Abstract
Key message
Viral-induced gene silencing of selected biosynthetic genes decreased overall carotenoid accumulation in California poppy. Regulation of carotenogenesis was linked with pigment sequestration, not changes in biosynthetic gene expression.
Abstract
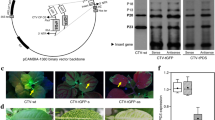
Genes of carotenogenesis are well described, but understanding how they affect carotenoid accumulation has proven difficult because of plant lethality when the pigments are lacking. Here, we used a Tobacco Rattle Virus-based virus-induced-gene-silencing (VIGS) approach in California poppy (Eschscholzia californica) to investigate how silencing of the carotenoid biosynthetic pathway genes affects carotenoid metabolite accumulation and RNA transcript abundance of the carotenoid biosynthetic pathway genes. VIGS of upstream (PDS and ZDS) and downstream (βOH and ZEP) genes reduced transcript abundance of the targeted genes in the poppy petals while having no effect on abundance of the other carotenogenesis genes. Silencing of PDS, ZDS, βOH and ZEP genes reduced total pigment concentration by 75–90% and altered petal colour. HPLC and LC-MS measurements suggested that petal colour changes were caused by substantially altered pigment profiles and quantity. Carotenoid metabolites were different to those normally detected in wild-type petals accumulated but overall carotenoid concentration was less, suggesting the chemical form of carotenoid was important for whether it could be stored at high amounts. In poppy petals, eschscholtzxanthin and retro-carotene-triol were the predominant carotenoids, present mainly as esters. Specific esterification enzymes for specific carotenoids and/or fatty acids appear key for enabling petal carotenoids to accumulate to high amounts. Our findings argue against a direct role for carotenoid metabolites regulating carotenogenesis genes in the petals of California poppy as transcript abundance of carotenogenesis genes studied was unchanged, while the petal carotenoid metabolite profile changed substantially.







Similar content being viewed by others
References
Al-Babili S, Hartung W, Kleinig H, Beyer P (1999) CPTA modulates levels of carotenogenic proteins and their mRNAs and affects carotenoid and ABA content as well as chromoplast structure in Narcissus pseudonarcissus flowers. Plant Biol 1:607–612
Altschul SF, Madden TL, Schaffer AA, Zhang JH, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Ampomah-Dwamena C, McGhie T, Wibisono R, Montefiori M, Hellens RP, Allan AC (2009) The kiwifruit lycopene beta-cyclase plays a significant role in carotenoid accumulation in fruit. J Exp Bot 60:3765–3779
Andrewes A, Engler G, Borch G, Strain H, Liaaen-Jensen S (1979) Absolute configuration of eschscholtzxanthin. Phytochemistry 18:303–309
Ariizumi T, Kishimoto S, Kakami R, Maoka T, Hirakawa H, Suzuki Y, Ozeki Y, Shirasawa K, Bernillon S, Okabe Y (2014) Identification of the carotenoid modifying gene PALE YELLOW PETAL 1 as an essential factor in xanthophyll esterification and yellow flower pigmentation in tomato (Solanum lycopersicum). Plant J 79:453–465
Baroli I, Do AD, Yamane T, Niyogi KK (2003) Zeaxanthin accumulation in the absence of a functional xanthophyll cycle protects Chlamydomonas reinhardtii from photooxidative stress. Plant Cell 15:992–1008
Borovsky Y, Tadmor Y, Bar E, Meir A, Lewinsohn E, Paran I (2013) Induced mutation in β-CAROTENE HYDROXYLASE results in accumulation of β-carotene and conversion of red to orange color in pepper fruit. Theor Appl Genet 126:557–565
Britton G (1995) Structure and properties of carotenoids in relation to function. FASEB J 9:1551–1558
