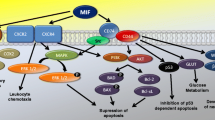
Abstract
Background
Macrophage migration inhibitory factor (MIF) was recently rediscovered as a cytokine, pituitary hormone, and glucocorticoid-induced immunomodulator. MIF is constitutively expressed in various cells and enhances production of inflammatory cytokines such as tumor necrosis factor-α, interleukin-1, and interferon γ. Recently, it was reported that MIF mRNA was overexpressed in prostatic tumors, which suggests that MIF is a protein involved in tumor cell growth beyond inflammatory and immune responses.
Materials and Methods
We examined the expression of MIF in the murine colon carcinoma cell line colon 26 by Western and Northern blot analyses and immunohistochemistry. Next, we investigated the effects of transforming growth factor (TGF) β, basic fibroblast growth factor (b-FGF), and platelet-derived growth factor (PDGF) on the expression of MIF mRNA. Furthermore, we examined whether MIF is involved in tumor cell proliferation, using an MIF anti-sense plasmid transfection technique.
Results
We demonstrated that MIF protein and its mRNA were highly expressed in colon 26 cells, using Western and Northern blot analyses, respectively. By immunohistochemical analysis, we found that MIF was localized largely in the cytoplasm of the tumor cells. In response to TGF-β, b-FGF, and PDGF, MIF mRNA expression was significantly up-regulated. Following this, we transfected the cells with an anti-sense MIF plasmid, which revealed that this treatment induced significant suppression of cell proliferation.
Conclusion
Although MIF plays multifunctional roles in a broad spectrum of pathophysiological states, little has been done to investigate the role of this protein in association with tumor growth. The current results suggest the possibility that MIF induces tumor cell growth in concert with other growth factors, which encouraged us to investigate a novel approach for tumor therapy using an anti-MIF antibody and an MIF anti-sense plasmid transfection technique.





Similar content being viewed by others
References
Anzano MA, Rieman D, Prichett W, Bowen-Pope DF, Grieg R. (1989) Growth factor production by human colon carcinoma cell lines. Cancer Res. 49: 2898–2904.
Calandra T, Bernhagen J, Mitchell RA, Mitchell J, Bucala R. (1994) The macrophage is an important and previously unrecognized source of macrophage migration inhibitory factor. J. Exp. Med. 179: 1895–1902.
Sakai M, Nishihira J, Hibiya Y, Koyama Y, Nishi S. (1994) Glutathione binding rat liver 13K protein is the homologue of the macrophage migration inhibitory factor. Biochem. Mol. Biol. Int. 33: 439–446.
Bacher M, Meinhardt A, Lan HY, et al. (1997) Macrophage migration inhibitory factor expression in experimentally induced endotoxemia. Am. J. Pathol. 150: 235–246.
Bucala R. (1996) MIF rediscovered: cytokine, pituitary hormone, and glucocorticoid-induced regulator of the immune response. FASEB J. 10: 1607–1613.
Wistow GJ, Shaughenessy MP, Lee DC, Hodin J, Zelenka PS. (1993) A macrophage migration inhibitory factor is expressed in the differentiating cells of the eye lens. Proc. Natl. Acad. Sci. U.S.A. 90: 1272–1275.
Shimizu T, Ohkawara A, Nishihira J, Sakamoto W. (1996) Identification of macrophage migration inhibitory factor (MIF) in human skin and its immunohistochemical localization. FEBS Lett. 381: 199–202.
Matsuda A, Tagawa Y, Matsuda H, Nishihira J. (1996) Identification and immunohistochemical localization of macrophage migration inhibitory factor in human cornea. FEBS Lett. 385: 225–228.
Lanahan A, Williams JB, Sanders LK, Nathans D. (1992) Growth factor-induced delayed early response genes. Mol. Cell. Biol. 12: 3919–3929.
Matsuda A, Tagawa Y, Matsuda H, Nishihira J. (1997) Expression of macrophage migration inhibitory factor in rat corneal wound healing. Invest. Ophthalmol. Vis. Sci. 38: 1555–1562.
