Abstract
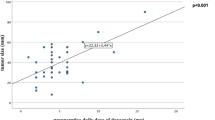
Pheochromocytoma resection is often complicated by intra-operative hypertension and post-resection hypotension. Factors associated with these hemodynamic alterations are not well defined. The aim of this study was to analyse the clinical-laboratory features associated with hemodynamic parameters during pheochromocytoma resection. Twenty-seven patients submitted to tumor resection — either open (no.=18) or video laparoscopic — between 1978–2007 were included. Nineteen received pre-operative α-blockers. Intra-operative hemodynamic data analysed were: maximum and minimum mean arterial blood pressure (MABP), no. of severe hypertensive (systolic BP >200 mmHg) and hypotensive episodes (MABP <60 mmHg), maximum and minimum heart rate (HR), no. of episodes of tachycardia and bradycardia, need to receive iv intra-operative treatment for hypertension and hypotension and the volume of fluids administered during surgery. Patients were 39.4±14.4-yr-old, 66% women. Intra-operative hemodynamic parameters were not different in patients submitted to open or video laparoscopic resection. Maximum intraoperative HR and the percentage of patients with HR>100 beats/min were higher in patients without pre-operative α - blocker treatment (no.=8). Pre-operative urinary vanylmandelic acid was positively associated with intra-operative maximum MABP (r=0.535, p=0.047) and with maximum transoperative systolic BP (r=0.805, p=0.016). Pre-operative urinary catecholamine (Pearson correlation r=0.575, p=0.03) and vanylmandelic acid (Pearson correlation r=0.605, p=0.04) levels were associated with maximum intra-operative MABP, adjusted for the presence of pheochromocytoma symptoms, surgical approach and pre-operative α-blockers. In conclusion, the degree of pre-operative catecholamine secretion was the most important aspect of transoperative BP control.
Similar content being viewed by others
References
Smythe GA, Edwards G, Graham P, Lazarus L. Biochemical diagnosis of pheochromocytoma by simultaneous measurement of urinary excretion of epinephrine and norepinephrine. Clin Chem 1992, 38: 486–92.
Stenström G, Svärdsudd K. Pheochromocytoma in Sweden 1958–1981. An analysis of the National Cancer Registry Data. Acta Med Scand 1986, 220: 225–32.
Pacak K, Linehan WM, Eisenhofer G, Walther MM, Goldstein DS. Recent advances in genetics, diagnosis, localization, and treatment of pheochromocytoma. Ann Intern Med 2001, 134: 315–29.
Orchard T, Grant CS, van Heerden JA, Weaver A. Pheochro-mocytoma—continuing evolution of surgical therapy. Surgery 1993, 114: 1153–8; discussion 8-9.
Plouin PF, Duclos JM, Soppelsa F, Boublil G, Chatellier G. Factors associated with perioperative morbidity and mortality in patients with pheochromocytoma: analysis of 165 operations at a single center. J Clin Endocrinol Metab 2001, 86: 1480–6.
Pacak K. Preoperative management of the pheochromocytoma patient. J Clin Endocrinol Metab 2007, 92: 4069–79.
Desmonts JM, Marty J. Anaesthetic management of patients with phaeochromocytoma. Br J Anaesth 1984, 56: 781–9.
Bravo EL, Tagle R. Pheochromocytoma: state-of-the-art and future prospects. Endocr Rev 2003, 24: 539–53.
Tauzin-Fin P, Sesay M, Gosse P, Ballanger P. Effects of perioperative alpha1 block on haemodynamic control during laparoscopic surgery for phaeochromocytoma. Br J Anaesth 2004, 92: 512–7.
van der Horst-Schrivers AN, Kerstens MN, Wolffenbuttel BH. Preoperative pharmacological management of phaeochromocytoma. Neth J Med 2006, 64: 290–5.
Newell KA, Prinz RA, Brooks MH, Glisson SN, Barbato AL, Freeark RJ. Plasma catecholamine changes during excision of pheochromocytoma. Surgery 1988, 104: 1064–73.
Eng C, Clayton D, Schuffenecker I, et al. The relationship between specific RET proto-oncogene mutations and disease phenotype in multiple endocrine neoplasia type 2. International RET mutation consortium analysis. JAMA 1996, 276: 1575–9.
Puñales MK, Graf H, Gross JL, Maia AL. RET codon 634 mutations in multiple endocrine neoplasia type 2: variable clinical features and clinical outcome. J Clin Endocrinol Metab 2003, 88: 2644–9.
Lenders JW, Pacak K, Walther MM, et al. Biochemical diagnosis of pheochromocytoma: which test is best? JAMA 2002, 287: 1427–34.
Sprung J, O’Hara JF Jr, Gill IS, Abdelmalak B, Sarnaik A, Bravo EL. Anesthetic aspects of laparoscopic and open adrenalectomy for pheochromocytoma. Urology 2000, 55: 339–43.
Kinney MA, Warner ME, vanHeerden JA, et al. Perianesthetic risks and outcomes of pheochromocytoma and paraganglioma resection. Anesth AnaIg 2000, 91: 1118–23.
Bravo EL, Tarazi RC, Gifford RW, Stewart BH. Circulating and urinary catecholamines in pheochromocytoma. Diagnostic and pathophysiologic implications. N Engl J Med 1979, 301: 682–6.
Gumbs AA, Gagner M. Laparoscopic adrenalectomy. Best Pract Res Clin Endocrinol Metab 2006, 20: 483–99.
Walz MK, Alesina PF, Wenger FA, et al. Laparoscopic and retroperitoneoscopic treatment of pheochromocytomas and retroperitoneal paragangliomas: results of 161 tumors in 126 patients. World J Surg 2006, 30: 899–908.
Amar L, Servais A, Gimenez-Roqueplo AP, Zinzindohoue F, Chatellier G, Plouin PF. Year of diagnosis, features at presentation, and risk of recurrence in patients with pheochromocytoma or secreting paraganglioma. J Clin Endocrinol Metab 2005, 90: 2110–6.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kramer, C.K., Leitão, C.B., Azevedo, M.J. et al. Degree of catecholamine hypersecretion is the most important determinant of intra-operative hemodynamic outcomes in pheochromocytoma. J Endocrinol Invest 32, 234–237 (2009). https://doi.org/10.1007/BF03346458
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03346458