Burns J, Fraser PD, Bramley PM (2003) Identification and quantification of carotenoids, tocopherols and chlorophylls in commonly consumed fruits and vegetables. Phytochemistry 62:939–947
Cazzonelli CI, Pogson BJ (2010) Source to sink: regulation of carotenoid biosynthesis in plants. Trends Plant Sci 15:266–274
Chen JC, Jiang CZ, Gookin TE, Hunter DA, Clark DG, Reid MS (2007) Chalcone synthase as a reporter in virus-induced gene silencing studies of flower senescence. Plant Mol Biol 2004 55:521–530
Chi W, Sun X, Zhang L (2013) Intracellular signaling from plastid to nucleus. Ann Rev Plant Biol 64:559–582
Conti A, Pancaldi S, Fambrini M, Michelotti V, Bonora A, Salvini M, Pugliesi C (2004) A deficiency at the gene coding for zeta-carotene desaturase characterizes the sunflower non dormant-1 mutant. Plant Cell Physiol 45:445–455
Cunningham FX, Lee H, Gantt E (2007) Carotenoid biosynthesis in the primitive red alga Cyanidioschyzon merolae. Eukaryot Cell 6:533–545
Deng X, Elomaa P, Nguyen CX, Hytönen T, Valkonen J, Teeri TH (2012) Virus-induced gene silencing for Asteraceae—a reverse genetics approach for functional genomics in Gerbera hybrida. Plant Biotechnol J 10:970–978
Egea I, Barsan C, Bian W, Purgatto E, Latché A, Chervin C, Bouzayen M, Pech J-C (2010) Chromoplast differentiation: current status and perspectives. Plant Cell Physiol 51:1601–1611
Enfissi E, Nogueira M, Bramley P, Fraser P (2017) The regulation of carotenoid formation in tomato fruit. Plant J 89(4):774–788
Fantini E, Falcone G, Frusciante S, Giliberto L, Giuliano G (2013) Dissection of tomato lycopene biosynthesis through virus-induced gene silencing. Plant Physiol 163:986–998
Farré G, Sanahuja G, Naqvi S, Bai C, Capell T, Zhu C, Christou P (2010) Travel advice on the road to carotenoids in plants. Plant Sci 179:28–48
Fernandez-Pozo N, Rosli HG, Martin GB, Mueller LA (2015) The SGN VIGS tool: user-friendly software to design virus-induced gene silencing (VIGS) constructs for functional genomics. Mol Plant 8:486–488
Fraser PD, Pinto MES, Holloway DE, Bramley PM (2000) Application of high-performance liquid chromatography with photodiode array detection to the metabolic profiling of plant isoprenoids. Plant J 24:551–558
Fraser PD, Enfissi EM, Halket JM, Truesdale MR, Yu D, Gerrish C, Bramley PM (2007) Manipulation of phytoene levels in tomato fruit: effects on isoprenoids, plastids, and intermediary metabolism. Plant Cell 19:3194–3211
Fu X, Kong W, Peng G, Zhou J, Azam M, Xu C, Grierson D, Chen K (2011) Plastid structure and carotenogenic gene expression in red-and white-fleshed loquat (Eriobotrya japonica) fruits. J Exp Bot 63:341–354
Galpaz N, Ronen G, Khalfa Z, Zamir D, Hirschberg J (2006) A chromoplast-specific carotenoid biosynthesis pathway is revealed by cloning of the tomato white-flower locus. Plant Cell 18:1947–1960
Kamffer Z, Bindon KA, Oberholster A (2010) Optimization of a method for the extraction and quantification of carotenoids and chlorophylls during ripening in grape berries (Vitis vinifera cv. Merlot). J Agric Food Chem 58:6578–6586
Kato M, Ikoma Y, Matsumoto H, Sugiura M, Hyodo H, Yano M (2004) Accumulation of carotenoids and expression of carotenoid biosynthetic genes during maturation in citrus fruit. Plant Physiol 134:824–837
Kilcrease J, Collins AM, Richins RD, Timlin JA, O’Connell MA (2013) Multiple microscopic approaches demonstrate linkage between chromoplast architecture and carotenoid composition in diverse Capsicum annuum fruit. Plant J 76:1074–1083
Kim SH, Ahn YO, Ahn M-J, Jeong JC, Lee H-S, Kwak S-S (2013) Cloning and characterization of an Orange gene that increases carotenoid accumulation and salt stress tolerance in transgenic sweetpotato cultures. Plant Physiol Biochem 70:445–454
