Abstract
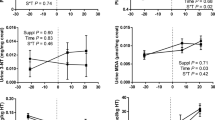
This study aimed to analyze the possible relationship between days to conception and different oxidative stress (OS) biomarkers and liver functional parameters in multiparous dairy cows. Besides, a fast reliable method for the accurate measurement of malondialdehyde (MDA) by liquid chromatography–tandem mass spectrometry was developed in several matrices. During lactation, the days to conception of 28 cows were determined for a retrospective study. According to this parameter, cows were divided into two groups: high and low days to conception (HDC and LDC, respectively). Blood, urine and liver biopsies were sampled 21 days before the expected calving date, and 7 and 21 days after calving. The method developed for MDA was validated according to international requirements. The lower limit of quantification was 0.25 µmol/L for plasma and urine and 10.00 µmol/L for liver tissue. No differences between groups were observed in the systemic concentration of non-esterified fatty acids, β-hydroxybutyric acid and liver triacylglycerol content (P > 0.05). Cholesterol concentration was higher in the LDC than in the HDC group (P < 0.05). Plasma 3-nitrotyrosine (3-NT) concentration was lower in the LDC than in the HDC group on day 21 post-calving (P < 0.05). Superoxide dismutase activity was higher in the LDC than in the HDC group (P < 0.05). Particularly, in the liver, 3-NT and MDA concentrations were lower in the LDC than in the HDC group (P < 0.05). These results allow inferring that the amelioration of OS biomarkers in plasma and liver could be related to a better reproductive performance of dairy cows.



Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
Abuelo A, Hernández J, Benedito JL, Castillo C (2015) The importance of the oxidative status of dairy cattle in the periparturient period: Revisiting antioxidant supplementation. J Anim Physiol Anim Nutr (berl) 99:1003–1016. https://doi.org/10.1111/jpn.12273
Akyuz E, Akyüz E, Kükürt A et al (2022) Evaluation of total sialic acid, paraoxonase activity and malondialdehyde in cows with subclinical paratuberculosis. J Hell Vet Med Soc 73:4283–4288
Angeli E, Rodríguez FM, Rey F et al (2019) Liver fatty acid metabolism associations with reproductive performance of dairy cattle. Anim Reprod Sci 208:106104. https://doi.org/10.1016/j.anireprosci.2019.06.016
Angeli E, Trionfini V, Gareis NC et al (2019b) Protein and gene expression of relevant enzymes and nuclear receptor of hepatic lipid metabolism in grazing dairy cattle during the transition period. Res Vet Sci 123:223–231. https://doi.org/10.1016/j.rvsc.2019.01.020
Angeli E, Barcarolo D, Durante L et al (2021) Effect of precalving body condition score on insulin signaling and hepatic inflammatory state in grazing dairy cattle. Domest Anim Endocrinol 76:106621. https://doi.org/10.1016/j.domaniend.2021.106621
Azizi S, Soleymani J, Khoubnasabjafari M et al (2017) Liquid chromatographic determination of malondialdehyde in plasma samples after liquid–liquid microextraction. Curr Anal Chem 14:416–422. https://doi.org/10.2174/1573411013666170703162443
Bandookwala M, Thakkar D, Sengupta P (2020) Advancements in the analytical quantification of nitroxidative stress biomarker 3-nitrotyrosine in biological matrices. Crit Rev Anal Chem 50:265–289. https://doi.org/10.1080/10408347.2019.1623010
Barcarolo D, Angeli E, Ribas LE et al (2022) Application of an optimized and validated LC–MS/MS method for the quantification of free 3-nitrotyrosine in plasma, urine and liver tissue of lactating dairy cows. Livest Sci 257:104852. https://doi.org/10.1016/j.livsci.2022.104852
Bello NM, Stevenson JS, Tempelman RJ (2012) Invited review: Milk production and reproductive performance: Modern interdisciplinary insights into an enduring axiom. J Dairy Sci 95:5461–5475. https://doi.org/10.3168/JDS.2012-5564
Berge AC, Vertenten G (2014) A field study to determine the prevalence, dairy herd management systems, and fresh cow clinical conditions associated with ketosis in western European dairy herds. J Dairy Sci 97:2145–2154. https://doi.org/10.3168/jds.2013-7163
Bertoni G, Trevisi E (2013) Use of the liver activity index and other metabolic variables in the assessment of metabolic health in dairy herds. Vet Clin North Am - Food Anim Pract 29:413–431. https://doi.org/10.1016/j.cvfa.2013.04.004
Bertoni G, Trevisi E, Han X, Bionaz M (2008) Effects of inflammatory conditions on liver activity in puerperium period and consequences for performance in dairy cows. J Dairy Sci 91:3300–3310. https://doi.org/10.3168/jds.2008-0995
Bertoni G, Trevisi E, Lombardelli R (2009) Some new aspects of nutrition, health conditions and fertility of intensively reared dairy cows. Ital J Anim Sci 8:491–518. https://doi.org/10.4081/ijas.2009.491
Bicalho MLS, Lima FS, Ganda EK et al (2014) Effect of trace mineral supplementation on selected minerals, energy metabolites, oxidative stress, and immune parameters and its association with uterine diseases in dairy cattle. J Dairy Sci 97:4281–4295. https://doi.org/10.3168/jds.2013-7832
Bioanalytical method validation. Guidance for industry (2018) United States Department of Health and Human Services. Food and Drug Administration (FDA), 1–41.
