Summary
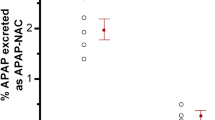
PhenacetinO-deethylation is a marker reaction of CYP450 1A2 activity. The drug-metabolizing enzyme is constitutively expressed in liver. In this study, an in vivo rat model for assessment of extrahepatic metabolism was used to investigate phenacetinO-deethylation and the alterations in the disposition of phenacetin due to the loss of liver function. Rats were divided into the model and normal control groups. The model was established according to our previously described method. The concentrations of phenacetin and its major metabolites acetaminophen, glucuronate-acetaminophen and sulfate-acetaminophen in plasma and urine were determined by HPLC. 30 min after intravenous administration of 0.16 % phenacetin 10 mg.kg−1, plasma acetaminophen in the model group was only 3.6 % of that in the control group (0.09±0.04 μg.mL−1 vs 2.49±0.85 μg.mL−1, n=8). 30 min after intragastric injection of 0.4 % phenacetin 30 mg.kg−1, plasma acetaminophen formation was very slight, about 8.6% of plasma phenacetin in the model group (0.74±0.43 μg.mL−1, acetaminophen vs 8.57±8.42 μg.mL−1 phenacetin) and 6.8% in the of plasma phenacetin in the model group (0.74±0.43 μg.mL−1 acetaminophen vs 8.57±8.42 μg.mL−1 phenacetin) and 6.8% in the control group (1.06±0.59 μg.mL−1 acetaminophen vs 15.47±7.21 μg.mL−1 phenacetin, n=8); no significant differences were observed in plasma phenacetin, total acetaminophen and the ratio of acetaminophen to phenacetin between control and model groups. In the urine collected for 3 h after intravenous administration of 0.16% phenacetin 10 mg.kg−1, the total receovery of acetaminophen (as free, glucuronate- and sulfate-acetaminophen) in the model group was 4.6% of that in the control group (4.47±4.27 μg vs 96.63±8.50 μg, n=6), but phenacetin recovery in the model group was 9 times higher than that in the control group (15.03±17.72 μg vs 1.66±0.50 μg). The results indicate that phenacetinO-deethylation in the extrahepatic tissues and the first-pass metabolism of the probe compound seem to be negligible in rats, but the renal excretion of phenacetin, as a compensation, dramatically increases in model rats.
Similar content being viewed by others
References
Peters, J. I., Iorishima, H., Ward, J. I., Coakley, C. J., Rimura, S. & Gonzalez, F. J. (1999): Role of CYP 1A2 in the toxicity of long-term phenacetin feeding in mice. Toxicol. Sci., 50, 82–89.
Bartoli, A., Xiaodong, S., Gatti, G., Cipolla, G., Marchiselli, R. & Prucca, E. (1996): The influence of ethnic factors and gender on CYP 1A2-mediated drug disposition: a comparative study in Caucasian and Chinese subjects using phenacetin as a marker substrate. Thr. Drug Monit., 18, 586–591.
Dong, S. X., Ping, Z. Z., Xiao, W. Z., Shu, C. C., Bartoli, A., Gatti, G., D’urso, S. & Perucca, E. (1998): Effect of active and passive cigarette smoking on CYP 1A2-mediated phenacetin disposition in Chinese subjects. Ther. Drug Monit., 20, 371–375.
Nelson D. R., Koymans, L. & Kamatahi, T. (1996): P450 superfamily: update on new sequences, gene mapping, accession numbers and nomenclature. Pharmacogenetics, 6, 1–42.
Kudo, S., Umehara, R., Hosokawa, M., Miyamoto, G., Chiba, K. & Satoh, T. (2000): Phenacetin deacetylase activity in human liver microsomes: distribution, kinetics, and chemical inhibition and stimulation. J. Pharmacol. Exp. Thr., 294, 80–88.
Tassaneeyakul, W., Birkett, D. J., Veronese, M. E., McManus, M. E., Tukey R. H., Quattrochi, L. C., Gelbion, H. V. & Miners, J. O. (1993): Specificity of substrate and inhibitor probes for human cytochromes P450 1A1 and 1A2. J. Pharmacol. Exp. Thr., 265, 401–407.
