Abstract
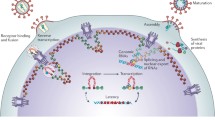
As a member of the Retrovirus family, human immunodeficiency virus (HIV), a causative agent of AIDS, replicates by integrating its genome into the host cell’s nuclear DNA. However, in contrast to most retroviruses that depend on mitotic dissolution of the nuclear envelope to gain access to the host cell’s genome, the HIV pre-integration complex can enter the nucleus of the target cell during the interphase. Such capacity greatly enhances HIV replication and allows the virus to productively infect terminally differentiated nonproliferating cells, such as macrophages. Infection of macrophages is a critical factor in the pathogenesis of diseases caused by HIV-1 and other lentiviruses. The mechanisms responsible for this unusual feature of HIV have enticed researchers since the early 90s, when the first characterization of the HIV-1 pre-integration complex was reported. Several viral factors, including matrix protein, integrase, viral protein R, and central DNA flap, have been proposed as regulators of HIV-1 nuclear import, only to be later shown as nonessential for this process. As a result, after more than a decade of intense research, there is still no consensus on which HIV-1 and cellular proteins control this critical step in HIV-1 replication. In this review, we will discuss recent advances and suggest possible solutions to the controversial issue of HIV-1 nuclear import.

Similar content being viewed by others
References
Bukrinsky MI, Haggerty S, Dempsey MP, Sharova N, Adzhubel A, Spitz L, Lewis P, Goldfarb D, Emerman M, and Stevenson M. (1993) A nuclear localization signal within HIV-1 matrix protein that governs infection of non-dividing cells. Nature 365:666–9.
Lewis PF, Emerman M. (1994) Passage through mitosis is required for oncoretro-viruses but not for the human immunodeficiency virus. J. Virol. 68:510–6.
Weinberg JB, Matthews TJ, Cullen BR, Malim MH. (1991) Productive human immunodeficiency virus type 1 (HIV-1) infection of nonproliferating human monocytes. J. Exp. Med. 174:1477–82.
Bukrinsky MI, Sharova N, Dempsey MP, Stanwick TL, Bukrinskaya AG, Haggerty S, Stevenson M. (1992) Active nuclear import of human immunodeficiency virus type 1 preintegration complexes. Proc. Natl. Acad. Sci. U.S.A. 89:6580–4.
Lewis P, Hensel M, Emerman M. (1992) Human immunodeficiency virus infection of cells arrested in the cell cycle. EMBO J. 11:3053–8.
Li G, Simm M, Potash MJ, Volsky DJ. (1993) Human immunodeficiency virus type 1 DNA synthesis, integration, and efficient viral replication in growth-arrested T cells. J. Virol. 67:3969–77.
Gallay P, Swingler S, Aiken C, Trono D. (1995) HIV-1 infection of nondividing cells: C-terminal tyrosine phosphorylation of the viral matrix protein is a key regulator. Cell 80:379–88.
Roe T, Reynolds TC, Yu G, Brown PO. (1993) Integration of murine leukemia virus DNA depends on mitosis. EMBO J. 12:2099–108.
Briggs JA, Wilk T, Welker R, Krausslich HG, Fuller SD. 2003. Structural organization of authentic, mature HIV-1 virions and cores. EMBO J. 22:1707–15.
Bukrinsky MI, Sharova N, McDonald TL, Pushkarskaya T, Tarpley WG, Stevenson M. (1993) Association of integrase, matrix, and reverse transcriptase antigens of human immunodeficiency virus type 1 with viral nucleic acids following acute infection. Proc. Natl. Acad. Sci. U.S.A. 90:6125–9.
Fassati A, Goff SP. (2001) Characterization of intracellular reverse transcription complexes of human immunodeficiency virus type 1. J. Virol. 75:3626–35.
Bowerman B, Brown PO, Bishop JM, Varmus HE. (1989) A nucleoprotein complex mediates the integration of retroviral DNA. Genes Dev. 3:469–78.
Miller MD, Farnet CM, Bushman FD. 1997. Human immunodeficiency virus type 1 preintegration complexes: studies of organization and composition. J. Virol. 71:5382–90.
Bukrinskaya A, Brichacek B, Mann A, Stevenson M. (1998) Establishment of a functional human immunodeficiency virus type 1 (HIV-1) reverse transcription complex involves the cytoskeleton. J. Exp. Med. 188:2113–25.
McDonald D, Vodicka MA, Lucero G, Svitkina TM, Borisy GG, Emerman M, Hope TJ. (2002) Visualization of the intracellular behavior of HIV in living cells. J. Cell Biol. 159:441–52.
