Abstract
Background
Lysozyme (LZ), a host-defense protein, contains an 18 amino-acid domain with high affinity binding for sugar-derived proteins or lipids, called advanced glycation endproducts (AGE), that are implicated in diabetes-and age-dependent complications (DC).
Materials and Methods
A) The effects of LZ on AGE-removal were tested in vivo. LZ was injected (200 ug/day, i.p., ×2 weeks) in non-obese diabetic (NOD), db/db (+/+) mice, and non-diabetic, AGE-infused Sprague-Dawley rats. B) LZ: AGE interactions with macrophage-like T1B-183 cells (Mf) and mesangial cells (MC) were tested in vitro.
Results
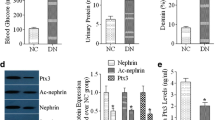
A) In NOD mice, LZ reduced the elevated basal serum AGE (sAGE) (p < 0.05), enhanced urinary AGE (uAGE) excretion by ~2-fold (p < 0.01), while it reduced albuminuria (UA), p < 0.005. In db/db mice, LZ infusion also reduced the elevated sAGE (p < 0.05), doubled uAGE excretion (p < 0.05), and decreased UA (p < 0.01). In addition, LZ maintained normal sAGE in normal rats infused with AGE-BSA, as it doubled the urinary AGE (uAGE) clearance (p < 0.01). B) LZ stimulated the uptake and degradation of 125I-labeled AGE-BSA and 25I-human serum AGE by Mf, while suppressing AGE-induced TNFα and IGF-I production. In MC, LZ suppressed the AGE-promoted PDGF-B, α1 type IV collagen, and tenascin mRNA levels, and restored the AGE-suppressed expression and activity of MMP-9, but not MMP-2.
Conclusion
LZ may act to: a) accelerate renal in-vivo AGE clearance, b) suppress macrophage and mesangial cell-specific gene activation in vitro, and c) improve albuminuria due to diabetes. These data suggest that LZ by sequestering AGEs may protect against diabetic renal damage.





Similar content being viewed by others
References
Baynes JW. (1994) Ageing growth factors; a role in diabetic vascular disease? J. Clin. Invest. 94: 2.
Bucala R, Vlassara H, Cerami A. (1992) Advanced glycosylation endproducts. In: Post-translational Modifications of Proteins. J.J. Harding and M.J.C. Crabbe, eds., CRC Press, Inc, Boca Raton, FL., pp: 53–59.
Schmidt AM, Yan SD, Wautier J-L, Stern D. (1999) Activation of receptor for advanced glycation endproducts: a mechanism for chronic vascular dysfunction in diabetic vasculopathy and atherosclerosis. Circulation Res. 84: 489–497.
Baynes JW. (1991) Role of oxidative stress in development of complications in diabetes. Diabetes 40: 405–12.
Makita Z, Bucala R, Rayfield EJ, et al. (1994) Reactive glycosylation endproducts in diabetic uraemia and treatment of renal failure. Lancet 343: 1519–1522.
Weiss MF, Ehrhard P, Kader-Attia FA, et al. (2000) Mechanisms for the formation of glycoxidation products in end-stage renal disease. Kidney International 57: 2571–2584.
Ankrah N-A, Appiah-Opong R. (1999) Toxicity of low levels of methylglyoxal: depletion of blood glutathione and adverse effects on glucose tolerance in mice. Toxicology Letters 109: 61–67.
Odani H, Shinzato T, Usami J, et al. (1998) Imidazolium crosslinks derived from reaction of lysine with glyoxal and methylglyoxal are increased in serum proteins of uremic patients: evidence for increased oxidative stress in uremia. FEBS Letters 427: 381–385.
Vlassara H, Bucala R, Striker L. (1994) Pathogenic effects of advance glycosylation: biochemical, biologic, and clinical implications for diabetes and aging. Lab. Invest. 70: 138–151.
Witko-Sarsat V, Friedlander M, Nguyen Khoa T, et al. (1998) Advanced oxidation protein products as novel mediators of inflammation and monocyte activation in chronic renal failure. J. Immunol. 161: 2524–2532.
Brownlee M, Vlassara H, Kooney A, et al. (1986) Aminoguanidine prevents diabetes-induced arterial wall protein cross-linking. Science 232: 1629–1632.
Vasan S, Zhang X, Zhang X, et al. (1996) An agent cleaving glucose-derived protein crosslinks in vitro and iin vivo. Nature 382: 275–278.
