Abstract
Objective
The characteristics of children with single-sided deafness (SSD) who become candidates for unilateral cochlear implantation (uCI) were identified.
Study design
In all, 118 children with SSD presenting from 2013–2019 to a tertiary pediatric children’s hospital were retrospectively assessed regarding candidacy for uCI.
Results
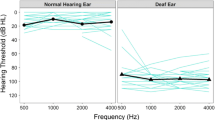
Of the 118 children, 103 had completed uCI candidacy assessment, while 15 were undergoing this assessment at the time of review. More than half of children did not go on to implantation (63/103, 61%), with the 2 main reasons being (1) half (31/63) did not meet candidacy criteria for implantation, most commonly due to cochlear nerve aplasia/hypoplasia (31/82 who were assessed with MRI, 38%) and (2) families (30/103; 29%) declined participation in the surgical arm of the trial. The most common etiologies of SSD in the 37/103 (36%) children who both met candidacy and consented to implantation were congenital cytomegalovirus (cCMV; 16/37, 43%), unknown (6/37, 16%), cochleovestibular anomaly and trauma (each 5/37, 14%).
Conclusions
Many children with SSD who present for implant candidacy assessment do not ultimately receive uCI. Major factors contributing to noncandidacy are cochlear nerve aplasia and parental acceptance of the intervention. While approximately half of children with SSD in our cohort were candidates for implantation, only 1/3 of the total cohort proceeded with implantation with the main predictors of acceptability of this intervention being an etiology (i.e., cCMV) that carries risk of progressive deterioration in the better hearing ear or SSD that was sudden in onset. These findings provide important insight into this new population of cochlear implant users and the emerging acceptance of intervention in children with SSD.
Zusammenfassung
Ziel
Es wurden Merkmale von Kindern mit einseitiger Taubheit („single-sided deafness“, SSD) herausgearbeitet, bei denen eine unilaterale Cochleaimplantation (uCI) infrage kam.
Studiendesign
Insgesamt wurden die Fälle von 118 Kindern mit SSD, die sich in der Zeit von 2013–2019 in einem Kinderkrankenhaus der Tertiärversorgung vorstellten, retrospektiv in Bezug auf die Eignung als Kandidat für eine uCI ausgewertet.
Ergebnisse
Von den 118 Kindern hatten 103 die vollständigen Voruntersuchungen für eine uCI durchlaufen, während 15 diese Voruntersuchungen zum Zeitpunkt der vorliegenden Arbeit noch absolvierten. Bei mehr als der Hälfte der Kindern kam es nicht zur Implantation (63/103, 61 %), Folgendes waren die beiden Hauptgründe: Zum einen erfüllte die Hälfte (31/63) nicht die Kriterien als Kandidat für eine Implantation, zumeist aufgrund einer Aplasie/Hypoplasie des N. cochlearis (31/82 mit erfolgter MRT-Untersuchung, 38 %), und zum anderen lehnten manche Familien (30/103, 29 %) die Teilnahme am chirurgischen Arm der Studie ab. Die häufigsten Ätiologien der SSD bei den 37/103 (36 %) Kindern, die sowohl die Kriterien als Kandidat für eine Implantation erfüllten als auch der Implantation zustimmten, bestanden aus kongenitaler Zytomegalovirusinfektion (cCMV; 16/37, 43 %), unbekannten Ursachen (6/37, 16 %), cochleovestibulären Anomalien und Traumata (je 5/37, 14 %).
Schlussfolgerung
Bei vielen Kindern mit SSD, die sich zur Voruntersuchung für eine uCI vorstellen, wurde letztlich keine uCI durchgeführt. Hauptfaktoren dafür sind die Aplasie des N. cochlearis und die nicht erteilte Zustimmung der Eltern zu der Intervention. Während annähernd die Hälfte der Kinder mit SSD in der untersuchten Kohorte Kandidaten für eine uCI waren, kam es nur bei einem Drittel der Gesamtkohorte zur Implantation; dabei waren die Hauptprädiktoren der Akzeptanz dieser Intervention eine Ätiologie (z. B. cCMV), bei der das Risiko einer progredienten Verschlechterung auf dem besser hörenden Ohr besteht, oder eine plötzlich aufgetretene SSD. Aus den vorliegenden Ergebnissen lassen sich wichtige Erkenntnisse über diese neue Population der Nutzer von Cochleaimplantaten und die sich entwickelnde Akzeptanz einer Intervention bei Kindern mit SSD ziehen.




