Abstract
Four strains with high phenanthrene-degrading ability were isolated from petroleum badly polluted soil. The strainPseudomonas sp. ZJF08 demonstrated the highest rate of degradation (138. 1 mg·L−1·day−1) among them and degraded 97.1% of the phenanthrene in one week. The activities of two key enzymes of ZJF08, polycyclic aromatic hydrocarbon dioxygenase and catechol-2,3-oxygenase (C23O), were also assayed during the degradation of phenanthrene. Both of them reached their maximums on the 2nd day of degradation. The C23O gene (C7) ofPseudomonas sp. ZJF08 was cloned and expressed inEscherichia coli, and its gene product was purified by a Ni-NTA-agarose column. The optimum temperature for the purified C23O was 40°C at pH 7.5 and the C23O activity could be still detected when the temperature reached 70°C. The results showed that the C23O fromPseudomonas sp. strain ZJF08 exhibited better thermostability than its homologs reported.
Similar content being viewed by others
References
Boopathy R. (2000). Factors limiting bioremediation technologies. Bioresource Technology, 75: 63–67.
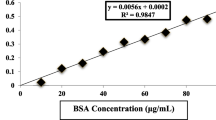
Bradford M.M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem., 72: 248–254.
Chen S.H., Aitken M.D. (1999). Salicylate stimulates the degradation of high-molecular weight polycyclic aromatic hydrocarbons byPseudomonas saccharophila P15. Environ. Sci. Technol., 33: 435–439.
Guieysse B., Viklund G., Toes A.C., Mattiasson B. (2004). Combined UV-biological degradation of PAHs. Chemosphere, 55: 1493–1499.
Hughes J.B., Beckles D.M., Chandra S.D., Ward C.H. (1997). Utilization of bioremediation processes for the treatment of PAH-contaminated sediments. J. Ind. Microbiol. Biotech., 18: 152–160.
Jacques R.J.S., Santos E.C., Bento F.M., Peralba M.C.R., Selbach P.A., Sá E.L.S., Camargo F.A.O. (2005). Anthracene biodegradation byPseudomonas sp. isolated from a petrochemical sludge landfarming site. Int. Biodeter. Biodeg., 56: 143–150.
Johnsen A.R., Wick L.Y., Harms L. (2005). Principles of microbial PAH-degradation in soil. Environ. Pollut., 133: 71–84.
Kahng H.Y., Nam K., Kukor J.J., Yoon B.J., Lee D.H., Oh D.C., Kam S.K., Oh K.H. (2002). PAH utilization byPseudomonas rhodesiae KK1 isolated from a former manufactured-gas plant site. Appl. Microbiol. Biotechnol., 60: 475–480.
Kanaly R.A., Haryama S. (2000). Biodegradation of high molecular-weight polycyclic aromatic hydrocarbons by bacteria. J. Bacteriol., 182: 2059–2067.
Kim D., Chae J-C., Jang J.Y., Zylstra G.J., Kim Y.M., Kang B.S., Kim E. (2005). Functional characterization and molecular modeling of methylcatechol 2,3-dioxygenase from o-xylene-degradingRhodococcus sp. strain DK17. Biochem. Biophys. Res. Comm., 326: 880–886.
Kiyohara H., Nagao K. (1978). The catabolism of phenanthrene and naphthalene by bacteria. J. Gen. Microbiol., 105: 69–75.
Kojima Y., Itada N., Hayaishi O. (1961). Metapyrocatechase: a new catechol-cleaving enzyme. J. Biol. Chem., 236: 2223–2228.
Langworthy D.E., Stapleton R.D., Sayler G.S., Findlay R.H. (2002). Lipid analysis of the response of a sedimentary microbial community to polycyclic aromatic hydrocarbons. Microb. Ecol., 43: 189–198.
Meyer S., Moser R., Neef A., Stahl U., Kämpfer P. (1999). Differential detection of key enzymes of polyaromatic-hydrocarbon-degrading bacteria using PCR and gene probes. Microbiology, 145: 1731–1741.
Moffett B.F., Walsh K.A., Harris J.A., Hill T.C.J. (2000). Analysis of bacterial community structure using 16S rDNA analysis. Anaerobe, 6: 129–131.
Nozaki M., Iwaki M., Nakai C., Saeki Y., Horiiki K., Kagamiyama H., Nakazawa T., Ebina Y., Inoue S., Nakazawa A. (1982). In: Nozaki M., Yamamoto S., Ishimura Y., Coon M.J., Ernster L., Estabrook R.W., Eds. Oxygenases and Oxygen Metabolism, Academic Press, New York, pp. 15–26.
Resnick S.M., Lee K., Gibson D.T. (1996). Diverse reactions catalyzed by naphthalene dioxygenase fromPseudomonas sp. strain NCIB 9816. J. Ind. Microbiol., 17: 438–457.
Samanta S.K., Singh O.V., Jain R.K. (2002). Polycyclic aromatic hydrocarbons: environmental pollution and bioremediation. Trends in Biotechnology, 20: 243–248.
Shamsuzzaman K.M., Barnsley E.A. (1974a). The regulation of naphthalene oxygenase inPseudomonads. J. Gen. Microbiol., 83: 165–170.
Shamsuzzaman K.M., Barnsley E.A. (1974b). The regulation of naphthalene metabolism inPseudomonads. Biochem. Biophys. Res. Comm., 60: 582–589.
Syakti A.D., Acquaviva M., Gliewicz M., Doumenq P., Bertrand J.C. (2004). Comparison ofn-eicosane and phenanthrene removal by pure and mixed cultures of two marine bacteria. Environ. Res., 96: 228–234.
Tao X.Q., Lu G.N., Dang Z., Yang C., Yi X.Y. (2007). Isolation of phenanthrene-degrading bacteria and characterization of phenanthrene metabolites. World J. Microbiol. Biotechnol., 23: 647–654.
Tian L., Ma P., Zhong J.J. (2002). Kinetics and key enzyme activities of phenanthrene degradation byPseudomonas mendocina. Process Biochemistry, 37: 1431–1437.
Wong J.W.C., Lai K.M., Wan C.K., Ma K.K., Fang M. (2002). Isolation and optimization of PAH degradative bacteria from contaminated soil for PAHs bioremediation. Water Air Soil Poll., 139: 1–3.
Zhang H., Kallimanis A., Koukkou A.I., Drainas C. (2004). Isolation and characterization of novel bacteria degrading polycyclic aromatic hydrocarbons from polluted Greek soils. Appl. Microbiol. Biotechnol., 65: 124–131.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Zhou, Y., Wei, J., Jiang, T. et al. Characterisation of a thermostable catechol-2,3-dioxygenase from phenanthrene-degradingPseudomonas sp. strain ZJF08. Ann. Microbiol. 57, 503–508 (2007). https://doi.org/10.1007/BF03175346
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03175346