Abstract
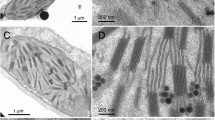
The structural reorganization of pea thylakoid systems in response to osmotic shock in a wide range of temperatures (36–70°C) was studied. At temperatures 40–46°C, the configuration of thylakoid systems changed from a flattened to a nearly round, whereas thylakoids themselves remained compressed. The percentage of thylakoids stacked into grana at 44°C decreased from 71 % in the control to 40 % in experimental samples, reaching 59 % at 48°C. At 44°C and above, thylakoid systems ceased to respond to the osmotic shock by disordering, in contrast to what happened at lower temperatures (36–43°C) and in the control, and retained the configuration inherent in thylakoid systems at these temperatures. At 50°C and above, the packing of thylakoids in grana systems changed, and thylakoids formed extended strands of pseudograna. Simultaneously, single thylakoids formed a network of anastomoses through local fusions. At temperatures of 60–70°C, thylakoid systems appeared as spherical clusters of membrane vesicles with different degree of separation.
Similar content being viewed by others
References
Armond, P.A., Björkman, O., Staehelin, L.A.: Dissociation of supramolecular complexes in chloroplast membranes. A manifestation of heat damage to the photosynthetic apparatus.–Biochim. biophys. Acta 601: 433–442, 1980.
Berry, J., Björkman, O.: Photosynthetic response and adaptation to temperature in higher plants.–Annu. Rev. Plant Physiol. 31: 491–543, 1980.
Bilger, W., Schreiber, U.: Chlorophyll luminescence as an indicator of stress-induced damage to the photosynthetic apparatus. Effects of heat stress in isolated chloroplasts.–Photosynth. Res. 25: 161–171, 1990.
Cao, J., Govindjee: Chlorophyll a fluorescence transient as an indicator of active and inactive Photosystem II in thylakoid membranes.–Biochim. biophys. Acta 1015: 180–188, 1990.
Cramer, W.A., Whitmarsh, J., Low, P.S.: Differential scanning calorimetry of chloroplast membranes: identification of an endothermic transition associated with the water-splitting complex of photosystem II.–Biochemistry 20: 157–162, 1981.
Gounaris, K., Brain, A.P.R., Quinn, P.J., Williams, W.P.: Structural and functional changes associated with heat-induced phase separations of non-bilayer lipids in chloroplast thylakoid membranes.–FEBS Lett. 153: 47–52, 1983.
Gounaris, K., Brain, A.P.R., Quinn, P.J., Williams, W.P.: Structural reorganisation of chloroplast thylakoid membranes in response to heat stress.–Biochim. biophys. Acta 766: 198–208, 1984.
Ignat’ev, A.P., Khorobrykh, S.A., Ivanov, B.N.: [Effect of high concentrations of magnesium ions on the rate of electron transport and proton exchange in thylakoid membranes of higher plants.]–Biofizika 46: 1075–1080, 2001. [In Russ.]
Kreslavskiï, V.D., Khristin, M.S.: [After-effect of heat shock on fluorescence induction and low-temperature fluorescence spectra in wheat leaves.]–Biofizika 45: 865–872, 2003. [In Russ.]
Lyutova, M.I., Tikhonov, A.N.: [After-effect of high temperature on photosynthesis and electron transport in wheat leaves.]–Biofizika 28: 284–287, 1983. [In Russ.]
Matos, M.C., Campos, P.S., Ramalho, J.C., Medeira, M.C., Maia, M.I., Semedo, J.M., Marques, N.M., Matos, A.: Photosynthetic activity and cellular integrity of the Andean legume Pachyrhizus ahipa (Wedd.) Parodi under heat and water stress.–Photosynthetica 40: 493–501, 2002.
Molotkovsky, Yu.G., Zheskova, I.M.: The influence of heating on the morphology and photochemical activity of isolated chloroplasts.–Biochem. biophys. Res. Commun. 20: 411–415, 1965.
Semenova, G.A.: The relationship between the transformation of thylakoid acyl lipids and the formation of tubular lipid aggregates visible on fracture faces.–J. Plant Physiol. 155: 669–677, 1999.
Semenova, G.A.: Effect of urea and distilled water on the structure of the thylakoid system.–J. Plant Physiol. 158: 1041–1050, 2001.
