Abstract
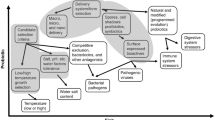
Gut health has major implications for the general health of food-producing animals such as the layer birds used in the egg industry. In order to modulate gut microbiota for the benefit of gut health, an understanding of the dynamics and details of the development of gut microbiota is critical. The present study investigated the phylogenetic composition of the gut microbiota of a commercial layer flock raised in cages from hatch to the end of the production cycle. This study also aimed to understand the establishment and development of gut microbiota in layer chickens. Results showed that the faecal microbiota was dominated by phyla Firmicutes and Proteobacteria in the rearing phase, but Bacteroidetes in mid lay and late lay phase. The gut microbiota composition changed significantly during the transfer of the flock from the rearing to the production shed. The richness and diversity of gut microbiota increased after week 6 of the flocks age and stabilized in the mid and late lay phase. The overall dynamics of gut microbiota development was similar to that reported in earlier studies, but the phylogenetic composition at the phylum and family level was different. The production stage of the birds is one of the important factors in the development of gut microbiota. This study has contributed to a better understanding of baseline gut microbiota development over the complete life cycles in layer chickens and will help to develop strategies to improve the gut health.
Key points
• Faecal microbiota of caged hens was dominated by phyla Firmicutes and Proteobacteria in the rearing phase.
• The gut microbiota composition changed significantly during the transfer of the flock from the rearing to the production shed.
• The richness and diversity of gut microbiota increased after week 6 of the flocks age and stabilized in the mid and late lay phase.







Similar content being viewed by others
Data availability
The 16S rRNA sequence data are available from the MG RAST database under the accession number mgl832560.
References
AECL (2019) Australian Eggs Annual Report. Sydney, NSW, Australia
Ahmad M, Krishnan S, Ramakrishna B, Mathan M, Pulimood A, Murthy S (2000) Butyrate and glucose metabolism by colonocytes in experimental colitis in mice. Gut 46(4):493–499
Alagawany M, Abd El-Hack ME, Farag MR, Sachan S, Karthik K, Dhama K (2018) The use of probiotics as eco-friendly alternatives for antibiotics in poultry nutrition. Environ Sci Pollut Res 25(11):10611–10618
Ashelford KE, Chuzhanova NA, Fry JC, Jones AJ, Weightman AJ (2005) At least 1 in 20 16S rRNA sequence records currently held in public repositories is estimated to contain substantial anomalies. Appl Environ Microbiol 71(12):7724–7736
Benjdia A, Martens EC, Gordon JI, Berteau O (2011) Sulfatases and a radical S-adenosyl-L-methionine (AdoMet) enzyme are key for mucosal foraging and fitness of the prominent human gut symbiont, Bacteroides thetaiotaomicron. J Biol Chem 286(29):25973–25982
Callaway T, Dowd S, Wolcott R, Sun Y, McReynolds J, Edrington T, Byrd J, Anderson R, Krueger N, Nisbet D (2009) Evaluation of the bacterial diversity in cecal contents of laying hens fed various molting diets by using bacterial tag-encoded FLX amplicon pyrosequencing. Poult Sci 88(2):298–302
Caporaso JG, Kuczynski J, Stombaugh J, Bittinger K, Bushman FD, Costello EK, Fierer N, Pena AG, Goodrich JK, Gordon JI (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7(5):335–336
De Cesare A, Sirri F, Manfreda G, Moniaci P, Giardini A, Zampiga M, Meluzzi A (2017) Effect of dietary supplementation with Lactobacillus acidophilus D2/CSL (CECT 4529) on caecum microbioma and productive performance in broiler chickens. PLoS One 12(5):e0176309