Bobe G, Young JW, Beitz DC (2004) Invited review: pathology, etiology, prevention, and treatment of fatty liver in dairy cows. J Dairy Sci 87:3105–3124. https://doi.org/10.3168/jds.S0022-0302(04)73446-3
Bogado Pascottini O, Probo M, LeBlanc SJ et al (2020) Assessment of associations between transition diseases and reproductive performance of dairy cows using survival analysis and decision tree algorithms. Prev Vet Med 176:104908. https://doi.org/10.1016/j.prevetmed.2020.104908
Bradford BJ, Yuan K, Farney JK et al (2015) Invited review: Inflammation during the transition to lactation: New adventures with an old flame. J Dairy Sci 98:6631–6650. https://doi.org/10.3168/jds.2015-9683
Buckley WT, Eigendorf GK, Dorward WJ (1986) A liver biopsy instrument for large animals. Can J Anim Sci 66:1137–1140. https://doi.org/10.4141/cjas86-125
Celi P, Merlo M, Barbato O, Gabai G (2012) Relationship between oxidative stress and the success of artificial insemination in dairy cows in a pasture-based system. Vet J 193:498–502. https://doi.org/10.1016/j.tvjl.2012.02.002
Chacha F, Gherissi DE, Lamraoui R et al (2022) Evaluation of body condition, daily milk production and biochemical parameters during the postpartum period according to calving season in Montbeliard dairy cows reared in the semi-arid region - Algeria. Vet Stanica 53:677–687. https://doi.org/10.46419/VS.53.6.3
Czauderna M, Kowalczyk J, Marounek M (2011) The simple and sensitive measurement of malondialdehyde in selected specimens of biological origin and some feed by reversed phase high performance liquid chromatography. J Chromatogr B Anal Technol Biomed Life Sci 879:2251–2258. https://doi.org/10.1016/j.jchromb.2011.06.008
De Vries A (2020) Symposium review: Why revisit dairy cattle productive lifespan? J Dairy Sci 103:3838–3845. https://doi.org/10.3168/jds.2019-17361
De Koster JD, Opsomer G (2013) Insulin resistance in dairy cows. Vet Clin North Am - Food Anim Pract 29:299–322. https://doi.org/10.1016/j.cvfa.2013.04.002
De Bie J, Langbeen A, Verlaet AAJ et al (2016) The effect of a negative energy balance status on β-carotene availability in serum and follicular fluid of nonlactating dairy cows. J Dairy Sci 99:5808–5819. https://doi.org/10.3168/jds.2016-10870
Dirandeh E, Sayyar MA, Ansari-Pirsaraei Z et al (2021) Peripheral leucocyte molecular indicators of inflammation and oxidative stress are altered in dairy cows with embryonic loss. Sci Rep 11:12771. https://doi.org/10.1038/s41598-021-91535-2
Đuričić D, Ljubić BB, Vince S et al (2020) Effects of dietary clinoptilolite supplementation on β-hydroxybutirate serum level and milk fat to protein ratio during early lactation in Holstein-Friesian cows. Microporous Mesoporous Mater 292:109766. https://doi.org/10.1016/J.MICROMESO.2019.109766
Edmonson AJ, Lean IJ, Weaver LD et al (1989) A body condition scoring chart for Holstein dairy cows. J Dairy Sci 72:68–78. https://doi.org/10.3168/jds.S0022-0302(89)79081-0
Esposito G, Irons PC, Webb EC, Chapwanya A (2014) Interactions between negative energy balance, metabolic diseases, uterine health and immune response in transition dairy cows. Anim Reprod Sci 144:60–71. https://doi.org/10.1016/j.anireprosci.2013.11.007
Esra Colakoglu H, Yazlik MO, Kaya U et al (2017) G MDA and GSH-Px activity in transition dairy cows under seasonal variations and their relationship with reproductive performance. J Vet Res 61:497–502. https://doi.org/10.1515/jvetres-2017-0067
Faul F, Erdfelder E, Lang A-G, Buchner A (2007) G*Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods 39:175–191. https://doi.org/10.3758/BF03193146