Semenova, G.A.: The thylakoid membrane in a wide pH range.–J. Plant Physiol. 159: 613–625, 2002.
Semenova, G.A., Agafonov, A.V., Opanasenko, V.K.: Light-induced reversible local fusions of thylakoid membranes in the presence of dibucaine or tetracaine.–Biochim. biophys. Acta 1285: 29–37, 1996.
Semenova, G.A., Khorobrykh, A.A.: [Structure, functional activity, and lipid composition of pea thylakoid systems during storage at–15 °C.]–Biol. Membr. 18: 259–264, 2001. [In Russ.]
Semenova, G.A., Vasilenko, I., Borovyagin, V.: Structural changes in thylakoid membranes of chilling-resistant and sensitive plants after heating and glycerol dehydration as revealed by 31P NMR and electron microscopy.–Biophys. Chem. 49: 59–69, 1994.
Shutilova, N.I.: [On the mechanism of photosynthetic oxidation of water in a dimeric oxygen-evolving complex of chloroplast photosystem II.]–Biofizika 45: 51–57, 2000. [In Russ.]
Shutilova, N.I., Klimov, V.V., Antropova, T.M., Shnyrov, V.L.: [On the mechanism of thermoinactivation of the oxygen-evolving photosystem II subchloroplast core complex.]–Biokhimiya 57: 1508–1518, 1992. [In Russ.]
Staehelin, L.A.: Chloroplast structure and supramolecular organization of photosynthetic membranes.–In: Staehelin, L.A., Arntzen, C.J. (ed.): Photosynthesis III. Pp. 1–84. Springer-Verlag, Berlin–Heidelberg–New York–Tokyo 1986.
Sundby, C., Melis, A., Mäenpää, P., Andersson, B.: Temperature-dependent changes in the antenna size of Photosystem II: Reversible conversion of Photosystem IIα to Photosystem IIβ.–Biochim. biophys. Acta 851: 475–483, 1986.
Thomas, P.G., Brain, A.P.R., Quinn, P.J., Williams, W.P.: Low pH and phospholipase A2 treatment induce the phase separation of non-bilayer lipids within pea chloroplast membranes.–FEBS Lett. 183: 161–166, 1985.
Thomas, P.G., Quinn, P.J., Williams, W.P.: The origin of photosystem I-mediated electron transport stimulation in heat-stressed chloroplasts.–Planta 167: 133–139, 1986.
Thompson, L.K., Blaylock, R., Sturtevant, J.M., Brudvig, G.W.: Molecular basis of the heat denaturation of photosystem II.–Biochemistry 28: 6686–6695, 1989.
Thompson, L.K., Sturtevant, J.M., Brudvig, G.W.: Differential scanning calorimetric studies of photosystem II: Evidence for a structural role of cytochrome b 559 in the oxygen evolving complex.–Biochemistry 25: 6161–6169, 1986.
Tikhonov, A.N., Ruuge, Ė.K.: [ESR study of electron transport in photosynthetic systems. VII. Effects of temperature on the processes of electron transport between two photosystems and the structural state of chloroplast membrane.]–Mol. Biol. 12: 1028–1036, 1978. [In Russ.]
Yamane, Y., Kashino, Y., Koike, H., Satoh, K.: Increases in the fluorescence F0 level and reversible inhibition of Photosystem II reaction center by high-temperature treatments in higher plants.–Photosynth. Res. 52: 57–64, 1997.
Yamane, Y., Kashino, Y., Koike, H., Satoh, K.: Effects of high temperatures on the photosynthetic systems in spinach: Oxygen-evolving activities, fluorescence characteristics and the denaturation process.–Photosynth. Res. 57: 51–59, 1998.
Yamashita, T., Butler, W.L.: Inhibition of chloroplasts by UV-irradiation and heat-treatment.–Plant Physiol. 43: 2037–2040, 1968.
Author information
Authors and Affiliations
Additional information
This revised version was published online in March 2005 with corrections to the page numbers.
Rights and permissions
About this article
Cite this article
Semenova, G.A. Structural reorganization of thylakoid systems in response to heat treatment. Photosynthetica 42, 521–527 (2004). https://doi.org/10.1007/S11099-005-0008-z
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/S11099-005-0008-z