Venkatakrishnan K., Von Moltke L. L. & Greenblatt D. J. (1998): Human cytochromes P450 mediating phenacetin O-deethylation in vitro: validation of the high affinity component as an index of CYP 1A2 activity. J. Pharm. Sci., 87, 1502–1507.
Deshpande, V. S., Genter, M. B., Jung, C. & Desai, P. B. (1999): Characterization of lidocaine metabolism by rat nasal microsomes: implications for nasal drug delivery. Eur. J. Drug. Metab. Pharmacok., 24, 177–182.
Anderson, K. E., Hammons, G. J., Kadbubar, F. F., Potter, J. D., Kederlik, K. R., Ilett, K. F., Minchin, R. F., Teitel, C. H., Chou, H. C., Martin, M. V., Guengerich, F. P., Barone, G. W., Lang, N. P. & Perterson, L. A. (1997): Metabolic activation of aromatic amines by human pancreas. Carcinogenesis, 18, 1085–1092.
Haehner, B. D., Gorski, J. C., Vandenbranden, M., Wrighton, S. A., Janardan, S. K., Watkins, P. B. & Hall, S. D. (1996): Bimodel distribution of renal cytochrome P450 3A activity in human. Mole. Pharmacol., 50, 52–59.
HE Ping, CUI Zhenfu, XIO Shaoqing, LUO Ming, WANG Jian, ZHANG Guoqing, ZHO Lin, LI Linfang & WU Mengchao (2001): An in vivo rat model for assessment of extrahepatic metabolism. J. Pharmacol. Toxi. Meth., 45, 181–185.
Zotz, R. B., Von Schonfeld, J., Erhard, J., Bereuer, N., Lange, R., Beste, M., Goebell, H. & Eigler, F. W. (1997): Value of an extended monoethylglycinexylidide formation test and other dynamic liver function tests in liver transplant donors. Transplantation, 63, 538–54.
Elin R. J., Fried, M. W., Sampson, M. J., Rudde, M., Kleiner, D. E. & Dibisceglie, A. M. (1997): Assessment of monoethylglycinexylidide as measure of liver function for patients with chronic viral hapatitis. Clin. Chemistry, 43, 1952–1957.
Oh, S. Y., Lee, C. H. & Ku, Y. S. (2000): Pharmacokinetics and hepatoprotective effects of 2-methylaminoethyl-4,4′-dimethoxy-5,6,5′,6′-dimethylenedioxybiphenyl-2-carboxylic acid-2′-carboxylate monohydrochloride in rats with CCl4-induced acute hepatic failure. J. Pharm. Pharmacol., 52, 1099–1103.
HE Ping, WU Mengchao, LI Lingfang & GUO Yajun (1998): Effect of glycyrrhizin pretreatment on metabolism of antipyrine and acetaminophen in mice. Chinese Pharmacol. Bull., 14, 516–519.
Robert, M. N., Stein, L., Penno, M. B., Passanant, G. T. & Elliot, S. M. (1984): Sources of interindividual variations in acetaminophen and antipyrine metabolism. Clin. Pharmacol. Ther., 36, 417–430.
Robert, M. N., Stein, L., Penno, M. B., Passanant, G. T. & Elliot, S. M. (1984): Sources of interindividual variations in acetaminophen and antipyrine metabolism. Clin. Pharmacol. Ther., 36, 417–430.
CUI Zhenfu, ZHENG Guoqing, LUO Ming, WANG Hualiang, WANG Jian, YAN Zhenlin, HE Ping, QIAN Qijun, WU Mengchao & GUO Yaojun (2000): The metabolism of phenacetin in the rats with diethylnitrosamine-induced hepatoma. Chinese Pharmacol. Bull., 16, 202–204.
Garland, W. A., Hsiao, K. C., Pantuck, E. J. & Conney, A. H. (1977): Quantitative determination of phenacetin and its metabolite acetaminophen by GLC-chemical ionization mass spectrometry. J. Pharm. Sci., 66, 340–344.
Liu, L. & Klaassen, C.D. (1996): Different mechanism of saturation of acetaminophen sulfate conjugation in mice and rats. Toxi. Appl. Pharmacol., 139, 128–134.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cui, Z.F., He, P., Luo, M. et al. PhenacetinO-deethylation in extrahepatic tissues of rats. Eur. J. Drug Metab. Pharmacokinet. 27, 107–111 (2002). https://doi.org/10.1007/BF03190424
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03190424