Nakamura S, Makita Z, Ishikawa S. (1997) Progression of nephropathy in spontaneous diabetic rats is prevented by OPB-9195, a novel inhibitor of advanced glycation. Diabetes 46: 895–899.
He CJ, Li J, Steffes MHM, et al. (1999) Low AGE-diet ameliorates nephropathy in non-obese diabetic (NOD) mice. Diabetes Supplement 1 48: A144.
Zheng F, He C, Li J, Vlassara H. (2000) Retriction of AGE content of food, without lowering protein intake prevents diabetic nephropathy (DN) in mice. Diabetes Supplement I 49: A161.
Sava G. (1996) Pharmacological aspects and therapeutic applications of lysozymes. In: Lysozymes: Model Enzymes in Biochemistry and Biology. P. Jolies, ed., Birkhauser Verlag Basel, Switzerland, pp: 433–449.
Li YM, Tan AX, Vlassara H. (1995) Antibacterial activity of lysozyme and lactoferrin is inhibited by binding of advanced glycation-modified proteins to a conserved motif. Nat. Med. 1: 1057–1061.
Mitsuhashi T, Li YM, Fishbane S, Vlassara H. (1997) Depletion of reactive advanced glycation endproducts from diabetic uremic sera by a lysozyme-linked matrix. J. Clin. Invest. 100: 847–854.
Makita Z, Vlassara H, Cerami A, Bucala R. (1992) Immuno-chemical detection of advanced glycosylation end products in vivo. J. Biol. Chem. 267: 5133–5138.
Mitsuhashi T, Vlassara H, Founds H, Li YM. (1997) Standardizing the Immunological easurement of Advanced Glycation Endproducts Using Normal Human Serum. J. Immunol. Methods 207: 79–88.
Lo TW, Westwood ME, McLellan AC, et al. (1994) Binding and modification of proteins by methylglyoxal under physiological conditions a kinetic and mechanistic study with N-alpha-acetylarginine, N-alpha-acetylcysteine, and N-alpha-acetyllysine, and bovine serum albumin. J. Biol. Chem. 269: 32299–32305.
Ahmed MU, Brinkmann Frye E, Degenhardt TP, et al. (1997) εN-(Carboxyethyl)lysine, a product of the chemical modification of proteins by methylglyoxal, increases with age in human lens proteins. Biochem. J. 324: 565–570.
Li YM, Steffes M, Donnelly T, et al. (1996) Prevention of cardiovascular and renal pathology of aging by the advanced glycation inhibitor aminoguanidine. Proc. Natl. Acad. Sci. USA 93: 3902–3907.
He C-J, Zheng F, Sabol J, et al. (2000) Differential expression and function of advanced glycation endproduct (AGE)-receptor genes in NOD mouse kidneys; possible role in diabetic and non-diabetic renal disease. Kidney Int. 58: 1931–1940.
Li JJ, Dickson D, Hof PR, et al. (1998) Receptors for advanced glycosylation endproducts in human brain: Role in brain homeostasis. Mol. Med. 4: 46–60.
Doi T, Vlassara H, Kirstein M, et al. (1992) Receptor-specific increase in extracellular matrix production in mouse mesangial cells by advanced glycosylation end products is mediated via Platelet-Derived Growth Factor. Proc. Natl. Acad. Sci. USA 89: 2873–2877.
Skolnick EY, Yang Z, Makita Z, et al. (1991) Human and rat mesangial cell receptors for glucose-modified proteins: potential role in kidney tissue remodelling and diabetic nephropathy. J. Exp. Med. 174: 931–939.
Elliot S, Striker L, Hattori M, et al. (1993) Mesangial cells from diabetic NOD mice constitutively secrete increased amounts of insulin-like growth factor-I. Endocr. J. 133: 1783–1788.
Peten EP, Striker L, Garcia-Perez A, et al. (1993) Studies by competitive PCR of glomerulosclerosis in growth hormone transgenic mice. Kidney Int. 43: S55–S58.
Lenz O, Eliot SE, Stetler-Stevenson WG. (2000) Matrix met-alloproteinases in renal development. J. Am. Soc. Nephrol. 11: 574–581.
Jacot TA, Striker GE, Stetler-Stevenson M, et al. (1996) Mesangial cells from transgenic mice with progressive glomerulosclerosis exhibit stable, phenotypic changes including undetectable MMP-9 and increased type IV collagen. Lab. Invest. 75: 791–790.
