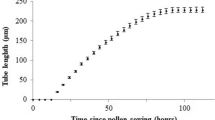
Summary
Pollen tubes grow by tip growth, with high levels of exocytosis at the apex. The commercial availability of FITC labelled α-linked dextrans provides a source of biologically inert tracers for endocytotic activity in pollen tubes. Growing tubes ofNicotiana andTradescantia were transferred to media containing 1% FD-4 for varying period of time before washing in control media and observation in a fluorescence microscope. Fluorescent material appeared to enter the pollen tubes only at the tip region, and to accumulate in vacuoles, starting with smaller vacuoles near the tip and spreading to the main vacuolated part of the tube. Mature tubes, with callose plugs, were only labelled up to the first complete plug from the tip, younger tubes without plugs were labelled into the pollen grain vacuole. The fluorescent material within the pollen tubes was shown to represent uptake of intact high molecular weight dextran by the following criteria: (i) free FITC and low molecular weight dextrans could not be detected in any of the media or pollen tubes using thin layer chromatography and (ii) pollen tube growth rates were unaffected by the fluorescent dextran, but were severely inhibited by low levels of free FITC. It was concluded that the dextrans entered the tubes by endocytosis, possibly in the tip region, and were then transferred to the vacuole system of the pollen tube.
Similar content being viewed by others
Abbreviations
- FITC:
-
fluorescein isothiocyanate
- FD:
-
fluorescent dextran
References
Cole L, Coleman J, Evans D, Hawes C (1990) Internalisation of fluorescein isothiocyanate-dextran by suspension-cultured plant cells. J Cell Sci 96: 721–730
— —, Kearns A, Morgan J, Hawes C (1991) The organic anion transport inhibitor, probenecid, inhibits the transport of Lucifer Yellow at the plasma membrane and at the tonoplast in suspension cultured plant cells. J Cell Sci 99: 545–555
Cornish EC, Pettitt JM, Bonig I, Clarke AE (1987) Developmentally controlled expression of a gene associated with self-incompatibility inNicotiana alata. Nature 326: 99–102
Derksen J, Pierson ES, Traas JA (1985) Microtubules in vegetative and generative cells of pollen tubes. Eur J Cell Biol 38: 142–148
- Rougen G, Rutten TLM, De Win AHN, Knuiman B, Sassen MMA, Lichtscheidl IK, Pierson ES (1993) Organelle distribution, exocytosis and endocytosis in pollen tubes. Planta (in press)
Fowke LC, Tanchak MA, Galway ME (1991) Ultrastructural cytology of the endocytotic pathway in plants. In: Hawes CR, Coleman JOD, Evans DE (eds) Endocytosis, exocytosis and vesicle traffic in plants. Cambridge University Press, Cambridge, pp 15–40
Gray JE, McClure BA, Bonig I, Anderson MA, Clarke AE (1991) Action of the style product of the self-incompatibility gene ofNicotiana alata (S-RNase) on in vitro-grown pollen tubes. Plant Cell 3: 271–283
Hann C (1991) Uptake of fluorescent dextran by endocytosis in pollen tubes ofTradescantia virginiana and in cells ofMorinda citrifolia. Thesis, University College Dublin, Dublin
Jahnen W, Batterham MP, Clarke AE, Moritz RL, Simpson RJ (1989 a) Identification, isolation, and N-terminal sequencing of style glycoproteins associated with self-incompatibility inNicotiana alata. Plant Cell 1: 493–499
—, Lush WM, Clarke AE (1989 b) Inhibition of in vitro pollen tube growth by isolated S-glycoproteins ofNicotiana alata. Plant Cell 1: 501–510
McClure BA, Gray JE, Anderson MA, Clarke AE (1990) Self-incompatibility inNicotiana alata involves degradation of pollen rRNA. Nature 347: 757–760
O'Driscoll D, Steer MW (1990) The development of a marker for estimating the rate of endocytosis in plant cells. Cell Biol Int Rep 14: 981–988
—, Wilson G, Steer MW (1991) Lucifer Yellow and fluorescein isothiocyanate uptake by cells ofMorinda citrifolia in suspension cultures is not confined to the endocytotic pathway. J Cell Sci 100: 237–241
- Read SM, Steer MW (1993) Determination of cell wall porosity by microscopy: walls of cultured cells and pollen tubes. Acta Bot Neerl (in press)
Oparka KJ, Murant EA, Wright KM, Prior DAM (1991) The drug probenecid inhibits the vacuolar accumulation of fluorescent anions in onion epidermal cells. J Cell Sci 99: 557–563
Picton JM, Steer MW (1983) Membrane recycling and the control of secretory activity in pollen tubes. J Cell Sci 63: 303–310
— — (1985) The effects of ruthenium red, lanthanum, fluorescein isothiocyanate and trifluoperazine on vesicle transport, vesicle fusion and tip extension in pollen tubes. Planta 163: 20–26
Preston RA, Murphy RF, Jones EW (1987) Apparent endocytosis of fluorescein isothiocyanate-conjugated dextran bySaccharomyces cerevisiae reflects uptake of low molecular weight impurities, not dextran. J Cell Biol 105: 1981–1987
Steer JM, Steer MW (1989) Pollen tube tip growth. New Phytol 111: 323–358
Steer MW (1988) Plasma membrane turnover in plant cells. J Exp Bot 39: 987–996
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
O'Driscoll, D., Hann, C., Read, S.M. et al. Endocytotic uptake of fluorescent dextrans by pollen tubes grown in vitro. Protoplasma 175, 126–130 (1993). https://doi.org/10.1007/BF01385010
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01385010