Summary
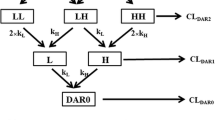
With the purpose of describing the MAb ior-R3’s kinetic behavior in disease state, this paper is focused on the study of this response using a human cancer (lung carcinoma cell line, H125) bearing nude mice animal model. This MAb was administered by a single 16 mg/Kg intravenous bolus dose and the blood samples were collected at several times ranging from 0 to 72 hours for serum drug quantification. The experimental data set was best fitted using a classical two-compartment mammilary pharmacokinetic (PK) model and the corresponding PK parameters were determined. Comparativel, the analysis of the more relevant physiologically-based PK parameters showed a significant enhancing of clearance as compound with the earlier reported study on healthy mice, increasing from 0.09 to 0.19 mL/h (p<0.01). However, the corresponding distribution volumes don’t seem to be altered by the tumor xenograft. We conclude that all of these evidences suggest a possible mechanism of receptor-mediated endocytosis (RME) as a major cause of this increased drug clearance which also contributed to the faster decrease of the drug disposition.
Similar content being viewed by others
References
Akaike, H. (1976): Statistical criteria for selecting a pharmacokinetic model. Math. Sci. 14: 5–11.
Blagoveshchenskaia, A.D., Sokolova, I., Kornilova, E., Nioliski, N.N. (1994): The dependence of the endocytosis of the EGFr on degree of receptor occupancy. Tsitologia. 36(7): 664–74.
Fernández, A., Pérez, R., Macías, A., Velanda, A., Alvarez, I., Ramos, M., Veloza, A. (1989): Generación y caracterización primaria de anticuerpos monoclonales contra el receptor del factor de crecimiento epidérmico. Interfer on y Biotecnología. 6, 289–298.
Garcia-Allan, C., Loughlin, J., Orton, T., Lord, P. (1997): Changes in protein and mRNA levels of EGF receptors in rat livers after administration of phenobarbitone or methylclofenapate. Arch. Toxicol. 71(7): 409–15.
Gibaldi, M., Perrier, D. (1982): Pharmacokinetics. Marcell Dekker Inc., New York.
Levy, G. (1988): Can Animal Models be Use to Explored Pharmacodynamics Problems of Clinical Interest? In: Topic in Pharmaceutical Sciences. Elsevier Sci. Publishers. BV (Biom. Div.), Sttugart, pp.
Levy, G. (1993): The case for preclinical pharmacodynamics. In: A. Yacobi et al. eds.), The integration of Pharmacokinetics, Pharmacodynamics and Toxicokinetics in rational drug development, by Plenum Press, New York, pp.
Moyer, J.D., Barbacci, E.G., Iwata, K.K., Arnold, L., Boman, B., Cunningham, A. (1997): Induction of apoptosis and cell cycle by CP 358–774, an inhibitor of EGF receptor tyrosine kinase. Cancer Res. 57 (21): 4838–48.
Opresko, C.K., Chang, C.P., Will, B., Burker, P., Gill, G., Wiley, H.S. (1995): Endocytosis of Lisosomal Targeting of EGFr are Mediated by Distinct Sequences Independent of the Tyrosine Kinase Domains. J. Biol. Chem. 270: 4325–4333.
Schlom, J., Horan-Hand, P., Geiner, J.W., Colcher, D., Shrivastav, S., Carrasquillo, J.A., Reynold, J.C., Larson, S.M., Raubitschek, A. (1990): Innovation that Influence the Pharmacology of Monoclonal antibody Guided Tumour Targetting. Cancer Research. 50, 820–827.
Stein, C.A., Khan, T.M., Khaled, Z., Tonkinson, J.I. (1995): Cell surface binding and cellular internalization properties of suramin, a novel antineoplastic agent. Clinical Cancer Research. 1: 509–517.
Fernández Sánchez E., Duconge J., Castillo R., Garcia I., Beausoleil I., Macias A. (2002): Monoclonal anti- egfr (iior r3) antibody’s pharmacokinetics studies on nude mice: a radio receptor analysis applied to serum drug quanntification. Journal of Pharmacy and Pharmacology, 54, January, 59–64.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Duconge, J., Fernández-Sánchez, E., Macías, A. et al. Monoclonal anti-EGFreceptor antibody (ior-R3) pharmacokinetic study in tumor bearing nude mice: Role of the receptor-mediated endocytosis on drug clearance. Eur. J. Drug Metab. Pharmacokinet. 27, 101–105 (2002). https://doi.org/10.1007/BF03190423
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03190423