Folch J, Lees M, Stanley GHS (1957) A simple method for the isolation and purification of total lipids from animal tissues. J Biol Chem 226:497–509
Folnožić I, Turk R, Duričić D et al (2015) Influence of body condition on serum metabolic indicators of lipid mobilization and oxidative stress in dairy cows during the transition period. Reprod Domest Anim 50:910–917. https://doi.org/10.1111/RDA.12608
Folnožić I, Samardžija M, Đuričić D et al (2019) Effects of in-feed clinoptilolite treatment on serum metabolic and antioxidative biomarkers and acute phase response in dairy cows during pregnancy and early lactation. Res Vet Sci 127:57–64. https://doi.org/10.1016/J.RVSC.2019.10.010
Gareis NC, Angeli E, Huber E et al (2018) Alterations in key metabolic sensors involved in bovine cystic ovarian disease. Theriogenology 120:138–146. https://doi.org/10.1016/j.theriogenology.2018.07.045
Garverick HA, Harris MN, Vogel-Bluel R et al (2013) Concentrations of nonesterified fatty acids and glucose in blood of periparturient dairy cows are indicative of pregnancy success at first insemination. J Dairy Sci 96:181–188. https://doi.org/10.3168/jds.2012-5619
Giordano JO, Fricke PM, Wiltbank MC, Cabrera VE (2011) An economic decision-making support system for selection of reproductive management programs on dairy farms. J Dairy Sci 94:6216–6232. https://doi.org/10.3168/jds.2011-4376
Ho E, Karimi Galougahi K, Liu C-C et al (2013) Biological markers of oxidative stress: Applications to cardiovascular research and practice. Redox Biol 1:483–491. https://doi.org/10.1016/j.redox.2013.07.006
Houghton R, Horro Pita C, Ward I, Macarthur R (2009) Generic approach to validation of small-molecule LC-MS/MS biomarker assays. Bioanalysis 1:1365–1374. https://doi.org/10.4155/bio.09.139
LeBlanc S (2010) Monitoring metabolic health of dairy cattle in the transition period. J Reprod Dev 56(Suppl):S29-35. https://doi.org/10.1262/jrd.1056S29
LeBlanc SJ, Duffield TF, Leslie KE et al (2002) The effect of prepartum injection of vitamin E on health in transition dairy cows. J Dairy Sci 85:1416–1426. https://doi.org/10.3168/jds.S0022-0302(02)74209-4
LeBlanc SJ, Leslie KE, Duffield TF (2005) Metabolic predictors of displaced abomasum in dairy cattle. J Dairy Sci 88:159–170. https://doi.org/10.3168/jds.S0022-0302(05)72674-6
Li X, Li S, Kellermann G (2015) A novel mixed-mode solid phase extraction coupled with LC-MS/MS for the re-evaluation of free 3-nitrotyrosine in human plasma as an oxidative stress biomarker. Talanta 140:45–51. https://doi.org/10.1016/j.talanta.2015.02.053
Loor JJ, Dann HM, Guretzky NAJ et al (2006) Plane of nutrition prepartum alters hepatic gene expression and function in dairy cows as assessed by longitudinal transcript and metabolic profiling. Physiol Genomics 27:29–41. https://doi.org/10.1152/physiolgenomics.00036.2006
Loor JJ, Everts RE, Bionaz M et al (2007) Nutrition-induced ketosis alters metabolic and signaling gene networks in liver of periparturient dairy cows. Physiol Genomics 32:105–116. https://doi.org/10.1152/physiolgenomics.00188.2007
López-Gatius F, García-Ispierto I, Santolaria P et al (2006) Screening for high fertility in high-producing dairy cows. Theriogenology 65:1678–1689. https://doi.org/10.1016/j.theriogenology.2005.09.027
Lucy MC (2007) Fertility in high-producing dairy cows: reasons for decline and corrective strategies for sustainable improvement. Soc Reprod Fertil Suppl 64:237–254
