Abstract.
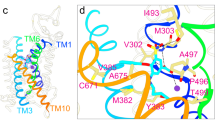
Recombinant protein, designated UAT, prepared from a cloned rat renal cDNA library functions as a selective voltage-sensitive urate transporter/channel when fused with lipid bilayers. Since we previously suggested that UAT may represent the mammalian electrogenic urate transporter, UAT has been functionally characterized in the presence and absence of potential channel blockers, several of which are known to block mammalian electrogenic urate transport. Two substrates, oxonate (a competitive uricase inhibitor) and pyrazinoate, that inhibit renal electrogenic urate transport also block UAT activity. Of note, oxonate selectively blocks from the cytoplasmic side of the channel while pyrazinoate only blocks from the channel's extracellular face. Like oxonate, anti-uricase (an electrogenic transport inhibitor) also selectively blocks channel activity from the cytoplasmic side. Adenosine blocks from the extracellular side exclusively while xanthine blocks from both sides. These effects are consistent with newly identified regions of homology to uricase and the adenosine A1/A3 receptor in UAT and localize these homologous regions to the cytoplasmic and extracellular faces of UAT, respectively. Additionally, computer analyses identified four putative α-helical transmembrane domains, two β sheets, and blocks of homology to the E and B loops of aquaporin-1 within UAT. The experimental observations substantiate our proposal that UAT is the molecular representation of the renal electrogenic urate transporter and, in conjunction with computer algorithms, suggest a possible molecular structure for this unique channel.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 13 October 1998/Revised: 28 January 1999
Rights and permissions
About this article
Cite this article
Leal-Pinto, E., Cohen, B. & Abramson, R. Functional Analysis and Molecular Modeling of a Cloned Urate Transporter/Channel. J. Membrane Biol. 169, 13–27 (1999). https://doi.org/10.1007/PL00005897
Issue Date:
DOI: https://doi.org/10.1007/PL00005897