Lee HS, Castle WS, Coates GA (2001) High-performance liquid chromatography for the characterization of carotenoids in the new sweet orange (Earlygold) grown in Florida, USA. J Chromatogr 913:371–377
Li L, Yuan H (2013) Chromoplast biogenesis and carotenoid accumulation. Arch Biochem Biophys 539:102–109
Li L, Lu S, Cosman KM, Earle ED, Garvin DF, O’Neill J (2006) β-Carotene accumulation induced by the cauliflower Or gene is not due to an increased capacity of biosynthesis. Phytochemistry 67:1177–1184
Li L, Yang Y, Xu Q, Owsiany K, Welsch R, Chitchumroonchokchai C, Lu S, Van Eck J, Deng X-X, Failla M (2012) The Or gene enhances carotenoid accumulation and stability during post-harvest storage of potato tubers. Mol Plant 5:339–352
Liu E, Page JE (2008) Optimized cDNA libraries for virus-induced gene silencing (VIGS) using tobacco rattle virus. Plant Methods 4:5
Liu H, Reavy B, Swanson M, MacFarlane SA (2002) Functional replacement of the tobacco rattle virus cysteine-rich protein by pathogenicity proteins from unrelated plant viruses. Virology 298:232–239
Lu S, Van Eck J, Zhou X, Lopez AB, O’Halloran DM, Cosman KM, Conlin BJ, Paolillo DJ, Garvin DF, Vrebalov J (2006) The cauliflower Or gene encodes a DnaJ cysteine-rich domain-containing protein that mediates high levels of β-carotene accumulation. Plant Cell 18:3594–3605
Lutzow M, Haaf F, Englert G, Beyer P, Kleinig H (1996) NADPH and protoporphyrin IX dependent conversion of violaxanthin to a retro-carotenoid in Narcissus pseudonarcissus chromoplasts in vitro. Phytochemistry 41:729–734
Lyons R, Booze-Daniels J (1986) Characteristics of the photoperiodic response of California poppy. J Am Soc Hortic Sci 111:593–596
Mann V, Harker M, Pecker I, Hirschberg J (2000) Metabolic engineering of astaxanthin production in tobacco flowers. Nat Biotech 18:888
Maoka T, Fujiwara Y, Hashimoto K, Takeda S, Takaragaki S, Ida K (2000) A new retro-carotenoid from the petals of the californian yellow poppy Eschscholtzia californica. J Nat Prod 63(9):1288–1289
Moehs CP, Tian L, Osteryoung KW, DellaPenna D (2001) Analysis of carotenoid biosynthetic gene expression during marigold petal development. Plant Mol Biol 45:281–293
Moise AR, Al-Babili S, Wurtzel ET (2013) Mechanistic aspects of carotenoid biosynthesis. Chem Rev 114:164–193
Namitha KK, Negi PS (2010) Chemistry and Biotechnology of Carotenoids. Crit Rev Food Sci Nutr 50:728–760
Nogueira M, Mora L, Enfissi EM, Bramley PM, Fraser PD (2013) Subchromoplast sequestration of carotenoids affects regulatory mechanisms in tomato lines expressing different carotenoid gene combinations. Plant Cell 25:4560–4579
Ohmiya A (2013) Qualitative and quantitative control of carotenoid accumulation in flower petals. Sci Hort 163:10–19
Pecker I, Chamovitz D, Linden H, Sandmann G, Hirschberg J (1992) A single polypeptide catalyzing the conversion of phytoene to zeta-carotene is transcriptionally regulated during tomato fruit ripening. Proc Nat Acad Sci 89:4962–4966
Qin GJ, Gu HY, Ma LG, Peng YB, Deng XW, Chen ZL, Qu LJ (2007) Disruption of phytoene desaturase gene results in albino and dwarf phenotypes in Arabidopsis by impairing chlorophyll, carotenoid, and gibberellin biosynthesis. Cell Res 17:471–482
Rodriguez-Concepcion M, Stange C (2013) Biosynthesis of carotenoids in carrot: an underground story comes to light. Arch Biochem Biophys 539:110–116
Ryu CM, Anand A, Kang L, Mysore KS (2004) Agrodrench: a novel and effective agroinoculation method for virus-induced gene silencing in roots and diverse Solanaceous species. Plant J 40:322–331
Schaub P, Al-Babili S, Drake R, Beyer P (2005) Why is Golden Rice golden (yellow) instead of red? Plant Physiol 138:441–450