Taunton J. (2001) Actin filament nucleation by endosomes, lysosomes and secretory vesicles. Curr. Opin. Cell Biol. 13:85–91.
Sodeik B, Ebersold MW, Helenius A. (1997) Microtubule-mediated transport of incoming herpes simplex virus 1 capsids to the nucleus. J. Cell Biol. 136:1007–21.
Suomalainen M, Nakano MY, Keller S, Boucke K, Stidwill RP, Greber UF. (1999) Microtubule-dependent plus- and minus end-directed motilities are competing processes for nuclear targeting of adenovirus. J. Cell Biol. 144:657–72.
de Noronha CM, Sherman MP, Lin HW, Cavrois MV, Moir RD, Goldman RD, Greene WC. (2001) Dynamic disruptions in nuclear envelope architecture and integrity induced by HIV-1 Vpr. Science 294:1105–8.
Gupta K, Ott D, Hope TJ, Siliciano RF, Boeke JD. (2000) A human nuclear shuttling protein that interacts with human immunodeficiency virus type 1 matrix is packaged into virions. J. Virol. 74:11811–24.
Zennou V, Petit C, Guetard D, Nerhbass U, Montagnier L, Charneau P. (2000) HIV-1 genome nuclear import is mediated by a central DNA flap. Cell 101: 173–85.
Sirven A, Pflumio F, Zennou V, Titeux M, Vainchenker W, Coulombel L, Dubart-Kupperschmitt A, Charneau P. (2000) The human immunodeficiency virus type-1 central DNA flap is a crucial determinant for lentiviral vector nuclear import and gene transduction of human hematopoietic stem cells. Blood 96:4103–10.
Van Maele B, De Rijck J, De Clercq E, Debyser Z. (2003) Impact of the central polypurine tract on the kinetics of human immunodeficiency virus type 1 vector transduction. J. Virol. 77:4685–94.
Dvorin JD, Bell P, Maul GG, Yamashita M, Emerman M, Malim MH. (2002) Reassessment of the roles of integrase and the central DNA flap in human immunodeficiency virus type 1 nuclear import. J. Virol. 76:12087–96.
Limon A, Nakajima N, Lu R, Ghory HZ, Engelman A. (2002) Wild-type levels of nuclear localization and human immunodeficiency virus type 1 replication in the absence of the central DNA flap. J. Virol. 76:12078–86.
von Schwedler U, Kornbluth RS, Trono D. (1994) The nuclear localization signal of the matrix protein of human immunodeficiency virus type 1 allows the establishment of infection in macrophages and quiescent T lymphocytes. Proc. Natl. Acad. Sci. U.S.A. 91:6992–6.
Nadler SG, Tritschler D, Haffar OK, Blake J, Bruce AG, Cleaveland JS. (1997) Differential expression and sequence-specific interaction of karyopherin alpha with nuclear localization sequences. J. Biol. Chem. 272:4310–5.
Dingwall C, Laskey RA. 1991. Nuclear targeting sequences—a consensus? Trends Biochem. Sci. 16:478–81.
Haffar OK, Popov S, Dubrovsky L, Agostini I, Tang H, Pushkarsky T, Nadler SG, Bukrinsky M. (2000) Two nuclear localization signals in the HIV-1 matrix protein regulate nuclear import of the HIV-1 pre-integration complex. J. Mol. Biol. 299:359–68.
Mattaj IW, Englmeier L. (1998) Nucleocytoplasmic transport: the soluble phase. Annu. Rev. Biochem. 67:265–306.
Rexach M, Blobel G. (1995) Protein import into nuclei: association and dissociation reactions involving transport substrate, transport factors, and nucleoporins. Cell 83:683–92.
Gallay P, Stitt V, Mundy C, Oettinger M, Trono D. (1996) Role of the karyopherin pathway in human immunodeficiency virus type 1 nuclear import. J. Virol. 70:1027–32.
Fouchier RA, Meyer BE, Simon JH, Fischer U, Malim MH. (1997) HIV-1 infection of non-dividing cells: evidence that the amino-terminal basic region of the viral matrix protein is important for Gag processing but not for post-entry nuclear import. EMBO J. 16:4531–9.
Freed EO, Englund G, Martin MA. (1995) Role of the basic domain of human immunodeficiency virus type 1 matrix in macrophage infection. J. Virol. 69:3949–54.
Reil H, Bukovsky AA, Gelderblom HR, Gottlinger HG. 1998. Efficient HIV-1 replication can occur in the absence of the viral matrix protein. EMBO J. 17:2699–708.
