Abstract
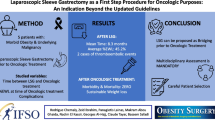
Background and aims
Lynch syndrome (LS) is the most common cause of hereditary colorectal cancer and is associated with an increased lifetime risk of gastric and duodenal cancers of 8–16% and 7%, respectively; therefore, we aim to describe an esophagogastroduodenoscopy (EGD) surveillance program for upper gastrointestinal (GI) precursor lesions and cancer in LS patients.
Methods
Patients who either had positive genetic testing or met clinical criteria for LS who had a surveillance EGD at our institution from 1996 to 2017 were identified. Patients were included if they had at least two EGDs or an upper GI cancer detected on the first surveillance EGD. EGD and pathology reports were extracted manually.
Results
Our cohort included 247 patients with a mean age of 47.1 years (SD 12.6) at first EGD. Patients had a mean of 3.5 EGDs (range 1–16). Mean duration of follow-up was 5.7 years. Average interval between EGDs was 2.3 years. Surveillance EGD detected precursor lesions in 8 (3.2%) patients, two (0.8%) gastric cancers and two (0.8%) duodenal cancers. Two interval cancers were diagnosed: a duodenal adenocarcinoma was detected 2 years, 8 months after prior EGD and a jejunal adenocarcinoma was detected 1 year, 9 months after prior EGD.
Conclusions
Our data suggest that surveillance EGD is a useful tool to help detect precancerous and cancerous upper GI lesions in LS patients. To our knowledge, this is the first study to examine a program of surveillance EGDs in LS patients. More data are needed to determine the appropriate surveillance interval.



Similar content being viewed by others
Availability of data and material
The datasets generated during and/or analyzed during the current study are not publicly available due to The Health Insurance Portability and Accountability Act of 1996 but are available from the corresponding author on reasonable request (de-identified).
References
Rustgi AK (2007) The genetics of hereditary colon cancer. Genes Dev 21(20):2525–2538. https://doi.org/10.1101/gad.1593107
Lynch HT, de la Chapelle A (1999) Genetic susceptibility to non-polyposis colorectal cancer. J Med Genet 36(11):801–818
Provenzale D, Gupta S, Ahnen DJ et al (2019) NCCN clinical practice guidelines in oncology. Genetic/familial high-risk assessment: colorectal. 3:2019 Accessed April 22, 2020. https://www.nccn.org/professionals/physician_gls/pdf/genetics_colon.pdf
Stoffel EM, Mangu PB, Gruber SB et al (2015) Hereditary colorectal cancer syndromes: American Society of Clinical Oncology Clinical Practice Guideline endorsement of the familial risk-colorectal cancer: European Society for Medical Oncology Clinical Practice Guidelines. J Clin Oncol Off J Am Soc Clin Oncol 33(2):209–217. https://doi.org/10.1200/JCO.2014.58.1322
Giardiello FM, Allen JI, Axilbund JE et al (2014) Guidelines on genetic evaluation and management of Lynch syndrome: a consensus statement by the US Multi-Society Task Force on colorectal cancer. Gastroenterology 147(2):502–526. https://doi.org/10.1053/j.gastro.2014.04.001
Syngal S, Brand RE, Church JM et al (2015) ACG clinical guideline: genetic testing and management of hereditary gastrointestinal cancer syndromes. Am J Gastroenterol 110(2):223–262; quiz 263. https://doi.org/10.1038/ajg.2014.435
Rubenstein JH, Enns R, Heidelbaugh J, Barkun A (2015) Clinical Guidelines Committee. American Gastroenterological Association Institute Guideline on the Diagnosis and Management of Lynch Syndrome. Gastroenterology 149(3):777–782; quiz e16–17. https://doi.org/10.1053/j.gastro.2015.07.036
Aarnio M, Sankila R, Pukkala E et al (1999) Cancer risk in mutation carriers of DNA-mismatch-repair genes. Int J Cancer 81(2):214–218. https://doi.org/10.1002/(sici)1097-0215(19990412)81:2%3c214::aid-ijc8%3e3.0.co;2-l
Møller P, Seppälä TT, Bernstein I et al (2018) Cancer risk and survival in path_MMR carriers by gene and gender up to 75 years of age: a report from the Prospective Lynch Syndrome Database. Gut 67(7):1306–1316. https://doi.org/10.1136/gutjnl-2017-314057
Park YJ, Shin K-H, Park J-G (2000) Risk of gastric cancer in hereditary nonpolyposis Colorectal Cancer in Korea. Clin Cancer Res 6(8):2994–2998
Aarnio M, Salovaara R, Aaltonen LA, Mecklin JP, Järvinen HJ (1997) Features of gastric cancer in hereditary non-polyposis colorectal cancer syndrome. Int J Cancer 74(5):551–555. https://doi.org/10.1002/(sici)1097-0215(19971021)74:5%3c551::aid-ijc13%3e3.0.co;2-9
Capelle LG, Van Grieken NCT, Lingsma HF et al (2010) Risk and epidemiological time trends of gastric cancer in Lynch syndrome carriers in the Netherlands. Gastroenterology 138(2):487–492. https://doi.org/10.1053/j.gastro.2009.10.051
Wroblewski LE, Peek RM, Wilson KT (2010) Helicobacter pylori and gastric cancer: factors that modulate disease risk. Clin Microbiol Rev 23(4):713–739. https://doi.org/10.1128/CMR.00011-10
