Abstract
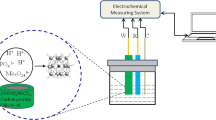
A zeolite-modified carbon paste electrode (CPE) has been used for the determination of 4-nitrophenol by differential pulse voltammetry (DPV). The electrochemical reduction of 4-nitrophenol at −1.0 V is carried out in a Britton-Robinson medium at pH 3.5. The cyclic voltammetric (CV) behaviour has been investigated to study the nature of the process. Studies on the effect of pH were carried out over the pH range 2–9 with the Britton-Robinson buffer solution, and the influence of pH on peak height and peak potential was analyzed. A linear relationship between peak intensity and concentration is obtained in the range 0.2–10 mg L−1, with a detection limit of 0.04 mg L−1; a relative standard deviation of 1.5% for a 5 mg L−1 4-nitrophenol concentration and a relative error of 2.6% were also obtained (n=11).
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received March 3, 1998. Revision December 10, 1998.
Rights and permissions
About this article
Cite this article
del Mar Cordero-Rando, M., Barea-Zamora, M., Barberá-Salvador, J. et al. Electrochemical Study of 4-Nitrophenol at a Modified Carbon Paste Electrode. Mikrochim Acta 132, 7–11 (1999). https://doi.org/10.1007/PL00010076
Issue Date:
DOI: https://doi.org/10.1007/PL00010076