Abstract
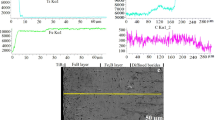
Stress corrosion cracking (SCC) is an established form of environmental attack in low karatage gold jewelry alloys. The cause of failure is often attributed to exposure to chlorinated solutions. At a certain gold content (usually greater than 14K) the alloy is widely believed protected from SCC. In this study, three commercial 18K gold alloys (yellow gold, nickel white gold, and palladium white gold) were tested in combination with three different household solutions to determine relative corrosion rates. These rates were determined using polarization tests. Once the maximum corrosion rate had been established, SCC tests using the constant potential dead weight method were used to determine time to failure. The resultant failure surfaces were examined to determine mode of fracture, which in all cases was predominantly intergranular. It was found that corrosion rates depended upon both the alloy system and the test solution. SCC was clearly demonstrated in 18K gold alloys, although in all cases times to failure were significantly greater than has been reported for lower karatage alloys. Palladium white gold was far more resistant to SCC than the other systems studied.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
7 References
R. Herrington, C. Stanley, and R. Symes, “Gold”, The Natural History Museum, London, 1999, p. 53
C.W. Corti,Gold Bull., 1999,32, 39
J.D. Fritz, B.W. Parks, H.W. Pickering,Scripta Metall 1988,22, 1063
I.A. Maier, S.A. Fernandez, J.R. Galvele,Corrosion Science 1995,37, 1
B.G. Ateya, H.W. Pickering,Corrosion Science 1996,38, 1245
L. Graf, Stress corrosion cracking in homogeneous alloys, inStress Corrosion Cracking and Embrittlement, edited by W.D. Robertson, Wiley, New York, 1956, pp. 48–60
J.S. Chen, M. Salmeron, T.M. Devine,Corrosion Science 1993,34, 2071
S.A. Serebrinsky, G.S. Duffo, and J.R. Galvele,Corrosion Science 1999,41, 191
W.S. Rapson and T. Groenewald,Gold Usage, Academic Press, London, 1978, pp. 57–64
G.S. Duffo, S.B. Farina, J.R. Galvele,Corrosion Science 2004,46, 1
G. Tammann,Z. Anorg. Allg. Chem. 1919,107, 1–240
M.G. Fontana, “Corrosion Engineering“, McGraw Hill, New York, 1986, pp. 172–173
F.L. LaQue, N.D. Greene, “Corrosion Basics“, NACE, Houston, TX, 1984, pp. 23–45
H.W. Pickering,Corrosion Science 1983,23, 1107
B.G. Ateya, G. Geh, A.H. Carim, H.W. Pickering,J. Electrochem. Soc. 2002,149, B27
J. Laurent, D. Landolt,Electrochem. Acta 1991,36, 49
J. Gniewek, J. Pezy, B.G. Baker, J. O’M. Bockris,J. Electrochem. Soc 1978,125, 17
U. Pittermann, R. Reining, K.G. Weil,J. Electrochem. Soc 1994,141, 3416
R. Pawlowych, D.L. Pile, H.W. Pickering, K.G. Weil,Physical Chemistry 1998,207, 113
Author information
Authors and Affiliations
Additional information
Colin C. Merriman is a graduate student in the School of Mechanical and Materials Engineering at Washington State University. He obtained a BS in Materials Science and Engineering from Washington State University in 2005. His thesis research is focused on understanding texture development in metals.
David F. Bahr is an Associate Professor in the School of Mechanical and Materials Engineering at Washington State University. He received the Bradley Stoughton Award for Young Teachers from ASM International in 2003 and is a recipient of a Presidential Early Career Award for Scientists and Engineers. Currently he serves as the Chair of the ASM/TMS Mechanical Behavior of Materials Committee and as a Key Reader forMetallurgical and Materials Transactions A. He was recently appointed to the ASM International Materials Reviews Committee.
M. Grant Norton is the Herman and Brita Lindholm Endowed Chair and Professor in the School of Mechanical and Materials Engineering at Washington State University and Associate Dean of Research and Graduate Programs in the College of Engineering and Architecture. During 1999/2000 he was a visiting professor at Oxford University. He has won several awards for teaching including the ASEE Outstanding Teaching Award for the Pacific Northwest. Currently, he serves as Editor ofJournal of Materials Science.
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License ( https://creativecommons.org/licenses/by-nc/2.0 ), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Merriman, C.C., Bahr, D.F. & Norton, M.G. Environmentally Induced Failure of Gold Jewelry Alloys. Gold Bull 38, 113–119 (2005). https://doi.org/10.1007/BF03215245
Issue Date:
DOI: https://doi.org/10.1007/BF03215245