Abstract
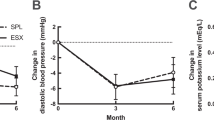
The pathogenesis of pseudohyperaldosteronism from licorice has been evaluated in 6 male volunteers taking daily 7 g of a commercial preparation of licorice for 7 days, corresponding to an intake of 500 mg/day of glycyrrhizic acid. Pseudohyperaldosteronism was evident during the treatment (increase of body weight, suppression of plasma renin activity and plasma aldosterone, reduction of serum potassium). The ratio (tetrahydro-cortisol+allo tetrahydrocortisol)/tetrahydrocortisone in urine increased in 5 cases after 3 days of treatment, without an increase of plasma mineralocorticoid activity (PMA). In the 6th case the urinary ratio was unchanged and PMA increased from the pretreatment value. After 7 days of therapy the ratio remained high and PMA was not measurable in 3 cases, while in the other 3 cases the ratio returned to pretreatment and PMA was higher than pretreatment value. We conclude that the pseudohyperaldosteronism from licorice is initially related to decreased activity of 11 μ-hydroxysteroid-dehydrogenase and afterwards also a direct effect of licorice derivatives on mineralocorticoid receptors becomes evident in some cases. In other cases however the effect on the enzyme is prevailing probably due to individual factors.
Similar content being viewed by others
References
Molhuysen J.A., Gerbandy J., De Vries L.A., De Jong J.C., Lenetra J.B., Turner K.P., Borst J.C.G. A licorice extract with desoxycorticosterone like action. Lancet 1: 381, 1950.
Ulmann A., Menard J., Corvol P. Binding of glycyrrhetinic acid to kidney mineralocorticoid and glucocorticoid receptors. Endocrinology 97: 46, 1975.
Armanini D., Karbowiak I., Funder J.W. Affinity of licorice derivatives for mineralocorticoid and glucocorticoid receptors. Clin. Endocrinol. (Oxf). 19: 606, 1983.
Armanini D., Karbowiak I., Krozowski Z., Funder J.W., Adam W.R. The mechanism of mineralocorticoid action of carbenoxolone. Endocrinology 111: 1683, 1982.
Armanini D., Wehling M., Weber P.C. Mineralocorticoid effector mechanism of licorice derivatives in human mononuclear leukocytes. J. Endocrinol. Invest. 12: 303, 1989.
Aterden L.M. Studies with glycyrrhetinic acid. Inhibition on metabolism of steroids in vitro. Biochem. J. 9: 75, 1958.
Tamura Y., Nishikawa T., Yamada K., Yamamoto M., Kumagai A. Effect of glycyrrhetinic acid and its derivatives on 4-Sulpha and 5 μ reductase in rat liver. Arzneimittelforschung 229: 47, 1979.
Latif S.A., Conca T.J., Morris D.J. The effect of the licorice derivative glycyrrhetinic acid,on hepatic 3 α- and 3 β-hydroxysteroid dehydrogenase and 5 α and 5 β-reductase pathways of metabolism of aldosterone in male rats Steroids 55: 52, 1990.
Stewart P.M., Wallace A.M., Valentino R., Burt D., Shackleton C.H., Edward C.R.W. Mineralocorticoid activity of licorice 11β hydroxysteroid dehydrogenase deficiency comes of age. Lancet 1: 82, 1987.
Farese R.W., Biglieri E.G., Shackleton C.H.L, Irony I., Gomez-Fontes R. Licorice-induced hypermineralocorticism. N. Engl. J. Med. 325: 1223, 1991.
Funder J.W., Pearce P.T., Smith R., Smith A. Mineralocorticoid action: target tissue specificity is enzyme, not receptor mediated. Science 242: 583, 1988.
Edwards C.R.W., Stewart P.M., Burt D., Brett L., McIntyre L., Sutanto W.S., de Kloet E.R., Monder C. Localization of 11β hydroxysteroid dehydrogenasetissue specific protector of the mineralocorticoid receptors. Lancet 2: 986, 1988.
Wilson R.C., Harbison M.D., Krozowski Z.S., Funder J.W., Shackleton C.H., Hanauske-Abel H.M., Wei J.Q., Hertecan J., Moran A., Neiberger R.E., Balfe J.W., Fattah A., Daneman D., Licholai T., New M. Several homozigous mutations in the gene for 11β-hydroxysteroid dehydrogenase type 2 in patients with apparent mineralocorticoid excess. J. Clin. Endocrinol. Metab. 80: 3145, 1995.
Bernardi M., D’Intino P., Trevisani F., Cantelli-Forti G.I., Raggi M.A., Turchetto E., Gasbarrini G. Effects of prolonged ingestion of graded doses of licorice by healthy volunteers. Life Science 55: 863, 1994.
Lan N.C., Matulich D.T., Stockigt J.R., Biglieri E.G., New M., Winter J.S.D., McKenzie J.K., Baxter J.D. Radioreceptor assay of plasma mineralocorticoid activity. Circ. Res. 46(suppl 1): 94, 1976.
Armanini D., Strasser T., Weber P.C. Characterization of aldosterone binding sites in human mononuclear leukocytes. Am. J. Physiol. 248: E388, 1985.
Ventura P., Visconti M., Pifferi G. A simple GLC. Analysis of ammonium glycyrrhizinate. Bollettino Chimico Farmaceutico 117: 211, 1978.
De Groot G., Koops R., Hogendorn E.A., Goewie C.E., Savelkoul T.J., Van Vloten P. Improvement of selectivity and sensitivity by column switching in the determination of glycyrrhizin and glycyrrhetinic acid in human plasma by high performance liquid chromatography. J. Chromatog. 456: 71, 1988.
Monder C., Shackleton C.H.L., Bradlow L., New M., Stoner E., Iohan F., Lakshmi V. The syndrome of apparent mineralocorticoid excess: its association with 11-β-hydroxysteroid-dehydrogenase and 5-β-reductase deficiency and some consequence for corticosteroid metabolism. J. Clin. Endocrinol. Metab. 63: 550, 1986.
Soro A., Ingram M.C., Tonolo G., Glorioso N., Fraser R. Evidence of coexisting changes in 11 hydroxysteroid dehydrogenase and 5-β-reductase activity in subjects with untreated essential hypertension. Hypertension 25: 67, 1995.
Armanini D., Witzgall H., Wehlig M., Kuhnle U., Weber P.C. Aldosterone receptors in different types of primary hyperaldosteronism. J. Clin. Endocrinol. Metab. 5: 101, 1987.
Armanini D., Zennaro M.C., Martella L., Pratesi C., Scali M., Zampollo V. Regulation of aldosterone receptors in hypertension. Steroids 58: 611, 1993.
Monder C., Shackleton C.H.L. 11-β-hydroxysteroid-dehydrogenase fact or fancy? Steroids 44: 383, 1986.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Armanini, D., Lewicka, S., Pratesi, C. et al. Further studies on the mechanism of the mineralocorticoid action of licorice in humans. J Endocrinol Invest 19, 624–629 (1996). https://doi.org/10.1007/BF03349029
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03349029