Characterization of the effects of Antimycin A upon high energy state quenching of chlorophyll fluorescence (qE) in spinach and pea chloroplasts. Photosynthe Res 12: 119–128
Rees D, Noctor G, Ruban AV, Crofts J, Young A and Horton P (1992) pH-dependent chlorophyll fluorescence quenching in spinach thylakoids from light treated or dark-adapted leaves. Photosynth Res 31: 11–19
Rutherford AW, Zimmermann J-L and Boussac A (1992) Oxygen evolution. In: Barber J (ed) The Photosystems: Structure, Function and Molecular Biology, pp 179–230. Elsevier, Amsterdam
Schlodder E and Meyer B (1987) pH-dependence of oxygen evolution and reduction kinetics of photooxidized chlorophylla II (P-680) in Photosystem II particles fromSynechococcus sp. Biochim Biophys Acta 890: 23–31
Schreiber U and Avron M (1979) Properties of ATP-driven reverse electron flow in chloroplasts. Biochim Biophys Acta 546: 436–447
Schreiber U and Neubauer C (1989) Correlation between dissipative fluorescence quenching at Photosystem II and 50 μ s recombination luminescence. FEBS Lett 258: 339–342
Schreiber U and Neubauer C (1990) O2-dependent electron flow, membrane energization and the mechanism of non-photochemical quenching of chlorophyll fluorescence. Photosynth Res 25: 279–293
Schuldiner S, Rottenberg H and Avron M (1972) Determination of a ΔpH in chloroplasts. 2. Fluorescent amines as a probe for the determination of ΔpH in chloroplasts. Eur J Biochem 25: 64–70
Semin BK, Ivanov II and Rubin AB (1989) Effects of calcium-channel blockers and activators on electron transport in pea chloroplasts. Biochim Biophys Acta 975: 239–243
Styring S, Virgin I, Ehrenberg A and Andersson B (1990) Strong light photoinhibition of electron transport in Photosystem II. Impairment of the function of the first quinone acceptor, QA. Biochim Biophys Acta 1015: 269–278
Thielen APG and van Gorkom HJ (1981) Redox potentials of electron acceptors in Photosystem IIα and IIβ. FEBS Lett 129: 205–209
Vass I, Styring S, Hundal T, Koivuniemi A, Aro E-M and Andersson B (1992) Reversible and irreversible intermediates during photoinhibition of Photosystem II. Stable reduced QA species promote chlorophyll triplet formation. Proc Natl Acad Sci USA 89: 1408–1412
Vernotte C, Etienne A-L and Briantais J-M (1979) Quenching of the Photosystem II chlorophyll fluorescence by the plastoquinone pool. Biochim Biophys Acta 545: 519–527
Walker D (1992) Transley Review No. 36, Excited leaves. New Phytol 121: 325–345
Weis E and Berry JA (1987) Quantum efficiency of Photosystem II in relation to energy-dependent quenching of chlorophyll fluorescence. Biochim Biophys Acta 894: 198–208
Weis E and Lechtenberg D (1989) Fluorescence analysis during steady state photosynthesis. Phil Trans R Soc London B 323: 253–268
van Wijk KJ and van Hasselt PR (1993) Photoinhibition of Photosystem II in vivo is preceded by down-regulation through light-induced acidification of the lumen: Consequences for the mechanism of photoinhibition in vivo. Planta 189: 359–368
Yocum CF (1991) Calcium activation of photosynthetic water oxidation. Biochim Biophys Acta 1059: 1–15
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Krieger, A., Weis, E. The role of calcium in the pH-dependent control of Photosystem II. Photosynth Res 37, 117–130 (1993). https://doi.org/10.1007/BF02187470
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02187470