Meyer-Siegler K, Hudson PB. (1996) Enhanced expression of macrophage migration inhibitory factor in prostatic adenocarcinoma metastases. Urology 48: 448–452.
Nishihira J, Kuriyama T, Sakai M, Nishi S, Ohki S, Hikichi K. (1995) The structure and physicochemical properties of rat liver macrophage migration inhibitory factor. Biochim. Biophys. Acta. 1247: 159–162.
Towbin H, Staehelin T, Gordon J. (1979) Electrophoretic transfer of protein from Polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc. Natl. Acad. Sci. U.S.A. 76: 4350–4354.
Schagger H, Von Jagow G. (1987) Tricine-sodium dodecyl sulfate-Polyacrylamide gel electrophoresis for the separation of proteins in the range from 1 to 100 kDa. Anal. Biochem. 166: 368–379.
Onodera S, Suzuki K, Matsuno T, Kaneda K, Kuriyama T, Nishihira J. (1997) Macrophage migration inhibitory factor induces phagocytosis of foreign particles by macrophages in autocrine and paracrine fashion. Immunology 92: 131–137.
Mitchell R, Bacher M, Bernhagen J, Pushkarshaya T, Seldin MF, Bucala R. (1995) Cloning and characterization of the gene for mouse macrophage migration inhibitory factor (MIF). J. Immunol. 154: 3863–3870.
Ohnishi Y, Sakamoto T, Fujii H, et al. (1997) Characterization of a liver metastatic variant of murine colon26 carcinoma cells. Tumor Biol. 18: 113–122.
Bloom BR, Bennett B. (1966) Mechanism of a reaction in vitro associated with delayed-type hypersensitivity. Science 153: 80–82.
David JR. (1966) Delayed hypersensitivity in vitro: its mediation by cell free substances forms by lymphoid cell-antigen interaction. Proc. Natl. Acad. Sci. U.S.A. 56: 72–77.
Weiser WY, Temple PA, Witek-Giannotti JS, Remold HG, Clark SC, David JR. (1989) Molecular cloning of a cDNA encoding a human macrophage migration inhibitory factor. Proc. Natl. Acad. Sci. U.S.A. 86: 7522–7526.
Bernhagen J, Calandra T, Mitchel RA, et al. (1993) MIF is the pituitary-derived cytokine that potentiates lethal endotoxaemia. Nature 365: 756–759.
Bacher M, Metz CN, Calandra T, et al. (1996) An essential regulatory role for macrophage migration inhibitory factor in T-cell activation. Proc. Natl. Acad. Sci. U.S.A. 93: 7849–7854.
Calandra T, Bernhagen J, Mets CN, et al. (1995) MIF as a glucocorticoid-induced modulator of cytokine production. Nature 377: 68–71.
Huang F, Newman E, Theodorescu D, Derbel RS, Friedman E. (1995) Transforming growth factor beta 1 (TGF beta 1) is an autocrine positive regulator of colon carcinoma U9 cells in vivo as shown by transfection of a TGF beta 1 antisense expression plasmid. Cell Growth Differ. 6: 1635–3642.
Hsu S, Huang F, Freiedman E. (1995) Platelet-derived growth factor-B increases colon cancer cell growth in vivo by a paracrine effect. J. Cell. Physiol. 165: 239–245.
Yamanaka Y, Friess H, Buchler M, et al. (1993) Overexpression of acidic and basic fibroblast growth factor in human pancreatic cancer correlates with advanced tumor stage. Cancer Res. 53: 5289–5296.
Acknowledgments
We are grateful to M. Suzuki and T. Kuriyama for their technical assistance, and to Y. Mizue of Sapporo Immunodiagnostic Laboratory for preparation of the anti-MIF antibody.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Takahashi, N., Nishihira, J., Sato, Y. et al. Involvement of Macrophage Migration Inhibitory Factor (MIF) in the Mechanism of Tumor Cell Growth. Mol Med 4, 707–714 (1998). https://doi.org/10.1007/BF03401765
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03401765