Summary
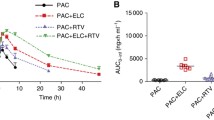
The MDR modulator, OC144-093, is a potential candidate for use in cancer therapy and exhibits potent biological activityin vitro andin vivo when combined with anticancer agents such as paclitaxel [1]. Its inhibitory interaction with P-glycoprotein (Pgp), the mdr1 gene product and a mechanistic participant in multidrug resistance [2], underlies its activity as a modulator of MDR. Having previously shown that OC144-093 is not a substrate for CYP3A [4] we first examined the effects of OC144-093 on paclitaxel metabolismin vitro. Using human liver microsomes, we have demonstrated that OC144-093 inhibited the CYP3A mediated metabolism of paclitaxel at high concentrations only (Ki=39.8±5.1 μM, n=3). Pharmacokinetic results also show that an oral dose of OC144-093, co-administered with paclitaxel caused negligible disturbance of the pharmacokinetic profile for paclitaxel when injected intravenously. In contrast, AUC values were elevated approximately 1.5-fold in all groups treated orally with paclitaxel and OC144-993. Cmax was enhanced approximately 2-fold in the co-dosed group. These characteristics are consistent with Pgp blockade in the gut enhancing oral bioavailability. Elimination properties of paclitaxel were affected only upon multiple dosing of OC144-093. These results warrant the further clinical assessment of OC144-093 as an MDR reversing agent.
Similar content being viewed by others
Abbreviations
- PgP:
-
P-glycoprotein
- MDR:
-
multidrug resistance
- HPLC:
-
high performance liquid chromatography
- AUC:
-
area under the curve
References
Newman MJ, Rodarte JC, Benbatoul KD, Romano SJ, Uyeda RT, Moran EJ, Dixon R, Guns ES and Mayer LD (2000) Discovery and Characterization of OC144-093, a novel inhibitor of Pgp mediated MDR. Cancer Research. 60(11):2964–72.
Bellamy William T (1996) P-glycoproteins and multidrug resistance. Annu. Rev. Pharmacol. Toxicol. 36:161–183.
Wacher VJ, Wu C-Y and Benet LZ (1995) Overlapping substrate specificities and tissue distribution of cytochrome P450 3A and P-glycoprotein: Implications for drug delivery and activity in cancer chemotherapy. Mol. Carcinogenesis. 13:129–134.
Guns ES, Bullock PL, Reimer MLJ, Dixon R and Mayer LD (2001) Assessment of the involvement of CYP3A in the in vitro metabolism of a new modulator of MDR in cancer chemotherapy, OC144-093, by human liver microsomes. Eur J Drug Metab Pharm 26(4):273–282.
Sonnichsen DS, Liu Q, Schuetz EG, Schuetz JD, Pappo A and Relling MV (1995). Variability in human cytochrome P450 paclitaxel metabolism. J Pharmacol Exp Ther. 275(2):566–75.
Endicott JA and Ling V (1989) The Biochemistry of P-Glycoprotein mediated multidrug resistance. Annu. Rev. Biochem. 58:137–71.
Cordon-Cardo C and O’Brien JP (1991) The multi-drug resistance phenotype in human cancer, in Important Advances in Oncology, (De Vita VT, Hellman S, Rosenberg SA) pp 19–38, Lippincott, Philadelphia.
Cordon CC, O’Brien JP, Casals D, Rittman-Grauer L., Biedler JL, Melamed MR and Bertino JR (1989) Multi-drug resistance gene (P-glycoprotein) is expressed by endothelial cells at blood-brain barrier sites. Proc. Natl. Acad. Sci. USA. 86:695–698.
Thiebaut F, Tsuruo T, Hamada H, Gottesman MM, Pastan I, Willingham MC (1989) Immunohistochemical localization in normal tissues of different epitopes in the multidrug transport protein P170: evidence for localization in brain capillaries and crossreactivity of one antibody with a muscle protein. J Histochem. 37:159–64.
Grogan TM, Spier CM, Salmon SE, Matzner M and Rybski J (1993) P-glycoprotein expression in human plasma cell myeloma: correlation with prior chemotherapy. Blood 81:490–95.
Drori S., Eytan, GD and Assaraf YG (1995) Potentiation of anticancer drug cytotoxicity by multidrug-resistance chemosensitizers involves alterations in membrane fluidity leading to increased membrane potential. Eur. J. Biochem. 228:1020–1029.
Pereira E, Teodori E, Dei D, Gualtieri F and Garnier-Suillerot A (1995) Reversal of multidrug resistance by verapamil analogues. Biochem. Pharmacol. 50(4):451–457.
