Abstract
Background
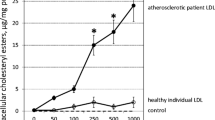
In the course of atherosclerosis, humans and apolipoprotein (apoE) Knockout (KO) mice exhibit an active cell-mediated and humoral immune process, both at the systemic level and within atheromata. Low density lipoproteins (LDL) infiltrate the vascular wall, where they are oxidatively modified. This oxidative modification may generate new epitopes for which tolerance is not achieved during ontogenesis. Such epitopes could constitute new targets for autoreactive immune responses that may have a physiopathological role in disease development.
Materials and Methods
Exposing mice to high dose of antigens during thymic T-cell education induces immunological tolerance to the administered antigens. We injected newborn apoE KO mice with oxidized LDL. They were fed a cholesterol-rich diet and aortic atherosclerosis, cell-mediated immune response, and T-cell repertoire were analyzed after 5 months.
Results
Injection of oxidized LDL at birth reduced not only the immune response to oxidized LDL, but also susceptibility to atherosclerosis in apoE mice. Injection of oxidized LDL induced T-cell tolerance due to clonal deletion, rather than anergy of the reactive T cells. The T-cell repertoire of apoE KO mice was affected by the development of the disease, whereas tolerization normalized it.
Conclusions
This study demonstrates that the immune response against oxidized LDL has a deleterious role in atherogenesis and that a fine-tuning of this response could modify the course of the disease.




Similar content being viewed by others
References
Plump AS, Smith JD, Hayek T, et al. (1992). Severe hypercholesterolemia and atherosclerosis in apolipoprotein E-deficient mice created by homologous recombination in ES cells. Cell 71: 343–353.
Piedrahita JA, Zhang SH, Hagaman JR, Oliver PM, Maeda N. (1992). Generation of mice carrying a mutant apolipoprotein E gene inactivated by gene targeting in embryonic stem cells. Proc. Natl. Acad. Sci. U.S.A. 89: 4471–4475.
Zhou X, Stemme S, Hansson GK. (1996). Evidence for a local immune response in atherosclerosis. CD4+ T cells infiltrate lesions of apolipoprotein-E-deficient mice. Am. J. Path. 149: 359–366.
Stemme S, Holm J, Hansson GK. (1992). T lymphocytes in human atherosclerotic plaques are memory cells expressing CD45RO and the integrin VLA-1. Arterioscler. Thromb. 12: 206–211.
Stemme S, Faber B, Holm J, Wiklund O, Witzum JL, Hansson GK. (1995). T lymphocytes from human atherosclerotic plaques recognize oxidized low density lipoprotein. Proc. Natl. Acad. Sci. U.S.A. 92: 3893–3897.
Palinski W, Ord VA, Plump AS, Breslow JL, Steinberg D, Witztum JL. (1994). ApoE-deficient mice are a model of lipoprotein oxidation in atherogenesis. Demonstration of oxidation-specific epitopes in lesions and high titers of autoantibodies to malondialdehyde-lysine in serum. Arterioscler. Thromb. 14: 605–616.
Salonen JT, Ylä-Herttuala S, Yamamoto R, et al. (1992). Autoantibody against oxidised LDL and progression of carotid atherosclerosis. Lancet 339: 883–887.
Esterbauer H, Ramos P. (1996). Chemistry and pathophysiology of oxidation of LDL. Rev. Physiol. Biochem. Pharmacol. 127: 31–64.
Nicoletti A, Caligiuri G, Törnberg I, Kodama T, Stemme S, Hansson GK. (1998). The macrophage scavenger receptor type A directs modified proteins to antigen presentation. Eur. J. Immunol. 28: 1–10.
Nicoletti A, Kaveri S, Caligiuri G, Bariety J, Hansson GK. (1998). Immunoglobulin treatment reduces atherosclerosis in apo E knockout mice. J. Clin. Invest. 102: 910–918.
Forsthuber T, Yip HC, Lehmann PV. (1996). Induction of TH1 and TH2 immunity in neonatal mice. Science 271: 1728–1730.
Caligiuri G, Nicoletti A, Zhou X, Tornberg I, Hansson GK. (1999). Effects of sex and age on atherosclerosis and autoimmunity in apoE-deficient mice. Atherosclerosis 145: 301–308.