Renkonen-Sinisalo L, Sipponen P, Aarnio M et al (2002) No support for endoscopic surveillance for gastric cancer in hereditary non-polyposis colorectal cancer. Scand J Gastroenterol 37(5):574–577. https://doi.org/10.1080/00365520252903134
Galiatsatos P, Labos C, Jeanjean M, Miller K, Foulkes WD (2017) Low yield of gastroscopy in patients with Lynch syndrome. Turk J Gastroenterol Off J Turk Soc Gastroenterol 28(6):434–438. https://doi.org/10.5152/tjg.2017.17176
Leach BH (2020) Lynch syndrome. American College of Gastroenterology. Accessed April 21, 2020. https://gi.org/topics/lynch-syndrome/
Vasen HFA, Blanco I, Aktan-Collan K et al (2013) Revised guidelines for the clinical management of Lynch syndrome (HNPCC): recommendations by a group of European experts. Gut 62(6):812–823. https://doi.org/10.1136/gutjnl-2012-304356
Yurgelun MB, Hampel H (2020) Recent advances in Lynch syndrome: diagnosis, treatment, and cancer prevention | Am Society Clin Oncol Educational Book. Accessed April 21. https://doi.org/10.1200/EDBK_208341
Vasen HF, Watson P, Mecklin JP, Lynch HT (1999) New clinical criteria for hereditary nonpolyposis colorectal cancer (HNPCC, Lynch syndrome) proposed by the International Collaborative group on HNPCC. Gastroenterology 116(6):1453–1456. https://doi.org/10.1016/s0016-5085(99)70510-x
Umar A, Boland CR, Terdiman JP et al (2004) Revised Bethesda Guidelines for hereditary nonpolyposis colorectal cancer (Lynch syndrome) and microsatellite instability. J Natl Cancer Inst 96(4):261–268. https://doi.org/10.1093/jnci/djh034
Danesh J (1999) Helicobacter pylori infection and gastric cancer: systematic review of the epidemiological studies. Aliment Pharmacol Ther 13(7):851–856. https://doi.org/10.1046/j.1365-2036.1999.00546.x
Uemura N, Okamoto S, Yamamoto S et al. (2001) Helicobacter pylori infection and the development of gastric cancer. https://doi.org/10.1056/NEJMoa001999.
Humans IWG (1994). On the E of CR to. Schistosomes, liver flukes and Helicobacter pylori. International Agency for Research on Cancer.
Kim J, Braun D, Ukaegbu C et al (2020) Clinical factors associated with gastric cancer in individuals with Lynch syndrome. Clin Gastroenterol Hepatol Off Clin Pract J Am Gastroenterol Assoc 18(4):830-837.e1. https://doi.org/10.1016/j.cgh.2019.07.012
Vasen HFA, Möslein G, Alonso A et al (2007) Guidelines for the clinical management of Lynch syndrome (hereditary non-polyposis cancer). J Med Genet 44(6):353–362. https://doi.org/10.1136/jmg.2007.048991
Funding
This work was supported by grant P30 CA008748/CA/NCI NIH HHS/United States.
Author information
Authors and Affiliations
Contributions
A.H.C. data curation: lead, formal analysis: lead, investigation: equal, methodology: equal, writing—original draft: lead, writing—review and editing: equal; J.J.Y. conceptualization: equal, data curation: equal, formal analysis: equal, investigation: equal, methodology: equal, project administration: equal, supervision: equal, writing—original draft: equal, writing—review and editing: equal; A.L. formal analysis: supporting, supervision: supporting, writing—review and editing: supporting; A.J.M. writing—review and editing: equal; J.S. data curation: supporting, supervision: supporting, writing—review and editing: supporting; J.M. data curation: equal; D.C. writing—review and editing: supporting; H.G. writing—review and editing: supporting; E.L. writing—review and editing: supporting; M.A.S. writing—review and editing: supporting; Z.K.S. writing—review and editing: supporting; E.K. funding acquisition: supporting; writing—review and editing: supporting; M.D. writing—review and editing: supporting; R.B.M. conceptualization: lead, formal analysis: equal, investigation: equal, methodology: equal, project administration: lead, supervision: lead, writing—original draft: equal, writing—review and editing: equal.
Corresponding author
Ethics declarations
Ethics approval
MSKCC’s Institutional Review Board approved this study.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
A.H.C., J.J.Y., A.L., A.J.M., J.S., J.M., D.C., H.G., E.L., M.A.S., E.K., M.D., and R.B.M. declare no conflicts of interest. Z.K.S. reports that an immediate family member holds consulting/advisory roles with Allergan, Adverum, Genentech/Roche, Gyroscope Tx, Novartis, Neurogene, Optos Plc, Regeneron, and Regenxbio.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ceravolo, A.H., Yang, J.J., Latham, A. et al. Effectiveness of a surveillance program of upper endoscopy for upper gastrointestinal cancers in Lynch syndrome patients. Int J Colorectal Dis 37, 231–238 (2022). https://doi.org/10.1007/s00384-021-04053-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-021-04053-y