Ziyadeh FN, Hoffman BB, Han DC, et al. (2000) Long-term prevention of renal insufficiency, excess matrix gene expression, and glomerular mesangial matrix expansion by treatment with monoclonal-TGFβ antibody in db/db diabetic mice. Proc. Natl. Acad. Sci. USA 97: 8015–8020.
Gugliucci A, Bendayan M. (1996) Renal fate of circulating advanced glycation end products (AGE): evidence for reabsorp-tion and catabolism of AGE peptides by renal proximal tubular cells. Diabetologia 39: 149–160.
Gugliucci A, Bendayan M. (1995) Reaction of advanced glycation endproducts with renal tissue from normal and streptozotocin-induced diabetic rats: an ultrastructural study using colloidal gold cytochemistry. J. Histochem. Cytochem. 43: 591–600.
Haas M, De Zeeuw D, van Zanten A, Meijer DKF. (1993) Quantification of renal low-molecular-weights protein handling in the intact rat. Kidney Int. 43: 949–954.
Haas, Kluppel ACA, Wartna ES, et al. (1997) Drug-targeting to the kidney: Renal delivery and degradation of a naproxenlysozyme conjugate in vivo. Kidney Int. 52: 1693–1699.
Kislinger T, Fu C, Huber B, et al. (1999) εN-(Carboxymethyl) lysine adducts of proteins are ligands for receptor for advanced glycation end products that activate cell signaling pathways and modulate gene expression. J. Biol. Chem. 274: 31740–31749.
Vlassara H, Brownlee M, Cerami A. (1986) Novel macrophage receptor for glucose-modified proteins is distinct from previously described scavenger receptors. J. Exp. Med. 164: 1301–1309.
Vlassara H, Bucala R, Stitt, A. (1998). Vascular complications of diabetes and the role of advanced glycosylation end products. In: An Introduction to Vascular Biology. A. Halliday, et al., eds., Cambridge University Press, UK, pp: 173–194.
Vlassara H, Brownlee M, Manogue KR, et al. (1988) Cachectin/tnf and IL-1 induced by glucose-modified proteins: role in normal tissue remodeling. Science 240: 1546–1548.
Thornalley PJ. (1996) Advanced glycation and the development of diabetic complications. Unifying the involvement of glucose, methylglyoxal and oxidative stress. Endocrinol. & Metabolism 3: 149–166.
Shinohara M, Thornalley PJ, Giardino I, et al. (1998) Over-expression of glyoxalase-I in bovine endothelial cells inhibits intracellular advanced glycation endproduct formation and prevents hyperglycemia-induced increases in macromolecular endocytosis. J. Clin. Invest. 101(5): 1142–1147.
Yang CW, Vlassara H, Striker GE, et al. (1995) Administration of AGEs in vivo induces genes implicated in diabetic glomerulosclerosis. Kidney. Int. Suppl. 49: 55–58.
Takada K, Ohno N, Yadomae T. (1994) Binding of lysozyme to lipopolysaccharide suppresses-tumor necrosis factor production in vivo. Infection and Immunity 62: 1171–1175.
Lee-Huang S, Huang PL, Sun Y, et al. (1999) Lysozyme and RNAses as anti-HIV components in β-core preparations of human chorionic gonadotropin. Proc. Natl. Acad. Sci, USA 96: 2678–2681.
Chen F, Crandall J, Zhu L, et al. (2000) Overexpression of human recombinant lysozyme (huLZ) by macrophages leads to enhanced glycotoxin uptake and clearance without cell activation: A new model approach for the prevention of diabetic complications. Diabetes Supplement 220: A54.
Park LKG, Raman KG, Lee Y, et al. (1998) Suppression of accelerated diabetic atherosclerosis by the soluble receptor for advanced glycation endproducts. Nat. Med. 4: 1025–1031.
Acknowledgments
This work was supported in part by an NIH grant (DK57126) and a program project grant from the Juvenile Diabetes Research Foundation (JDRF-4-1999-697). We thank Ms. Ina Katz for invaluable editorial assistance and Dr. Michael A. Yamin for critically reviewing this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Contributed by R. Bucala.
Rights and permissions
About this article
Cite this article
Zheng, F., Cai, W., Mitsuhashi, T. et al. Lysozyme Enhances Renal Excretion of Advanced Glycation Endproducts In Vivo and Suppresses Adverse AGE-mediated Cellular Effects In Vitro: A Potential AGE Sequestration Therapy for Diabetic Nephropathy?. Mol Med 7, 737–747 (2001). https://doi.org/10.1007/BF03401963
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03401963