Abstract
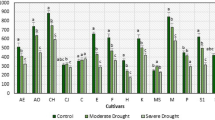
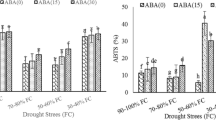
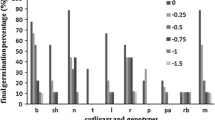
Differences in abscisic acid (ABA) accumulation between two olive cultivars were studied by enzyme-linked immunosorbent assay in roots and leaves, leaf water potential (Ψl), stomatal conductance (g s) as well as photosynthetic rate (A) were also determined in well-watered (WW) and water-stressed (WS) plants of two olive cultivars ‘Chemlali’ and ‘Chetoui’. ‘Chemlali’ was able to maintain higher leaf CO2 assimilation rate and leaf stomatal conductance throughout the drought cycle when compared with ‘Chetoui’. Furthermore, leaf water potential of ‘Chemlali’ decreased in lower extent than in Chetoui in response to water deficit. Interestingly, significant differences in water-stress-induced ABA accumulation were observed between the two olive cultivars and reflect the degree of stress experienced. Chemlali, a drought tolerant cultivar, accumulated lower levels of ABA in their leaves to regulate stomatal control in response to water stress compared to the drought sensitive olive cultivar ‘Chetoui’ which accumulated ABA in large amount.








Similar content being viewed by others
References
Allen GJ, Chu SP, Harrington CL, Schumacher K, Hoffmann T, Tang YY, Grill E, Schroeder JI (2001) A defined range of guard cell calcium oscillation parameters encodes stomatal movements. Nature 411:1053–1057. doi:10.1038/35082575
Bacelar EA, Santos DL, Moutinho-Pereira JM, Lopes JI, Gonçalves BC, Ferreira TC, Correia CM (2007) Physiological behaviour, oxidative damage and antioxidative protection of olive trees grown under different irrigation regimes. Plant Soil 292:1–12. doi:10.1007/s11104-006-9088-1
Bacon MA, Wilkinson S, Davies WJ (1998) pH-regulated leaf cell expansion in droughted plants is abscisic acid dependent. Plant Physiol 118:1507–1515. doi:10.1104/pp.118.4.1507
Ben Temime S, Campeol E, Luigi Cioni P, Daoud D, Zarrouk M (2006) Volatile compounds from Chétoui olive oil and variations induced by growing area. Food Chem 99:315–325. doi:10.1016/j.foodchem.2005.07.046
Boujnah D (1997) Variations morphologiques anatomiques et écophysiologiques en rapport avec la résistance à la sécheresse chez l’olivier (Olea europaea L.). PhD Thesis, University of Gent, Belgium
Chartzoulakis K, Patakas A, Bosabalidis AM (1999) Changes in water relations, photosynthesis and leaf anatomy induced by intermittent drought in two olive cultivars. Environ Exp Bot 42:113–120. doi:10.1016/S0098-8472(99)00024-6
Chartzoulakis K, Loupasaki M, Bertaki M, Androulakis I (2002) Effects of NaCl salinity on growth, ion content and CO2 assimilation rate of six olive cultivars. Sci Hortic (Amsterdam) 96:235–247. doi:10.1016/S0304-4238(02)00067-5
Chaves MM, Moraco JP, Pereira JS (2003) Understanding plant responses to drought from genes to whole plant. Funct Plant Biol 30:239–264. doi:10.1071/FP02076
Cornic G (2000) Drought stress inhibits photosynthesis by decreasing stomatal aperture—not by affecting ATP synthesis. Trends Plant Sci 5:187–188. doi:10.1016/S1360-1385(00)01625-3
Davies WJ, Zhang J (1991) Root signals and the regulation of growth and development of plants in drying soil. Ann Rev Plant Physiol Plant Mol Biol 42:55–76. doi:10.1146/annurev.pp.42.060191.000415