Lucy MC, Butler ST, Garverick HA (2014) Endocrine and metabolic mechanisms linking postpartum glucose with early embryonic and foetal development in dairy cows. Animal 8(Suppl 1):82–90. https://doi.org/10.1017/S1751731114000482
Masarone M, Rosato V, Dallio M et al (2018) Role of oxidative stress in pathophysiology of nonalcoholic fatty liver disease. Oxid Med Cell Longev 2018:1–14. https://doi.org/10.1155/2018/9547613
Mee JF (2007) The role of the veterinarian in bovine fertility management on modern dairy farms. Theriogenology 68(Suppl 1):S257–S265. https://doi.org/10.1016/j.theriogenology.2007.04.030
Mikulková K, Kadek R, Filípek J, Illek J (2020) Evaluation of oxidant/antioxidant status, metabolic profile and milk production in cows with metritis. Ir Vet J 73:8. https://doi.org/10.1186/S13620-020-00161-3
Moreno-González D, Alcántara-Durán J, Addona SM, Beneito-Cambra M (2018) Multi-residue pesticide analysis in virgin olive oil by nanoflow liquid chromatography high resolution mass spectrometry. J Chromatogr A 1562:27–35. https://doi.org/10.1016/j.chroma.2018.05.053
Olynk NJ, Wolf CA (2008) Economic analysis of reproductive management strategies on US commercial dairy farms. J Dairy Sci 91:4082–4091. https://doi.org/10.3168/jds.2007-0858
Ospina PA, Nydam DV, Stokol T, Overton TR (2010) Associations of elevated nonesterified fatty acids and β-hydroxybutyrate concentrations with early lactation reproductive performance and milk production in transition dairy cattle in the northeastern United States. J Dairy Sci 93:1596–1603. https://doi.org/10.3168/jds.2009-2852
Patton J, Kenny DA, McNamara S et al (2007) Relationships among milk production, energy balance, plasma analytes, and reproduction in Holstein-Friesian cows. J Dairy Sci 90:649–658. https://doi.org/10.3168/jds.S0022-0302(07)71547-3
Pilz J, Meineke I, Gleiter CH (2000) Measurement of free and bound malondialdehyde in plasma by high-performance liquid chromatography as the 2,4-dinitrophenylhydrazine derivative. J Chromatogr B Biomed Sci Appl 742:315–325. https://doi.org/10.1016/S0378-4347(00)00174-2
Ponnampalam EN, Kiani A, Santhiravel S et al (2022) The importance of dietary antioxidants on oxidative stress, meat and milk production, and their preservative aspects in farm animals: antioxidant action, animal health, and product quality-invited review. Animals 12(23):3279. https://doi.org/10.3390/ani12233279
Radabaugh MR, Nemirovskiy OV, Misko TP et al (2008) Immunoaffinity liquid chromatography-tandem mass spectrometry detection of nitrotyrosine in biological fluids: Development of a clinically translatable biomarker. Anal Biochem 380:68–76. https://doi.org/10.1016/j.ab.2008.05.019
Rivera H, Lopez H, Fricke PM (2005) Use of intravaginal progesterone-releasing inserts in a synchronization protocol before timed AI and for synchronizing return to estrus in Holstein heifers. J Dairy Sci 88:957–968. https://doi.org/10.3168/jds.S0022-0302(05)72763-6
Roche JF (2006) The effect of nutritional management of the dairy cow on reproductive efficiency. Anim Reprod Sci 96:282–296. https://doi.org/10.1016/j.anireprosci.2006.08.007
Rutherford AJ, Oikonomou G, Smith RF (2016) The effect of subclinical ketosis on activity at estrus and reproductive performance in dairy cattle. J Dairy Sci 99:4808–4815. https://doi.org/10.3168/jds.2015-10154
Sayiner S, Darbaz I, Ergene O, Aslan S (2021) Changes in antioxidant enzyme activities and metabolic parameters in dairy cows during different reproductive periods. Theriogenology 159:116–122. https://doi.org/10.1016/j.theriogenology.2020.10.024