Similar content being viewed by others

References
Gordon K, Henkin Y, Kral A (2015) Asymmetric hearing during development: The aural preference syndrome and treatment options. Pediatrics 136(1):141–153
Thomas JP, Neumann K, Dazert S, Voelter C (2017) Cochlear implantation in children with congenital single-sided deafness. Otol Neurotol 38(4):496–503
Arndt S, Laszig R, Aschendorff A, Hassepass F, Beck R, Wesarg T (2017) Cochlear implant treatment of patients with single-sided deafness or asymmetric hearing loss. HNO 65(Suppl 2):98–108. https://doi.org/10.1007/s00106-016-0297-5
Arndt S, Prosse S, Laszig R, Wesarg T, Aschendorff A, Hassepass F (2015) Cochlear implantation in children with single-sided deafness: Does aetiology and duration of deafness matter? Audiol Neurootol 20(Suppl 1):21–30
Dancer J, Burl NT, Waters S (1995) Effects of unilateral hearing loss on teacher responses to the SIFTER. Screening instrument for targeting educational risk. Am Ann Deaf 140(3):291–294
Morita S, Suzuki M, Iizuka K (2010) Non-organic hearing loss in childhood. Int J Pediatr Otorhinolaryngol 74(5):441–446
Sharma A, Dorman MF, Kral A (2005) The influence of a sensitive period on central auditory development in children with unilateral and bilateral cochlear implants. Hear Res 203(1–2):134–143
Newton V (2009) Paediatric audiological medicine, 2nd edn. Wiley, Hoboken
Sokolov M, Gordon KA, Polonenko M, Blaser SI, Papsin BC, Cushing SL (2019) Vestibular and balance function is often impaired in children with profound unilateral sensorineural hearing loss. Hear Res 372:52–61. https://doi.org/10.1016/j.heares.2018.03.032
Wolter NE, Cushing SL, Vilchez-Madrigal LD et al (2016) Unilateral hearing loss is associated with impaired balance in children: A pilot study. Otol Neurotol 37(10):1589–1595
Lieu JE (2004) Speech-language and educational consequences of unilateral hearing loss in children. Arch Otolaryngol Head Neck Surg 130(5):524–530
Lieu JE, Tye-Murray N, Fu Q (2012) Longitudinal study of children with unilateral hearing loss. Laryngoscope 122(9):2088–2095
Rachakonda T, Shimony JS, Coalson RS, Lieu JE (2014) Diffusion tensor imaging in children with unilateral hearing loss: A pilot study. Front Syst Neurosci 8:87
Schmithorst VJ, Holland SK, Ret J, Duggins A, Arjmand E, Greinwald J (2005) Cortical reorganization in children with unilateral sensorineural hearing loss. Neuroreport 16(5):463–467
Wang X, Fan Y, Zhao F et al (2014) Altered regional and circuit resting-state activity associated with unilateral hearing loss. PLoS ONE 9(5):e96126
Yang M, Chen HJ, Liu B et al (2014) Brain structural and functional alterations in patients with unilateral hearing loss. Hear Res 316:37–43
Haffey T, Fowler N, Anne S (2013) Evaluation of unilateral sensorineural hearing loss in the pediatric patient. Int J Pediatr Otorhinolaryngol 77(6):955–958
Arndt S, Aschendorff A, Laszig R et al (2011) Comparison of pseudobinaural hearing to real binaural hearing rehabilitation after cochlear implantation in patients with unilateral deafness and tinnitus. Otol Neurotol 32(1):39–47
Van de Heyning P, Vermeire K, Diebl M, Nopp P, Anderson I, De Ridder D (2008) Incapacitating unilateral tinnitus in single-sided deafness treated by cochlear implantation. Ann Otol Rhinol Laryngol 117(9):645–652
Vermeire K, Van de Heyning P (2009) Binaural hearing after cochlear implantation in subjects with unilateral sensorineural deafness and tinnitus. Audiol Neurootol 14(3):163–171
Buechner A, Brendel M, Saalfeld H, Litvak L, Frohne-Buechner C, Lenarz T (2010) Results of a pilot study with a signal enhancement algorithm for HiRes 120 cochlear implant users. Otol Neurotol 31(9):1386–1390
Firszt JB, Holden LK, Reeder RM, Cowdrey L, King S (2012) Cochlear implantation in adults with asymmetric hearing loss. Ear Hear 33(4):521–533
Stelzig Y, Jacob R, Mueller J (2011) Preliminary speech recognition results after cochlear implantation in patients with unilateral hearing loss: A case series. J Med Case Rep 5:343
Tavora-Vieira D, De Ceulaer G, Govaerts PJ, Rajan GP (2015) Cochlear implantation improves localization ability in patients with unilateral deafness. Ear Hear 36(3):e93–e98
Tavora-Vieira D, Marino R, Acharya A, Rajan GP (2015) The impact of cochlear implantation on speech understanding, subjective hearing performance, and tinnitus perception in patients with unilateral severe to profound hearing loss. Otol Neurotol 36(3):430–436
