Summary
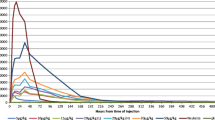
The biological potency of 2 recombinant human granulocyte colony-stimulating factors (rG-CSF) formulated for clinical use (lenograstim and filgrastim) together with a nonmarketed Escherichia coli-derived rG-CSF were compared by duplicate neutrophil colony assays in 3 normal human marrows. Serial doubling dilutions of each rG-CSF (128 to 0.008 ng/ml) were used. Neutrophil colonies were blindly scored at day 14. Lenograstim stimulated neutrophil colony formation at doses 16 times lower than that of the 2 nonglycosylated rG-CSFs and was twice as potent as filgrastim at maximal colony stimulation. Qualitative differences in colony size were also noted. Both nonglycosylated rG-CSFs (filgrastim and research-use rG-CSF) gave inferior qualitative and quantitative results, suggesting that glycosylation gives lenograstim a potency advantage.
Similar content being viewed by others
References
Dube S, Fisher JW, Powell JS. Glycosylation at specific sites of erythropoietin is essential for biosynthesis, secretion and biological function. Journal of Biological Chemistry 263: 17516–17521, 1988
Iscove NN. Culture of lymphocytes and haematopoietic cells in serum-free medium. In Barns et al. (Eds). Methods for serum-free culture of neuronal and lymphoid cells, pp. 169–185, Alan R. Liss, New York, 1984
Iscove NN, Guilbert LJ, Weyman C. Complete replacement of serum in primary cultures of erythropoietin-dependent red cell precursors (CFU-E) by albumin, transferrin, iron, unsaturated fatty acid, lecithin and cholesterol. Experimental Cell Research 126: 121–126, 1980
Kishita M, Motojima H, Oeda M, et al. Stability of granulocyte colony stimulating factor (G-CSF) in serum. Clinical Report 26: 221–224, 1992
Kubota N, Orita T, Hattori K, et al. Structural characterization of natural and recombinant human granulocyte colony-stimulation factors. Journal of Biochemistry 107: 486–492, 1990
Mancilla J, Schindler R, Dinarello CA. Importance of glycosylation for receptor binding and biological activity of IL-6. In Oppenheim et al. (Eds). Progress in Leucocyte Biology, Vol. 10A. Molecular and Cellular Biology of Cytokines; Second International Workshop on Cytokines, South Carolina, Dec 10–14 51–66. ISBN 0-471-56835-X, 1989
Oh-eda M, Hasegawa M, Hattori K, et al. O-linked sugar chain of human granulocyte colony stimulating factor protects it against polymerization and denaturation allowing it to retain its biological activity. Journal of Biological Chemistry 265: 11432–11435, 1990
Watari K, Asano S, Shirafuji N, et al. Serum granulocyte colony stimulating factor levels in healthy volunteers and patients with various disorders as estimated by enzyme immunoassay. Blood 73: 117–122, 1989
World Health Organization (WHO), BS/92.1711. International collaborative study for the proposed international Standards for Granulocyte macrophage-colony stimulating factor and granulocyte-colony stimulating factor. World Health Organization Technical Report Series, in press, 1994
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nissen, C., Dalle Carbonare, V. & Moser, Y. In Vitro Comparison of the Biological Potency of Glycosylated versus Nonglycosylated rG-CSF. Drug Invest 7, 346–352 (1994). https://doi.org/10.1007/BF03258477
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03258477