Radel S, Bankusli I, Mayhew E and Rustum YM (1988) The effects of verapamil and a tiapamil analogue, DMDP, on adriamycin-induced cytotoxicity in P388 adriamycin-resistant and-sensitive leukemia in vitro and in vivo. Cancer Chemother. Pharmacol. 21:25–30.
Sikic BI, Fisher GA and Lum BL (1997) Modulation and prevention of multidrug resistance by inhibitors of P-glycoprotein. Cancer Chemother. Pharmacol. 40(Suppl):S13-S19.
Keller RP, Altermatt HJ, Nooter K, Poschmann G, Laissue JA, Bollinger P and Hiestand PC (1992) SDZ PSC833, a non-immunosuppressive cyclosporin: its potency in overcoming P-glycoprotein mediated multidrug resistance of murine leukemia. Int J. Cancer 50:593–597.
Erlichman C, Moore M, Thiessen JJ, Kerr IG, Walker S, Goodman P, Bjarnason G, Deangelis C. and Bunting P (1993) Phase I pharmacokinetic study of Cyclosporin A combined with Doxorubicin. Cancer Research. 53:4837–4842.
Kerr J, Graham J, Cummings J, Morrison JG, Thompson GG, Brodie MJ and Kaye SB (1986) The effect of verapamil on the pharmacokinetics of adriamycin. Cancer Chemother. Pharmacol. 18:239–242.
Zhou-Pan XR, Seree E, Zhou XJ, Placidi M, Maurel P, Barra Y and Rahmani R (1993) Involvement of Human Liver Cytochrome P450 3A in Vinblastine Metabolism: Drug Interactions. Cancer Research 53: 5121–5126.
Rushing DA, Raber SR, Rodvold KA, Piscitelli SC, Plank GS, Tewksbury DA (1994) The effects of Cyclosporine on the Pharmacokinetics of Doxorubicin in Patients with Small Cell Lung Cancer. Cancer 74:834–841.
Henricsson S, Lindholm A and Aravoglou M (1990) Cyclosporin metabolism in human liver microsomes and its inhibition by other drugs. Pharmacology and Toxicology 66:49–52.
Marre F, Sanderink G-J, De Sousa G, Gaillard C, Martinet M and Rahmani R (1996) Hepatic Biotransformations of Docetaxel (Taxotere”) in vitro: Involvement of the CYP3A Subfamily in Humans. Cancer Research 56:1296–1302.
Kivisto KT, Kroemer HK and Eichelbaum M (1995) The role of human cytochrome P450 enzymes in the metabolism of anticancer agents: implications for drug interactions. Br. J. Clin. Pharmacol. 40:523–530.
Van Asperen J, Van Tellingen O, Sparreboom A, Schinkel AH, Borst P, Nooijen WJ and Beijnen JH (1997) Enhanced oral bioavailability of paclitaxel in mice treated with the P-glycoprotein blocker SDZPSC 833. Br J Cancer. 76(9):1181–3.
Purba HS, Back DJ and Orme LLE (1987) Tolbutamide 4-hydroxylase activity of human liver microsomes: Effect of inhibitors. Br. J. Clin. Pharmacol. 24:230–234.
Desai PB, Duan JZ, Zhu YW and Kouzi S (1998) Human liver microsomal metabolism of paclitaxel and drug interactions. Eur J Drug Metab Pharmacokinet. 23(3):417–24.
Maurice M, Pitchard L, Daujat M, Fabre I, Joyeux H, Domergue J and Maurel P (1992) Effects of imidazole derivatives on cytochrome P-450 from human hepatocytes in primary culture. Fed. Am. Soc. Exp. Biol. 6:752–758.
Chang T, Benet LZ, and Hebert MF (1996) The effect of water-soluble vitamin E on cyclosporine pharmacokinetics in healthy volunteers. Clin. Pharmacol. Ther. 59:297–303.
Fischer V, Rodriguez-Gascon A, Heitz F, Tynes R, Hauck C, Cohen D, Vickers AE (1998) The multidrug resistance modulator valspodar (PSC 833) is metabolized by human cytochrome P450 3A. Implications for drug-drug interactions and pharmacological activity of the main metabolite. Drug Metab. Dispos. 26:802–11.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Guns, E.S., Denyssevych, T., Dixon, R. et al. Drug interaction studies between paclitaxel (Taxol) and OC144-093 — A new modulator of MDR in cancer chemotherapy. Eur. J. Drug Metab. Pharmacokinet. 27, 119–126 (2002). https://doi.org/10.1007/BF03190426
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03190426