DeSantis TZ, Hugenholtz P, Larsen N, Rojas M, Brodie EL, Keller K, Huber T, Dalevi D, Hu P, Andersen GL (2006) Greengenes, a chimera-checked 16S rRNA gene database and workbench compatible with ARB. Appl Environ Microbiol 72(7):5069–5072
Donoghue DJ (2003) Antibiotic residues in poultry tissues and eggs: human health concerns? Poult Sci 82(4):618–621
Duncan SH, Belenguer A, Holtrop G, Johnstone AM, Flint HJ, Lobley GE (2007) Reduced dietary intake of carbohydrates by obese subjects results in decreased concentrations of butyrate and butyrate-producing bacteria in feces. Appl Environ Microbiol 73(4):1073–1078
Egan M, Van Sinderen D (2018) Carbohydrate metabolism in bifidobacteria. In: Mattarelli P, Biavati B, Holzapfel WH, Wood BJB (eds) The Bifidobacteria and related organisms: biology, taxonomy, applications, 1st edn. Elsevier, San Diego, pp 145–164
Fadrosh DW, Ma B, Gajer P, Sengamalay N, Ott S, Brotman RM, Ravel J (2014) An improved dual-indexing approach for multiplexed 16S rRNA gene sequencing on the Illumina MiSeq platform. Microbiome 2(1):6
Forte C, Manuali E, Abbate Y, Papa P, Vieceli L, Tentellini M, Trabalza-Marinucci M, Moscati L (2018) Dietary Lactobacillus acidophilus positively influences growth performance, gut morphology, and gut microbiology in rurally reared chickens. Poult Sci 97(3):930–936
Gole VC, Caraguel CG, Sexton M, Fowler C, Chousalkar KK (2014) Shedding of Salmonella in single age caged commercial layer flock at an early stage of lay. Int J Food Microbiol 189:61–66
Guo F-F, Yu T-C, Hong J, Fang J-Y (2016) Emerging roles of hydrogen sulfide in inflammatory and neoplastic colonic diseases. Front Physiol 7:156
Hy-Line Management guide (2018). Hy-Line Brown Management Guide. Hy-Line International© BRN.COM.AUS rev. 1-14-19. 1-29.
Johnson TJ, Youmans BP, Noll S, Cardona C, Evans NP, Karnezos TP, Ngunjiri JM, Abundo MC, Lee C-W (2018) A consistent and predictable commercial broiler chicken bacterial microbiota in antibiotic-free production displays strong correlations with performance. Appl Environ Microbiol 84(12):e00362–e00318
Kers JG, Velkers FC, Fischer EA, Hermes GD, Stegeman JA, Smidt H (2018) Host and environmental factors affecting the intestinal microbiota in chickens. Front Microbiol 9:235
Khan S, Moore RJ, Stanley D, Chousalkar KK (2020) Gut microbiota of laying hens and its manipulation with prebiotics and probiotics to enhance gut health and food safety. Appl Environ Microbiol 86
Knudsen BE, Bergmark L, Munk P, Lukjancenko O, Priemé A, Aarestrup FM, Pamp SJ (2016) Impact of sample type and DNA isolation procedure on genomic inference of microbiome composition. MSystems 1(5):e00095–e00016
Liu L, Lin L, Zheng L, Tang H, Fan X, Xue N, Li M, Liu M, Li X (2018) Cecal microbiome profile altered by Salmonella enterica serovar Enteritidis inoculation in chicken. Gut pathogens 10(1):34
Louis P, Flint HJ (2007) Development of a semiquantitative degenerate real-time PCR-based assay for estimation of numbers of butyryl-coenzyme A (CoA) CoA transferase genes in complex bacterial samples. Appl Environ Microbiol 73(6):2009–2012
Lumpkins BS, Batal AB, Lee M (2008) The effect of gender on the bacterial community in the gastrointestinal tract of broilers. Poult Sci 87(5):964–967. https://doi.org/10.3382/ps.2007-00287
Mahdavi A, Rahmani H, Pourreza J (2005) Effect of probiotic supplements on egg quality and laying hen’s performance. Int J Poult Sci 4(7):488–492
Manichanh C, Rigottier-Gois L, Bonnaud E, Gloux K, Pelletier E, Frangeul L, Nalin R, Jarrin C, Chardon P, Marteau P (2006) Reduced diversity of faecal microbiota in Crohn’s disease revealed by a metagenomic approach. Gut 55(2):205–211
