Abstract
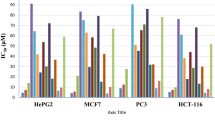
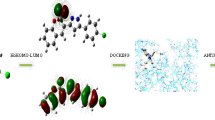
The behavior and reactivity of a coumarin-based chalcone bearing a pyrazole core 3 toward malononitrile, hydrazine, thiourea, hydrazinecarbothioamide, 2-aminoaniline, and 6-aminothiouracil were investigated as an attempt to design and synthesize a novel series of pyrazole-based heterocycles, viz. pyranocoumarin, diazepine, pyrimidochromene, triazepine, benzodiazepine, and pyrimidopyrimidine derivatives. DFT based on quantum chemical computation outlines the structure optimization of the intermediate that reacted to achieve the desired product. The antitumor activity screening against HePG-2 and MCF-7 cancer cell lines disclosed that the most potent compounds against two cell lines were 9 and 17 as compared to doxorubicin which may be attributed to their presence in more tautomeric structures. Also, the minimized energy, dipole moment, ionization potential, transferred electrons, and charge density distribution disclosed that the greater value of 0.126 and 0.8 for pyrazole derivatives 9 and 17, respectively, indicates the maximum transfer of electron and hence greater tendency of scavenging radicals and rapidly reduce oxygen to superoxide.














Similar content being viewed by others
References
B. Orlikova, D. Tasdemir, F. Golais, M. Dicato, M. Diederich, Genes Nutr. 6, 125 (2011)
R. Kenchappa, Y.D. Bodke, A. Chandrashekar, M.A. Sindhe, S.K. Peethambar, Arabian J. Chem. 10, S3895 (2017)
A.D. Singh, A. Kumar, S. Saini, H. Prabha, Asian Pacific. J. Health Sci. 4, 84 (2017)
K.N. Anees Rahman, J. Haribabu, C. Balachandran, N.S.P. Bhuvanesh, R. Ramasamy Karvembu, A. Sreekanth, Polyhedron 135, 26 (2017)
M.M. Abdou, Arab. J. Chem. 10, S3664 (2017)
K.N.M. Halim, S.K. Ramadan, S.A. Rizk, M.A. El-Hashash, Synth. Commun. 50(8), 1159 (2020)
S.K. Ramadan, K.N.M. Halim, S.A. Rizk, M.A. El-Hashash, J. Iran. Chem. Soc. 17, 1575 (2020)
K.N.M. Halim, S.A. Rizk, M.A. El-Hashash, S.K. Ramadan, J. Heterocycl. Chem. 58(2), 636 (2021)
S.K. Ramadan, W.S.I. Abou-Elmagd, Synth. Commun. 48(18), 2409 (2018)
S.K. Ramadan, A.K. El-Ziaty, R.S. Ali, J. Heterocycl. Chem. 58, 290 (2021)
S.K. Ramadan, S.S. Shaban, A.I. Hashem, Synth. Commun. 50(2), 185 (2020)
E.A.E. El-Helw, A.A. El-Badawy, J. Heterocycl. Chem. 57(6), 2354 (2020)
S.K. Ramadan, E.A.E. El-Helw, H.A. Sallam, Heterocycl. Commun. 25(1), 107 (2019)
S.K. Ramadan, H.A. Sallam, J. Heterocycl. Chem. 55(8), 1942 (2018)
W.S.I. Abou-Elmagd, A.K. El-Ziaty, M.I. Elzahar, S.K. Ramadan, A.I. Hashem, Synth. Commun. 46, 1197 (2016)
E.A.E. El-Helw, M.M. Gado, A.K. El-Ziaty, J. Iran. Chem. Soc. 17, 1479 (2020)
S. Maddila, S. Gorle, N. Seshadri, P. Lavanya, S.B. Jonnalagadda, Arab. J. Chem. 9, 681 (2016)
D. Kumar, S.I. Khan, B.L. Tekwani, P. Ponnan, D.S. Rawat, Eur. J. Med. Chem. 89, 490 (2015)
C.M. Bhalgat, M.I. Ali, B. Ramesh, G. Ramu, Arab. J. Chem. 7, 986 (2014)
P.J. Chen, A. Yang, Y.F. Gu, X.S. Zhang, K.P. Shao, D.Q. Xue, P. He, T.F. Jiang, Q.R. Zhang, H.M. Liu, Bioorg. Med. Chem. Lett. 24, 2741 (2014)