Gordon KA, Wong DD, Papsin BC (2013) Bilateral input protects the cortex from unilaterally-driven reorganization in children who are deaf. Brain 136(Pt 5):1609–1625
Hassepass F, Aschendorff A, Wesarg T et al (2013) Unilateral deafness in children: Audiologic and subjective assessment of hearing ability after cochlear implantation. Otol Neurotol 34(1):53–60
Tavora-Vieira D, Rajan GP (2015) Cochlear implantation in children with congenital and Noncongenital unilateral deafness. Otol Neurotol 36(8):1457–1458
Polonenko MJ, Papsin BC, Gordon KA (2017) Cortical activity in children with single-sided deafness pre- and post-implantation. Conference of the Association for Research in Otolaryngology 2017, Baltimore
Sokolov M, Cushing SL, Polonenko M, Blaser SI, Papsin BC, Gordon KA (2017) Clinical characteristics of children with single-sided deafness presenting for candidacy assessment for unilateral cochlear implantation. Curr Otorhinolaryngol Rep 5(4):275–285
Daya H, Figueirido JC, Gordon KA, Twitchell K, Gysin C, Papsin BC (1999) The role of a graded profile analysis in determining candidacy and outcome for cochlear implantation in children. Int J Pediatr Otorhinolaryngol 49(2):135–142
van der Knaap MS, Vermeulen G, Barkhof F, Hart AA, Loeber JG, Weel JF (2004) Pattern of white matter abnormalities at MR imaging: Use of polymerase chain reaction testing of Guthrie cards to link pattern with congenital cytomegalovirus infection. Radiology 230(2):529–536
Clemmens CS, Guidi J, Caroff A et al (2013) Unilateral cochlear nerve deficiency in children. Otolaryngol Head Neck Surg 149(2):318–325
Stehel EK, Shoup AG, Owen KE et al (2008) Newborn hearing screening and detection of congenital cytomegalovirus infection. Pediatrics 121(5):970–975
Grosse SD, Ross DS, Dollard SC (2008) Congenital cytomegalovirus (CMV) infection as a cause of permanent bilateral hearing loss: A quantitative assessment. J Clin Virol 41(2):57–62
Barbi M, Binda S, Caroppo S, Ambrosetti U, Corbetta C, Sergi P (2003) A wider role for congenital cytomegalovirus infection in sensorineural hearing loss. Pediatr Infect Dis J 22(1):39–42
Park AH, Duval M, McVicar S, Bale JF, Hohler N, Carey JC (2014) A diagnostic paradigm including cytomegalovirus testing for idiopathic pediatric sensorineural hearing loss. Laryngoscope 124(11):2624–2629
Dahle AJ, Fowler KB, Wright JD, Boppana SB, Britt WJ, Pass RF (2000) Longitudinal investigation of hearing disorders in children with congenital cytomegalovirus. J Am Acad Audiol 11(5):283–290
Lanzieri TM, Chung W, Flores M et al (2017) Hearing loss in children with asymptomatic congenital cytomegalovirus infection. Pediatrics 139(3):e20162610
Carlsson PI, Hall M, Lind KJ, Danermark B (2011) Quality of life, psychosocial consequences, and audiological rehabilitation after sudden sensorineural hearing loss. Int J Audiol 50(2):139–144
Noguchi Y, Takahashi M, Ito T, Fujikawa T, Kawashima Y, Kitamura K (2016) Delayed restoration of maximum speech discrimination scores in patients with idiopathic sudden sensorineural hearing loss. Auris Nasus Larynx 43(5):495–500
Li LP, Shiao AS, Chen KC et al (2012) Neuromagnetic index of hemispheric asymmetry prognosticating the outcome of sudden hearing loss. PLoS ONE 7(4):e35055
Polonenko MJ, Papsin BC, Gordon KA (2017) Children with single-sided deafness use their cochlear implant. Ear Hear 38(6):681–689. https://doi.org/10.1097/AUD.0000000000000452
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
S.L. Cushing: Speaker’s Bureau Cochlear Corporation. Oticon, Interacoustics. K.A. Gordon and B.C. Papsin: Speaker’s Bureau Cochlear Corporation. M. Sokolov, V. Papaioannou and M. Polonenko declare that they have no competing interests.
For this article no studies with human participants or animals were performed by any of the authors. All studies performed were in accordance with the ethical standards indicated in each case.
Rights and permissions
About this article
Cite this article
Cushing, S.L., Gordon, K.A., Sokolov, M. et al. Etiology and therapy indication for cochlear implantation in children with single-sided deafness. HNO 67, 750–759 (2019). https://doi.org/10.1007/s00106-019-00729-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00106-019-00729-8
Keywords
- Unilateral deafness
- Cochlear nerve aplasia
- Cochlear nerve hypoplasia
- Cytomegalovirus
- Pediatrics
- Hearing loss