Mon KKZ, Saelao P, Halstead MM, Chanthavixay G, Chang H-C, Garas L, Maga EA, Zhou H (2015) Salmonella enterica serovars Enteritidis infection alters the indigenous microbiota diversity in young layer chicks. Front Vet Sci 2(61). https://doi.org/10.3389/fvets.2015.00061
Nahashon SN, Nakaue HS, Mirosh LW (1996) Performance of single comb white leghorn fed a diet supplemented with a live microbial during the growth and egg laying phases. Anim Feed Sci Technol 57(1-2):25–38
Ngunjiri JM, Taylor KJ, Abundo MC, Jang H, Elaish M, Mahesh K, Ghorbani A, Wijeratne S, Weber BP, Johnson TJ (2019) Farm stage, bird age, and body site dominantly affect the quantity, taxonomic composition, and dynamics of respiratory and gut microbiota of commercial layer chickens. Appl Environ Microbiol 85(9):e03137–e03118
Nordentoft S, Mølbak L, Bjerrum L, De Vylder J, Van Immerseel F, Pedersen K (2011) The influence of the cage system and colonisation of Salmonella enteritidis on the microbial gut flora of laying hens studied by T-RFLP and 454 pyrosequencing. BMC Microbiol 11(1):187
Oakley BB, Kogut MH (2016) Spatial and temporal changes in the broiler chicken cecal and fecal microbiomes and correlations of bacterial taxa with cytokine gene expression. Front Vet Sci 3:11
Ott S, Musfeldt M, Wenderoth D, Hampe J, Brant O, Fölsch U, Timmis K, Schreiber S (2004) Reduction in diversity of the colonic mucosa associated bacterial microflora in patients with active inflammatory bowel disease. Gut 53(5):685–693
Parada Venegas D, De la Fuente MK, Landskron G, González MJ, Quera R, Dijkstra G, Harmsen HJ, Faber KN, Hermoso MA (2019) Short chain fatty acids (SCFAs)-mediated gut epithelial and immune regulation and its relevance for inflammatory bowel diseases. Front Immunol 10:277
Rey FE, Gonzalez MD, Cheng J, Wu M, Ahern PP, Gordon JI (2013) Metabolic niche of a prominent sulfate-reducing human gut bacterium. Proc Natl Acad Sci 110(33):13582–13587
Roediger W (1982) Utilization of nutrients by isolated epithelial cells of the rat colon. Gastroenterology 83(2):424–429
Scheppach W (1994) Effects of short chain fatty acids on gut morphology and function. Gut 35(1 Suppl):S35–S38
Siegerstetter SC, Schmitz-Esser S, Magowan E, Wetzels SU, Zebeli Q, Lawlor PG, O'Connell NE, Metzler-Zebeli BU (2017) Intestinal microbiota profiles associated with low and high residual feed intake in chickens across two geographical locations. PLoS One 12(11):e0187766. https://doi.org/10.1371/journal.pone.0187766
Stanley D, Denman SE, Hughes RJ, Geier MS, Crowley TM, Chen H, Haring VR, Moore RJ (2012) Intestinal microbiota associated with differential feed conversion efficiency in chickens. Appl Microbiol Biotechnol 96(5):1361–1369. https://doi.org/10.1007/s00253-011-3847-5
Stanley D, Geier MS, Denman SE, Haring VR, Crowley TM, Hughes RJ, Moore RJ (2013a) Identification of chicken intestinal microbiota correlated with the efficiency of energy extraction from feed. Vet Microbiol 164(1-2):85–92
Stanley D, Geier MS, Hughes RJ, Denman SE, Moore RJ (2013b) Highly variable microbiota development in the chicken gastrointestinal tract. PLoS One 8(12):e84290
Stanley D, Hughes RJ, Moore RJ (2014) Microbiota of the chicken gastrointestinal tract: influence on health, productivity and disease. Appl Microbiol Biotechnol 98(10):4301–4310
Stanley D, Hughes RJ, Geier MS, Moore RJ (2016) Bacteria within the gastrointestinal tract microbiota correlated with improved growth and feed conversion: challenges presented for the identification of performance enhancing probiotic bacteria. Front Microbiol 7:187