S.I. Pretorius, W.J. Breytenbach, C. De. Kock, P.J. Smith, D.D. N’Da, Bioorg. Med. Chem. 21, 269 (2013)
D.G. Powers, D.S. Casebier, D. Fokas, W.J. Ryan, J.R. Troth, D.L. Coffen, Tetrahedron 54, 4085 (1998)
S.A. Rizk, M.A. El-Hashash, K.K. Mostafa, Egypt. J. Chem. 51, 116 (2008)
S.A. Rizk, G. Elsayed, M.A. El-Hashash, J. Iran. Chem. Soc. 15, 2093 (2018)
M.A. El-Hashash, S.A. Rizk, F.A. El-Bassiouny, D.B. Guirguis, S.M. Khairy, L. Guirguis, Egypt J. Chem. 60, 407 (2017)
M.A. El-Hashash, K.M. Darwish, S.A. Rizk, F.A. El-Basiouny, Pharmaceuticals 4(7), 1032 (2011)
A.M. Youssef, A.K. El-Ziaty, W.S.I. Abou-Elmagd, S.K. Ramadan, J. Heterocycl. Chem. 52(1), 278 (2015)
A.R.I. Morsy, S.K. Ramadan, M.M. Elsafty, Med. Chem. Res. 29, 979 (2020)
S.K. Ramadan, E.Z. Elrazaz, K.A.M. Abouzid, A.M. El-Naggar, RSC Adv. 10, 29475 (2020)
A.M. El-Naggar, S.K. Ramadan, Synth. Commun. 50(14), 2188 (2020)
S.K. Ramadan, A.I. Hashem, W.S.I. Abou-Elmagd, A.K. El-Ziaty, J. Heterocycl. Chem. 54(6), 3711 (2017)
S.K. Ramadan, E.A.E. El-Helw, J. Chem. Res. 42, 332 (2018)
N.M. Gad, W.S.I. Abou-Elmagd, D.S.A. Haneen, S.K. Ramadan, Synth. Commun. 51(9), 1384 (2021)
S.K. Ramadan, A.K. El-Ziaty, E.A.E. El-Helw, Synth. Commun. 51(8), 1272 (2021)
E.A.E. El-Helw, A.I. Hashem, Synth. Commun. 50(7), 1046 (2020)
E.A.E. El-Helw, A.R.I. Morsy, A.I. Hashem, J. Heterocycl. Chem. 58(4), 1003 (2021)
A.A. El-Badawy, A.S. Elgubbi, E.A.E. El-Helw, J. Sulfur Chem. 42, 295 (2021)
H.A. Sallam, A.S. Elgubbi, E.A.E. El-Helw, Synth. Commun. 50(13), 2066 (2020)
M.S. Salem, E.A.E. El-Helw, H.A.Y. Derbala, Russ. J. Bioorg. Chem. 46(1), 77 (2020)
E.A.E. El-Helw, H.A. Sallam, A.S. Elgubbi, Synth. Commun. 49(20), 2651 (2019)
S.K. Ramadan, E.A.E. El-Helw, M.E. Azab, Russ. J. Org. Chem. 55(12), 1940 (2019)
P. Rathelot, N. Azas, H. El-Kashef, F. Delmas, C.D. Giorgio, P. Timon-David, J. Maldonado, P. Vanelle, Eur. J. Med. Chem. 37, 671 (2002)
J.-P. Li, J.-K. Qiu, H.-J. Li, G.-S. Zhang, J. Chin. Chem. Soc. 58, 268 (2011)
F. El-Taib Heakal, S.A. Rizk, A.E. Elkholy, J. Mol. Struct. 1152, 328 (2018)
L.E. Chirlian, M.M. Francl, J. Comput. Chem. 8, 894 (1987)
A.E. Elkholy, S.A. Rizk, A.M. Rashad, J. Mol. Struct. 1175, 788 (2019)
R. Dhingra, V. Margulets, S.R. Chowdhury, J. Thliveris, D. Jassal, P. Fernyhough, G.W. Dorn, L.A. Kirshenbaum, Proc. Natl. Acad. Sci. USA 111, E5537 (2014)
B. Kalyanaraman, Redox Biol. 29, 101394 (2020)
X. Fang, H. Wang, D. Han, E. Xie, X. Yang, J. Wei, S. Gu, F. Gao, N. Zhu, X. Yin, Q. Cheng, P. Zhang, W. Dai, Proc. Natl. Acad. Sci. U. S. A. 116, 2672 (2019)
T. Mosmann, J. Immunol. Methods 65, 55 (1983)
F. Denizot, R. Lang, J. Immunol. Methods 22, 271 (1986)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ramadan, S.K., Rizk, S.A. Synthesis, density functional theory, and cytotoxic activity of some heterocyclic systems derived from 3-(3-(1,3-diphenyl-1H-pyrazol-4-yl)acryloyl)-2H-chromen-2-one. J IRAN CHEM SOC 19, 187–201 (2022). https://doi.org/10.1007/s13738-021-02298-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-021-02298-6