Schledz M, Al Babili S, Lintig JV, Haubruck H, Rabbani S, Kleinig H, Beyer P (1996) Phytoene synthase from Narcissus pseudonarcissus: functional expression, galactolipid requirement, topological distribution in chromoplasts and induction during flowering. Plant J 10:781–792
Takaichi S (2011) Carotenoids in algae: distributions, biosyntheses and functions. Mar Drugs 9:1101–1118
Takaichi S, Mochimaru M (2007) Carotenoids and carotenogenesis in cyanobacteria: unique ketocarotenoids and carotenoid glycosides. Cell Mol Life Sci 64:2607–2619
Tekleyohans DG, Lange S, Becker A (2013) Virus-induced gene silencing of the alkaloid-producing basal eudicot model plant Eschscholzia californica (California Poppy). Virus-induced Gene Silencing Methods Protocols:83–98
Tian S, Li L, Chai W, Shah S, Gong Z (2014) Effects of silencing key genes in the capsanthin biosynthetic pathway on fruit color of detached pepper fruits. BMC Plant Biol 14(1):314
Toledo-Ortiz G, Huq E, Rodríguez-Concepción M (2010) Direct regulation of phytoene synthase gene expression and carotenoid biosynthesis by phytochrome-interacting factors. Proc Nat Acad Sci 107:11626–11631
Wakelin AM, Lister CE, Conner AJ (2003) Inheritance and biochemistry of pollen pigmentation in California poppy (Eschscholzia californica Cham.). Int J Plant Sci 164:867–875
Wan CY, Wilkins TA (1994) A modified hot borate method significantly enhances the yield of high-quality RNA from cotton (Gossypium-hirsutum L.). Anal Biochem 223:7–12
Wege S, Scholz A, Gleissberg S, Becker A (2007) Highly efficient virus-induced gene silencing (VIGS) in California Poppy (Eschscholzia californica): an evaluation of VIGS as a strategy to obtain functional data from non-model plants. Ann Bot 100:641–649
Wellburn AR (1994) The spectral determination of chlorophyll-a and chlorophyll-b, as well as total carotenoids, using various solvents with spectrophotometers of different resolution. J Plant Physiol 144:307–313
Xu F, Yuan Q, Dong H (2006) Determination of lycopene and β-carotene by high-performance liquid chromatography using sudan I as internal standard. J Chromatogr B 838:44–49
Yamamizo C, Kishimoto S, Ohmiya A (2010) Carotenoid composition and carotenogenic gene expression during Ipomoea petal development. J Exp Bot 61:709–719
Yu BY, Lydiate DJ, Young LW, Schafer UA, Hannoufa A (2008) Enhancing the carotenoid content of Brassica napus seeds by downregulating lycopene epsilon cyclase. Transgenic Res 17:573–585
Zhu C, Bai C, Sanahuja G, Yuan D, Farré G, Naqvi S, Shi L, Capell T, Christou P (2010) The regulation of carotenoid pigmentation in flowers. Arch Biochem Biophys 504:132–141
Acknowledgements
Thanks to Philippa Barrell (Plant & Food Research, Lincoln) for providing the initial California poppy petal cDNA and to Dr Mark Fiers (Plant & Food Research, Lincoln) for providing sequences from the California poppy 454 sequence database. Thanks also to Ian King for managing the plants in the containment facility and to Dinesh Kumar for providing the pTRV plasmids. Thanks to Dr Nigel Joyce (Plant & Food Research, Lincoln) for performing LCMS. We thank Duncan Hedderley for the statistical analysis. We thank F. Hofmann-La Roche for supplying zeaxanthin as a purified standard for analysis of carotenoid pigments.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Salim Al-Babili.
Dedicated to the memory of Michael McManus, September 1957-July 2015.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhou, J., Hunter, D.A., Lewis, D.H. et al. Insights into carotenoid accumulation using VIGS to block different steps of carotenoid biosynthesis in petals of California poppy. Plant Cell Rep 37, 1311–1323 (2018). https://doi.org/10.1007/s00299-018-2314-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-018-2314-5