Stecher B, Chaffron S, Käppeli R, Hapfelmeier S, Freedrich S, Weber TC, Kirundi J, Suar M, McCoy KD, von Mering C (2010) Like will to like: abundances of closely related species can predict susceptibility to intestinal colonization by pathogenic and commensal bacteria. PLoS Pathog 6(1):e1000711
Videnska P, Sisak F, Havlickova H, Faldynova M, Rychlik I (2013) Influence of Salmonella enterica serovar Enteritidis infection on the composition of chicken cecal microbiota. BMC Vet Res 9(1):140. https://doi.org/10.1186/1746-6148-9-140
Videnska P, Sedlar K, Lukac M, Faldynova M, Gerzova L, Cejkova D, Sisak F, Rychlik I (2014) Succession and replacement of bacterial populations in the caecum of egg laying hens over their whole life. PLoS One 9(12)
Walker AW, Duncan SH, Leitch ECM, Child MW, Flint HJ (2005) pH and peptide supply can radically alter bacterial populations and short-chain fatty acid ratios within microbial communities from the human colon. Appl Environ Microbiol 71(7):3692–3700
Wrzosek L, Miquel S, Noordine M-L, Bouet S, Chevalier-Curt MJ, Robert V, Philippe C, Bridonneau C, Cherbuy C, Robbe-Masselot C (2013) Bacteroides thetaiotaomicron and Faecalibacterium prausnitzii influence the production of mucus glycans and the development of goblet cells in the colonic epithelium of a gnotobiotic model rodent. BMC Biol 11(1):1-13, 61
Yan W, Sun C, Yuan J, Yang N (2017) Gut metagenomic analysis reveals prominent roles of Lactobacillus and cecal microbiota in chicken feed efficiency. Sci Rep 7:45308. https://doi.org/10.1038/srep45308
Yang J, Martínez I, Walter J, Keshavarzian A, Rose DJ (2013) In vitro characterization of the impact of selected dietary fibers on fecal microbiota composition and short chain fatty acid production. Anaerobe 23:74–81
Zakrzewski M, Proietti C, Ellis JJ, Hasan S, Brion M-J, Berger B, Krause L (2016) Calypso: a user-friendly web-server for mining and visualizing microbiome–environment interactions. Bioinformatics 33(5):782–783
Acknowledgements
Mr. Nitish Joat is a recipient of postgraduate scholarship from the University of Adelaide. Help from Andrea McWhorter and Dr. Samiullah Khan is acknowledged. We would like to thank Ms Sonali Deshmukh for her technical assistance in the lab.
Funding
This research was financially supported by Australian Eggs under the grant number 18AEC.
Author information
Authors and Affiliations
Contributions
NJ, HV, RM, DS, KC contributed to the study design, animal trials, sample processing, and critical revision of the manuscript. NJ and KC collected samples from animals, NJ processed the samples for DNA extraction, HV and RM performed 16S rRNA gene sequencing, DS and NJ analysed the data, and NJ drafted the manuscript. All authors approved the manuscript for publication.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The experimental setup was approved by the University of Adelaide, Animal Ethics Committee under Approval Number No. S-2018-015. The protocol was carried out in accordance with the guidelines specified in the Australian Code for the Care and Use of Animals for Scientific Purposes 8th edition 2013.
Consent for publication
All authors have approved the submission of the manuscript.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(PDF 3332 kb)
Rights and permissions
About this article
Cite this article
Joat, N., Van, T.T.H., Stanley, D. et al. Temporal dynamics of gut microbiota in caged laying hens: a field observation from hatching to end of lay. Appl Microbiol Biotechnol 105, 4719–4730 (2021). https://doi.org/10.1007/s00253-021-11333-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-021-11333-8