Gallay P, Hope T, Chin D, Trono D. (1997) HIV-1 infection of nondividing cells through the recognition of integrase by the importin/karyopherin pathway. Proc. Natl. Acad. Sci. U.S.A. 94:9825–30.
Bouyac-Bertoia M, Dvorin JD, Fouchier RA, Jenkins Y, Meyer BE, Wu LI, Emerman M, Malim MH. 2001. HIV-1 infection requires a functional integrase nls. Mol. Cell 7:1025–35.
Limon A, Devroe E, Lu R, Ghory HZ, Silver PA, Engelman A. 2002. Nuclear localization of human immunodeficiency virus type 1 preintegration complexes (PICs): V165A and R166A are pleiotropic integrase mutants primarily defective for integration, not PIC nuclear import. J. Virol. 76:10598–607.
Fassati A, Gorlich D, Harrison I, Zaytseva L, Mingot JM. (2003) Nuclear import of HIV-1 intracellular reverse transcription complexes is mediated by importin 7. EMBO J. 22:3675–85.
Jakel S, Gorlich D. 1998. Importin beta, transportin, RanBP5, and RanBP7 mediate nuclear import of ribosomal proteins in mammalian cells. EMBO J. 17:4491–502.
Heinzinger NK et al. (1994) The Vpr protein of human immunodeficiency virus type 1 influences nuclear localization of viral nucleic acids in nondividing host cells. Proc. Natl. Acad. Sci. U.S.A. 91:7311–5.
Emerman M, Bukrinsky M, Stevenson M. (1994) HIV-1 infection of non-dividing cells. Nature 369:107–8.
Dahl K, Martin K, Miller G. (1987) Differences among human immunodeficiency virus strains in their capacities to induce cytolysis or persistent infection of a lymphoblastoid cell line immortalized by Epstein-Barr virus. J. Virol. 61:1602–8.
Stewart SA, Poon B, Jowett JB, Chen IS. (1997) Human immunodeficiency virus type 1 Vpr induces apoptosis following cell cycle arrest. J. Virol. 71:5579–92.
Connor RI, Chen BK, Choe S, Landau NR. (1995) Vpr is required for efficient replication of human immunodeficiency virus type-1 in mononuclear phagocytes. Virology 206:935–44.
Nie Z, Bergeron D, Subbramanian RA, Yao XJ, Checroune F, Rougeau N, Cohen EA. (1998) The putative alpha helix 2 of human immunodeficiency virus type 1 Vpr contains a determinant which is responsible for the nuclear translocation of proviral DNA in growth-arrested cells. J. Virol. 72:4104–15.
Jenkins Y, McEntee M, Weis K, Greene WC. (1998) Characterization of HIV-1 Vpr nuclear import: analysis of signals and pathways. J. Cell Biol. 143:875–85.
Popov S, Rexach M, Ratner L, Blobel G, Bukrinsky M. (1998) Viral protein R regulates docking of the HIV-1 preintegration complex to the nuclear pore complex. J. Biol. Chem. 273:13347–52.
Popov S, Rexach M, Zybarth G, Reiling N, Lee MA, Ratner L, Lane CM, Moore MS, Blobel G, Bukrinsky M. (1998) Viral protein R regulates nuclear import of the HIV-1 pre-integration complex. EMBO J. 17:909–17.
Vodicka MA, Koepp DM, Silver PA, Emerman M. (1998) HIV-1 Vpr interacts with the nuclear transport pathway to promote macrophage infection. Genes Dev. 12:175–85.
Fouchier RA et al. (1998) Interaction of the human immunodeficiency virus type 1 Vpr protein with the nuclear pore complex. J. Virol. 72:6004–13.
Le Rouzic E et al. (2002) Docking of HIV-1 Vpr to the nuclear envelope is mediated by the interaction with the nucleoporin hCG1. J. Biol. Chem. 277:45091–8.
Weiss RA. (2003) HIV and AIDS: looking ahead. Nat. Med. 9:887–91.
Turelli P et al. (2001) Cytoplasmic recruitment of INI1 and PML on incoming HIV preintegration complexes: interference with early steps of viral replication. Mol. Cell 7:1245–54.
Nermut MV, Fassati A. (2003) Structural analyses of purified human immunodeficiency virus type 1 intracellular reverse transcription complexes. J. Virol. 77:8196–206.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bukrinsky, M. A Hard Way to the Nucleus. Mol Med 10, 1–5 (2004). https://doi.org/10.1007/BF03401996
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03401996