Abstract
Purpose
To determine how the probability of fentanyl-induced cough is affected by patient characteristics and/or anesthetic technique.
Methods
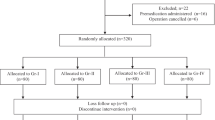
We analyzed data from a cohort of 1,311 adult patients undergoing elective surgery under general anesthesia, accompanied byiv fentanyl. The following data were collected: patient demographics, history of cigarette smoking, presence of bronchial asthma or chronic obstructive pulmonary disease, administration of angiotensin converting enzyme inhibitors; and anesthetic technique, including: preanesthetic anxiolytic medication, prior use of atropine, epidural lidocaine, a priming dose of vecuronium, and the dose ofiv fentanyl. Associations between individual variables in the clinical evaluation model and the likelihood of fentanyl-induced cough were characterized by calculating odds ratios. Multiple logistic regression analysis was used to examine the independent contribution of each variable while controlling for all variables.
Results
Fentanyl-induced cough was independently associated with the following: aging, cigarette smoking, a prior epidural injection of lidocaine, and a priming dose of vecuronium. Fentanyl-induced cough was unaffected by gender, the presence of either bronchial asthma or chronic obstructive pulmonary disease, or prior use of atropine.
Conclusions
Fentanyl-induced cough may be suppressed by aging, cigarette smoking, prior epidural injection of lidocaine, or a priming dose of vecuronium. These findings may allow insights into the mechanism of this phenomenon, thereby leading to its prevention.
Résumé
Objectif
Déterminer comment la probabilité de toux induite par le fentanyl est modifiée par les caractéristiques du patient et/ou la technique anesthésique.
Méthode
Nous avons analysé les données sur 1 311 patients adultes qui ont subi une opération réglée sous anesthésie générale accompagnée de fentanyl iv. Nous avons noté les données suivantes: les caractéristiques des patients, les antécédents de tabagisme, la présence d’asthme bronchique ou de maladie pulmonaire obstructive chronique, l’administration d’inhibiteurs de l’enzyme de conversion de l’angiotensine. La technique anesthésique comprenait: la médication préanesthésique anxiolytique, l’usage antérieur d’atropine, l’anesthésie péridurale avec lidocaïne, la dose d’amorçage de vécuronium et la dose iv de fentanyl. Les associations entre les variables individuelles dans le modèle d’évaluation clinique et la probabilité de toux induite par le fentanyl ont été définies par le calcul du risque relatif. Une analyse de régression logistique multifactorielle a servi à vérifier la contribution indépendante de chaque variable tout en contrôlant pour toutes les variables.
Résultats
La toux induite par le fentanyl a été indépendamment associée aux données suivantes: l’âge, le tabagisme, une injection péridurale antérieure de lidocaïne et une dose d’amorçage de vécuronium. La toux induite par le fentanyl n’était pas modifiée par le sexe du patient, l’asthme bronchique ou la maladie pulmonaire obstructive chronique ou l’usage antérieure d’atropine.
Conclusion
La toux induite par le fentanyl peut être supprimée par le vieillissement, le tabagisme, une injection péridurale antérieure de lidocaïne ou une dose d’amorçage de vécuronium. Ces résultats peuvent donner un aperçu du mécanisme de ce phénomène et conduire à sa prévention.
Article PDF
Similar content being viewed by others
References
Böhrer H, Fleischer F, Werning P. Tussive effect of a fentanyl bolus administered through a central venous catheter. Anaesthesia 1990; 45:18–21.
Phua WT, Teh BT, Jong W, Lee TL, Tweed WA. Tussive effect of a fentanyl bolus. Can J Anaesth 1991; 38:330–4.
Tweed WA, Dakin D. Explosive coughing after fentanyl injection. Anesth Analg 2001; 92:1442–3.
Agarwal A, Azim A, Ambesh S, et al. Sabutamol, beclomethasone or sodium chromoglycate suppress coughing induced by iv fentanyl. Can J Anesth 2003; 50:297–300.
Pandey CK, Raza M, Ranjan R, et al. Intravenous lidocaine suppresses fentanyl-induced coughing: a double-blind, prospective, randomized placebo-controlled study. Anesth Analg 2004; 99:1696–8.
Widdicombe JG. Neurophysiology of the cough reflex. Eur Respir J 1995; 8:1193–1202.
Israili ZH, Hall WD. Cough and angioneurotic edema associated with angiotensin-converting enzyme inhibitor therapy. A review of the literature and pathophysiology. Ann Intern Med 1992; 117:234–42.
Fuller RW, Dixon CM, Barnes PJ. Bronchoconstrictor response to inhaled capsaicin in humans. J Appl Physiol 1985; 58:1080–4.
Bickerman HA, Barach AL, Drimmer F. The experimental production of cough in human subjects induced by citric acid aerosols. Preliminary studies on the evaluation of antitussive agents. Am J Med Sci 1954; 228:156–63.
Sheppard D, Rizk NW, Boushey HA, Bethel RA. Mechanism of cough and bronchoconstriction induced by distilled water aerosol. Am Rev Respir Dis 1983; 127:691–4.
Pontoppidan H, Beecher HK. Progressive loss of protective reflexes in the airway with the advance of age. JAMA 1960; 174:2209–13.
Fujimura M, Kasahara K, Kamio Y, Naruse M, Hashimoto T, Matsuda T. Female gender as a determinant of cough threshold to inhaled capsaicin. Eur Respir J 1996; 9:1624–6.
Dicpinigaitis PV. Cough reflex sensitivity in cigarette smokers. Chest 2003; 123:685–8.
Christensen V, Ladegaard-Pedersen HJ, Skovsted P. Intravenous lidocaine as a suppressant of persistent cough caused by bronchoscopy. Acta Anaesthesiol Scand Suppl 1978; 67:84–6.
Dicpinigaitis PV, Grimm DR, Lesser M. Cough reflex sensitivity in subjects with cervical spinal cord injury. Am J Respir Crit Care Med 1999; 159:1660–2.
Irwin RS, Madison JM. The persistently troublesome cough. Am J Respir Crit Care Med 2002; 165:1469–74.
O’Connell F, Thomas VE, Fuller RW, Pride NB, Karlsson JA. Effect of clonidine on induced cough and bronchoconstriction in guinea pigs and healthy humans. J Appl Physiol 1994; 76:1082–7.
Dicpinigaitis, PV, Rauf K. The influence of gender on cough reflex sensitivity. Chest 1998; 113:1319–21.
Doherty MJ, Mister R, Pearson MG, Calverley PM. Capsaicin responsiveness and cough in asthma and chronic obstructive pulmonary disease. Thorax 2000; 55:643–9.
Karlsson JA, Sant’Ambrogio G, Widdicombe J. Afferent neural pathways in cough and reflex bronchoconstriction. J Appl Physiol 1988; 65:1007–23.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was supported in part by Grant-in-Aid 17591624 from Japan Society for the Promotion of Science.
Competing interests: None declared.
Rights and permissions
About this article
Cite this article
Oshima, T., Kasuya, Y., Okumura, Y. et al. Identification of independent risk factors for fentanyl-induced cough. Can J Anesth 53, 753–758 (2006). https://doi.org/10.1007/BF03022790
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03022790