Sordillo LM, Aitken SL (2009) Impact of oxidative stress on the health and immune function of dairy cattle. Vet Immunol Immunopathol 128:104–109. https://doi.org/10.1016/j.vetimm.2008.10.305
Sordillo LM, Mavangira V (2014) The nexus between nutrient metabolism, oxidative stress and inflammation in transition cows. Anim Prod Sci 54:1204–1214. https://doi.org/10.1071/AN14503
Surai PF, Kochish II, Fisinin VI, Juniper DT (2019) Revisiting oxidative stress and the use of organic selenium in dairy cow nutrition. Animals 9(7):462. https://doi.org/10.3390/ani9070462
Talukder S, Kerrisk KL, Gabai G, Celi P (2017) Role of oxidant–antioxidant balance in reproduction of domestic animals. Anim Prod Sci 57:1588. https://doi.org/10.1071/AN15619
van Dorland HA, Richter S, Morel I et al (2009) Variation in hepatic regulation of metabolism during the dry period and in early lactation in dairy cows. J Dairy Sci 92:1924–1940. https://doi.org/10.3168/jds.2008-1454
Venjakob PL, Pieper L, Heuwieser W, Borchardt S (2018) Association of postpartum hypocalcemia with early-lactation milk yield, reproductive performance, and culling in dairy cows. J Dairy Sci 101:9396–9405. https://doi.org/10.3168/jds.2017-14202
Acknowledgements
We thank Med. Vet. Ernesto Quercia for his professional assistance and Biogenesis Bago S.A. for providing products for this project. We also thank the staff members of the Laboratorio de Biología Celular y Molecular Aplicada, Instituto de Ciencias Veterinarias del Litoral (ICIVET Litoral), Consejo Nacional de Investigaciones Científicas y Tecnológicas (CONICET)—Universidad Nacional del Litoral (UNL), Santa Fe, Argentina.
Funding
This study was supported by grants from the Argentine National Agency for the Promotion of Science and Technology (ANPCyT; PICT 2016–0456), from CONICET (PIP 11220210100067CO) and from Universidad Nacional del Litoral (CAI + D 50020150100079LI), Santa Fe, Argentina. VM, FR, HHO and GJH are research career members and EA, DB and LER are fellows of the National Scientific Research Council (CONICET, Argentina).
Author information
Authors and Affiliations
Contributions
Conceptualization: EA, DB, LER and SMA; Methodology: EA, DB, LER and SM; Formal analysis and investigation: EA, VM and FR; Writing—review and editing: EA, DB, LER, SMA, HHO and GJH; Funding acquisition: HHO and GJH; Resources: HHO and GJH; Supervision: GJH.
Corresponding author
Ethics declarations
Ethics approval
The procedures performed in this study were approved by the Ethics Committee of the Faculty of Veterinary Sciences of the Universidad Nacional del Litoral (Protocol number FCV-1055850–21, Santa Fe, Argentina) and were in agreement with the Guide for the Care and Use of Agricultural Animals in Research and Teaching (Federation of Animal Science Societies, 2010).
Consent to participate
All authors participated voluntarily in the research.
Consent to publish
All authors agree to the content of the paper for publication.
Competing interests
The authors declare no competing interests.
Conflicting interests
The authors declare no conflicts of interest with respect to the research, authorship, and/or publication of this article.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Angeli, E., Barcarolo, D., Ribas, L.E. et al. Biomarkers of oxidative stress and liver function in early lactation and their relationship with the reproductive efficiency of multiparous grazing dairy cows in Argentina. A retrospective study. Vet Res Commun 47, 1817–1830 (2023). https://doi.org/10.1007/s11259-023-10134-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11259-023-10134-w