Davies WJ, Mansfield TA, Hetherington AM (1990) Sensing of soil water status and the regulation of plant growth and development. Plant Cell Environ 13:709–719. doi:10.1111/j.1365-3040.1990.tb01085.x
Desikan R, Cheung MK, Bright J, Henson D, Hancock JT, Neill SJ (2004) ABA, hydrogen peroxide and nitric oxide signalling in stomatal guard cells. J Exp Bot 55:205–212. doi:10.1093/jxb/erh033
Fernández JE, Moreno F, Giron IF, Blazquez OM (1997) Stomatal control of water use in olive tree leaves. Plant Soil 190:179–192. doi:10.1023/A:1004293026973
Flexas J, Escalona JM, Medrano H (1999) Water stress induces different levels of photosynthesis and electron transport rate regulation in grapevines. Plant Cell Environ 22:39–48. doi:10.1046/j.1365-3040.1999.00371.x
García-Mata C, Lamattina L (2002) Nitric oxide and abscisic acid cross talk in guard cells. Plant Physiol 128:790–792. doi:10.1104/pp.011020
Giorio P, Sorrentino G, D’andria R (1999) Stomatal behavior, leaf water status and photosynthetic response in field-grown olive trees under water deficit. Environ Exp Bot 42:95–104. doi:10.1016/S0098-8472(99)00023-4
Gowing DJG, Jones HG, Davies WJ (1993) Xylem-transported abscisic acid: the relative importance of its mass and its concentration in the control of stomatal aperture. Plant Cell Environ 16:453–459. doi:10.1111/j.1365-3040.1993.tb00892.x
Guerfel M, Baccouri B, Boujnah D, Zarrouk M (2007) Seasonal changes in water relations and gas exhange in leaves of two Tunisian olive (Olea europaea L.) cultivars under water deficit. J Hortic Sci Biotechnol 82:721–726
Hartung W, Sauter A, Hose E (2002) Abscisic acid in the xylem: where does it come from, where does it go to? J Exp Bot 53:27–32. doi:10.1093/jexbot/53.366.27
Jackson GE, Irvine J, Grace J, Khalil AAM (1995) Abscisic acid concentrations and fluxes in droughted conifer saplings. Plant Cell Environ 18:13–22. doi:10.1111/j.1365-3040.1995.tb00539.x
Jarvis AJ, Davies WJ (1997) Whole-plant ABA flux and the regulation of water loss in Cedrella odorata. Plant Cell Environ 20:521–527. doi:10.1046/j.1365-3040.1997.d01-90.x
Jarvis AJ, Mansfield TA, Davies WJ (1999) Stomatal behaviour, photosynthesis and transpiration under rising CO2. Plant Cell Environ 22:639–648. doi:10.1046/j.1365-3040.1999.00407.x
Jia W, Zhang J (1999) Stomatal closure is induced rather by prevailing xylem abscisic acid than by accumulated amount of xylem-derived abscisic acid. Physiol Plant 106:268–275. doi:10.1034/j.1399-3054.1999.106303.x
Kwak JM, Moon J-H, Murata Y, Kuchitsu K, Leonhardt N, DeLong A, Schroeder JI (2002) Disruption of a guard cell-expressed protein phosphatase 2A regulatory subunit, RCN1, confers abscisic acid insensitivity in Arabidopsis. Plant Cell 14:2849–2861. doi:10.1105/tpc.003335
Laouar S (1977) Caractéristiques écophysiologiques et aspects de l’économie de l’eau de l’olivier et de l’oranger. Thèse de Doctorat d’Etat en Sciences. Université Paris VII, p 247
Liu F, Jensen CR, Shahanzari A, Andersen MN, Jacobsen SE (2005) ABA regulated stomatal control and photosynthetic water use efficiency of potato (Solanum tuberosum L.) during progressive soil drying. Plant Sci 168:831–836. doi:10.1016/j.plantsci.2004.10.016
Luan S (2002) Signalling drought in guard cells. Plant Cell Environ 25:229–237. doi:10.1046/j.1365-3040.2002.00758.x