Palinski W, Ylä-Herttuala S, Rosenfeld ME, et al. (1990). Antisera and monoclonal antibodies specific for epitopes generated during oxidative modification of low density lipoprotein. Arteriosclerosis 10: 325–335.
Palinski W, Rosenfeld ME, Ylä-Herttuala S, et al. (1989). Low density lipoprotein undergoes oxidative modification in vivo. Proc. Natl. Acad. Sci. U.S.A. 86: 1372–1376.
Zhou X, Paulsson G, Stemme S, Hansson GK. (1998). Hypercholesterolemia is associated with a T helper (Th) 1/Th2 switch of the autoimmune response in atherosclerotic apo E-knockout mice. J. Clin. Invest. 101: 1717–1725.
Gascoigne NR, Chien Y, Becker DM, Kavaler J, Davis MM. (1984). Genomic organization and sequence of T-cell receptor beta-chain constant- and joining-region genes. Nature 310: 387–391.
Patten P, Yokota T, Rothbard J, Chien Y, Arai K, Davis MM. (1984). Structure, expression and divergence of T-cell receptor beta-chain variable regions. Nature 312: 40–46.
Barth RK, Kim BS, Lan NC, et al. (1985). The murine T-cell receptor uses a limited repertoire of expressed V beta gene segments. Nature 316: 517–523.
Behlke MA, Chou HS, Huppi K, Loh DY. (1986). Murine T-cell receptor mutants with deletions of beta-chain variable region genes. Proc. Natl. Acad. Sci. U.S.A. 83: 767–771.
Chou HS, Anderson SJ, Louie MC, et al. (1987). Tandem linkage and unusual RNA splicing of the T-cell receptor beta-chain variable-region genes. Proc. Natl. Acad. Sci. U.S.A. 84: 1992–1996.
Louie MC, Nelson CA, Loh DY. (1989). Identification and characterization of new murine T cell receptor beta chain variable region (V beta) genes. J. Exp. Med. 170: 1987–1998.
Pannetier C, Cochet M, Darche S, Casrouge A, Zoller M, Kourilsky P. (1993). The sizes of the CDR3 hypervariable regions of the murine T-cell receptor beta chains vary as a function of the recombined germ-line segments. Proc. Natl. Acad. Sci. U.S.A. 90: 4319–4323.
Koide Y, Kaidoh T, Yanagawa T, Yoshida TO. (1993). A comparative study on T cell receptor V beta gene usages: spleen cells from the non-obese diabetic (NOD) mouse and its non-diabetic sister strain, the ILI mouse, and infiltrating T cells into pancreata of NOD mice. Microbiol. Immunol. 37: 653–659.
Palinski W, Horkkö S, Miller E, et al. (1996). Cloning of monoclonal autoantibodies to epitopes of oxidized lipoproteins from apolipoprotein E-deficient mice. Demonstration of epitopes of oxidized low density lipoprotein in human plasma. J. Clin. Invest. 98: 800–814.
Beverly B, Kang SM, Lenardo MJ, Schwartz RH. (1992). Reversal of in vitro T cell clonal anergy by IL-2 stimulation. Int. Immunol. 4: 661–671.
Essery G, Feldmann M, Lamb JR. (1988). Inter-leukin-2 can prevent and reverse antigen-induced unresponsiveness in cloned human T lymphocytes. Immunology 64: 413–417.
Pannetier C, Even J, Kourilsky P. (1995). T-cell repertoire diversity and clonal expansions in normal and clinical samples. Immunol. Today 16: 176–181.
Acknowledgment
We thank Ingrid Törnberg, Kerstin Carlson, Inger Bodin, and Anita Larsson for excellent technical assistance. This work was supported by the Swedish Medical Research Council (proj. 6816) and Heart-Lung Foundation, the Johnson and Wallenberg Foundations, King Gustaf V 80th Anniversary Fund, Tore Nilsson Foundation, and the AFA Research Fund, A. N. was the recipient of a Marie Curie postdoctoral research fellowship from the European Union.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by H. Wigzell
Rights and permissions
About this article
Cite this article
Nicoletti, A., Paulsson, G., Caligiuri, G. et al. Induction of Neonatal Tolerance to Oxidized Lipoprotein Reduces Atherosclerosis In ApoE Knockout Mice. Mol Med 6, 283–290 (2000). https://doi.org/10.1007/BF03401937
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03401937