Osório ML, Breia E, Rodrigues A, Osório J, Le Rouxc X, Daudetd FA, Ferreira I, Chaves MM (2006) Limitations to carbon assimilation by mild drought in nectarine trees growing under field conditions. Environ Exp Bot 55:235–247. doi:10.1016/j.envexpbot.2004.11.003
Schroeder JI, Kwak JM, Allen GJ (2001) Guard cell abscisic acid signaling and engineering of drought hardiness in plants. Nature 410:327–330. doi:10.1038/35066500
Shinozaki K, Yamaguchi-Shinozak K (2007) Gene networks involved in drought stress response and tolerance. J Exp Bot 58:221–227. doi:10.1093/jxb/erl164
Sofo A, Manfreda S, Dichio B, Fiorentino M, Xiloyannis C (2007) The olive tree: a paradigm for drought tolerance in Mediterranean climates. Hydrol Earth Syst Sci Discuss 4:2811–2835
Tattini M, Traversi ML (2009) On the mechanism of salt tolerance in olive (Olea europaea L.) under low- or high-Ca2+ supply. Environ Exp Bot 65:72–81. doi:10.1016/j.envexpbot.2008.01.005
Tognetti R, d’Andria R, Morelli G, Calandrelli D, Fragnito F (2004) Irrigation effects on daily and seasonal variations of trunk sap flow and leaf water relations in olive trees. Plant Soil 263:249–264. doi:10.1023/B:PLSO.0000047738.96931.91
Tognetti R, d’Andria R, Lavini A, Morelli G (2006) The effect of deficit irrigation on crop yield and vegetative development of Olea europaea L. (cvs. Frantoio and Leccino). Eur J Agron 25:356–364. doi:10.1016/j.eja.2006.07.003
Trejo CL, Clephan A, Davies WJ (1995) How do stomata read Abscisic acid signal? Plant Physiol 109:803–811
Villalobos FJ, Morian A, Fereres E (2002) Stomatal and photosynthesis responses of olive (Olea europaea L.) leaves to water deficits. Plant Cell Environ 25:387–395
Zhang J, Davies WJ (1989a) Abscisic acid produced in dehydrating roots may enable the plant to measure the water status of the soil. Plant Cell Environ 12:73–81. doi:10.1111/j.1365-3040.1989.tb01918.x
Zhang J, Davies WJ (1989b) Sequential responses of whole plant water relations towards prolonged soil drying and the mediation by xylem sap ABA concentrations in the regulation of stomatal behaviour of sunflower plants. New Phytol 113:167–174. doi:10.1111/j.1469-8137.1989.tb04703.x
Zhang J, Davies WJ (1990a) Changes in the concentration of ABA in xylem sap as a function of changing soil water status will account for changes in leaf conductance. Plant Cell Environ 13:277–285. doi:10.1111/j.1365-3040.1990.tb01312.x
Zhang J, Davies WJ (1990b) Does ABA in the xylem control the rate of leaf growth in soil-dried maize and sunflower plants. J Exp Bot 41:1125–1132. doi:10.1093/jxb/41.9.1125
Zhang J, Davies WJ (1991) Antitranspiration activity in xylem sap of maize plants. J Exp Bot 42:317–321. doi:10.1093/jxb/42.3.317
Zhang J, Jia W, Yang J, Ismail AM (2006) Role of ABA in integrating plant responses to drought and salt stresses. Field Crops Res 97:111–119. doi:10.1016/j.fcr.2005.08.018
Zhu J-K (2002) Salt and drought stress signal transduction in plants. Annu Rev Plant Physiol Plant Mol Biol 53:247–273. doi:10.1146/annurev.arplant.53.091401.143329
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by W. Filek.
Rights and permissions
About this article
Cite this article
Guerfel, M., Beis, A., Zotos, T. et al. Differences in abscisic acid concentration in roots and leaves of two young Olive (Olea europaea L.) cultivars in response to water deficit. Acta Physiol Plant 31, 825–831 (2009). https://doi.org/10.1007/s11738-009-0298-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11738-009-0298-z